 |
A 69-year-old Black male presented for a routine eye exam with a nebulous complaint of ocular discomfort and itching, OD>OS of one month’s duration. He reported no visual loss, pain or diplopia.
His pertinent medical history included reports of hypertension and gout. Current medications included clopidogrel (Plavix, Bristol-Myers Squibb), hydrochlorothiazide and aspirin. The patient denied allergies of any kind.
Diagnostic Data
Ocular examination revealed best-corrected visual acuities to be 20/20 OU through -1.00/+2.50 DS spectacles. External evaluation uncovered normal motilities and visual fields, normal color and stereo with no evidence of afferent pupillary defect. Refraction was negligibly different between eyes.
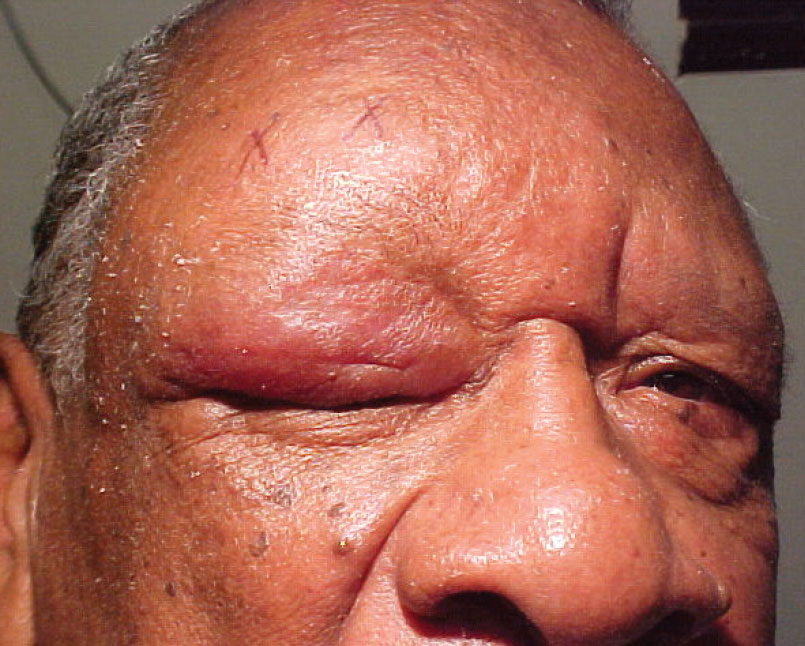
The pertinent anterior segment finding OD is demonstrated in the photograph.
Goldmann applanation tonometry was 15mm Hg. Dilated fundus examination revealed normal and quiet posterior segments with no peripheral pathology OU.
Additional Testing
 |
|
What seemed like a routine case of contact dermatitis actually turned out to be far less commonplace. Click image to enlarge. |
Other efforts might include palpation of the region to ensure the absence of cellulitis. Also, topical sodium fluorescein could be used to assess the corneal surface for damage and to evaluate the status of the lacrimal lake.
Back for More
The patient was diagnosed with acute contact dermatitis and allergic blepharoconjunctivitis OD. He was educated to remove possible triggers, use supportive measures (e.g., artificial tears, cold compresses) and was prescribed a topical mast cell stabilizing/antihistamine agent. He was scheduled to return for follow-up in five to 14 days.
The patient did not return for his scheduled follow-up but did come in within one month with a recurrent acute episode. His chief complaint at the return visit was worsening eyelid edema OD and onset of horizontal diplopia accompanied by constant mild pain behind the right eye.
Forced duction testing was positive OD, suggesting a restrictive etiology. Exophthalmometry (base 103) measured 25mm OD and 21mm OS. There was a 3mm ptosis OD produced by the weight of the edematous lid tissue. There was no afferent defect. Slit lamp exam confirmed upper lid edema and revealed possible lacrimal gland enlargement OD. Mild bulbar conjunctival injection OD was also observed. Posterior segment findings remained unchanged.
New Revelations
Upon further questioning, the patient revealed a history of cancer of an unknown type that had been in remission for 10 years. A call was made to the patient’s oncologist, who confirmed a history of stage IVa marginal zone lymphoma characterized by diffuse lymphadenopathy that was successfully treated with fludarabine-based combination therapy. His most recent bloodwork was done two months prior was and was interpreted as unremarkable.
Topical supportive dermatologic treatment was suggested to reduce discomfort from the adnexal swelling. Topical tear preparations were prescribed to increase lubrication of the ocular surface and reduce local symptoms. Based on suspicions of ocular involvement, the patient was referred for an immediate evaluation by his oncologic team.
The patient’s oncologist agreed with our findings and—because suspicion of orbital NHL was high—ordered an MRI of the brain and orbits. The MRI report revealed a soft tissue mass in the superior lateral aspect of the right orbit in the region of the lacrimal gland, consistent with lymphoma.
A biopsy confirmed a small B-cell neoplasm consistent with an extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT). The neoplasm was comprised of B-cells (90%) and T-cells (10%).
An immediate PET/CT scan and bone marrow biopsy was ordered by the oncology team for the purposes of assessing the status of all organ systems and restaging the cancer. The results of the imaging studies found that the disease was localized to the right orbit only.
The patient was treated with external beam radiation therapy. The prognosis is good and treatment is currently ongoing.
Non-Hodgkin’s Lymphoma
The non-Hodgkin’s lymphomas (NHL) are a heterogeneous group of B-cell and T-cell neoplasms that arise primarily in the lymph nodes. They can affect any organ of the body.1
The orbit is an uncommon primary site (i.e., site of first discovery) for NHL, accounting for <1% of primary presentations and 5-14% of all extranodal presentations.(2)12 It is the most common orbital malignancy; however, accounting for 24% of all cases.2,5
The most common types of lymphoma that occur in the orbit include MALT, follicular lymphoma, large B-cell lymphoma and mantle cell lymphoma.
Following diagnosis, a full staging workup is mandatory for to prep for treatment.21 Orbital lymphoma is typically treated with radiation therapy if the disease is localized and chemotherapy or combination therapy in cases demonstrating systemic spread.21
NHL results from uncontrolled proliferation of lymphocytes. 12 The neoplasms originate from B-lymphocytes (80%), T-lymphocytes (14%) or natural killer cells (6%).6 Neoplasms may also be a combination or “mixed” cell type.12
Many nomenclature systems have been developed over time to classify NHL. Examples of current classification systems include the Revised European-American Lymphoma classification and the World Health Organization classification of hematopoietic and lymphoid neoplasms, which designate dozens of distinct histologic entities.1
The prognosis for the disease varies depending upon its severity, from weeks to many years. Non-Hodgkin’s lymphomas are traditionally responsive to chemotherapy. However, over half of patients experience a recurrence.12 Recurrent lesions typically occur in extranodal sites, including the lung, salivary glands and the ocular adnexa.6
Ocular Involvement
Orbital lymphoma refers to a lesion occurring in the conjunctiva, lacrimal gland, eyelid and or ocular musculature.6 Orbital involvement in non-Hodgkin’s lymphoma is rare, accounting for only 1% of all NHL and 8% to 10% of extranodal NHL.6 Although the orbit is an uncommon site of involvement, it is the most common orbital malignancy, accounting for 24% of all cases.2,5
NHL can present at any time during the course of the disease. It has been documented to occur even in cases demonstrating prolonged remission.12 Orbital lymphomas can arise in the orbit or represent a manifestation of disseminated disease.6 Secondary orbital involvement may appear late after primary diagnosis or may be part of a generalized relapse. It may also represent the only active site of disease.2 The majority of orbital lymphomas are primary extranodal neoplasms. Secondary tumors in patients with disseminated lymphoma account for approximately 10% to 32% of cases.19
Nearly all orbital lymphomas are of B-cell phenotype. The most common subtype of primary orbital lymphoma is extranodal MALT (35% to 80% of cases). Follicular lymphoma (~20%), diffuse large B-cell lymphoma (~8%), and other histological subtypes such as mantle cell lymphoma, small lymphocytic lymphoma and lymphoplasmacytic lymphoma represent the other subtypes seen.17,19,21
Although orbital lymphoma can occur at any age, it most commonly occurs in adults age 50 to 70 years with increasing incidence in susceptible patients with advancing age. There is no gender predilection, although some studies show a slight female predominance.19,20 Most orbital lymphomas present unilaterally; however, bilateral cases are documented to occur in 10% of cases.6
The most common presentation is a palpable mass (64%), followed by eye irritation (28%), ptosis (20%), proptosis (18%), excessive tearing (16%), blurry vision (11%) and pain in the eye (3%).21 Other ocular signs and symptoms include diplopia, periorbital edema and increased intraocular pressure if orbital congestion causes decreased uveoscleral outflow.12
The differential diagnosis of orbital lymphoma includes inflammatory eye diseases such as thyroid associated orbitopathy and idiopathic orbital inflammatory syndrome, orbital infiltration, orbital infection and other orbital tumors.
Following identification of a mass lesion, the first step in the diagnosis of orbital lymphoma is a CT scan or MRI with contrast enhancement of the orbit. This is followed by a surgical biopsy of any detected mass. The imaging studies are used to aid in the assessment of a lesion’s location, size and degree of infiltration. They cannot provide a definitive diagnosis. A tissue biopsy is essential to confirm the diagnosis of NHL and also helps in staging. Once a biopsy has confirmed NHL, a systemic workup is indicated to rule out systemic spread and is vital for optimal treatment planning. The systemic workup includes complete blood count with differential and platelets, liver and kidney function tests, peripheral blood film, bone marrow biopsy, chest X-ray and CT of the orbit, chest and abdomen.19,21
The treatment of choice for the majority of patients with localized tumors is external beam irradiation. The modality has an average success rate of 97%.12 Despite this, frequent treatment-related complications and risk of recurrence are factors to be considered.19 Distant relapse occurs in about 20% of instances.12
Long-term ocular complications secondary to radiation treatment are observed in up to 50% of patients and include dry eye secondary to damage to the lacrimal and meibomian glands, cataract formation and radiation retinopathy. There is no universally accepted agreement with respect to the optimal radiation dose for successful tumor treatment with minimal complications due to the rarity of the disease.12,19
Disseminated orbital lymphoma is treated by chemotherapy.15 Complete responses are observed in 67% to 100% of patients. Local recurrence is the predominant cause of treatment failure, occurring in up to 29% of patients. Distant relapses may also occur.19
There is limited evidence regarding the efficacy of rituximab, a monoclonal chimeric anti-CD20 antibody, in patients with MALT lymphoma. It has recently been used for patients relapsing with lymphoma that expresses the specific CD20 antigen. Only a few case reports have been published confirming a high activity of rituximab. Even so, early recurrence is common. Further investigations with longer follow-up are needed to better define the role of rituximab in the management of orbital lymphoma. 19
Prognosis
The prognosis of patients with orbital lymphoma is generally favorable. A high percentage present with localized disease (stage I) with only 10% to 15% having disseminated disease (stage IV) at the time of initial diagnosis. Most patients have prolonged disease-free intervals and only about 10% of patients die from lymphoma. Negative prognostic factors include discovery at a non-conjunctival site, discovery at an advanced disease stage, nodal involvement, discovery at age 60 years or older and elevated serum lactate dehydrogenase (LDH) levels. 19
Despite the indolent course of extranodal marginal zone lymphoma, the lesions have a reputation for recurrence in extranodal sites, including the lung, salivary glands, as well as other ocular adnexal sites. Therefore, long-term follow-up with six-month examinations is recommended.4
Orbital involvement of non-Hodgkin’s lymphoma is a rare presentation but may occur at any time during the course of the disease. Orbital lymphoma can be an initial presentation of NHL, either solitary or systemic, or may represent dissemination of previously diagnosed lymphoma.17 If the disease is confined to the orbit, the prognosis for survival is good, and local treatment with radiotherapy is effective. In patients with systemic involvement, chemotherapy is required in addition to the orbital radiotherapy. If a patient with a history of NHL, even if in prolonged remission, presents with subtle ocular signs and symptoms such as lid edema, proptosis or diplopia, orbital lymphoma should be suspected.
Dr. Gurwood thanks Dr. Julie Marsh for contributing this case.
Dr. Gurwood is a professor of clinical sciences at The Eye Institute of the Pennsylvania College of Optometry at Salus University. He is a co-chief of Primary Care Suite 3. He is attending medical staff in the department of ophthalmology at Albert Einstein Medical Center, Philadelphia. He has no financial interests to disclose.
| 1. Alexander, Dominik D., Mink, Pamela J. et al. The non-Hodgkin lymphomas: A review of the epidemiologic literature. Int. J. Cancer 2007; 120:1-39. 2. Bairey, Osnat, MD, Kremer, Israel, MD, et al. Orbital and Adnexal Involvement in Systemic Non-Hodgkin’s Lymphoma. Cancer 1994; 73: 2395-99. 3. Bessell, E.M., Henk, J.M. et al. Ocular Morbidity After Radiotherapy of Orbital and Conjunctival Lymphoma. Eye 1987;1:90-96. 4. Coupland SE, Hummel M, Stein H. Ocular adnexal lymphomas: five case presentations and a review of the literature. Surv Ophthalmol. 2002;47(5):470-90. 5. Demirci H, Shields CL, Shields JA, Honavar SG, Mercado GJ, Tovilla JC. Orbital tumors in the older adult population. Ophthalmology 2002;109(2):243-8. 6. Essadi I, Tazy EM, et. al. Primary non Hodgkin’s lymphoma of the orbit: A case report. Medical Case Studies 2011; 2(2):19-21. 7. Evans, Linda S. and Hancock, Barry W. Non-Hodgkin lymphoma. Lancet. 2003; 362: 139-46. 8. Galieni, Piero, Polito, Ennio, et. al. Localized Orbital Lymphoma. Haematologica 1997;82:436-439. 9. Jenkins, C., Rose, GE. et. al. Clinical features associated with survival of patients with lymphoma of the ocular adnexa. Eye 2003;17:809-820. 10. Karcioglu, Zeynel A. Orbital Lymphoma in Orbital Tumors: Diagnosis and Treatment. Springer 2005: 133-140. 11. Liu, Grant, Volpe, Nicholas J., Galetta, Steven E. Orbital Disease in Neuro-Ophthalmology in Neuro-Ophthalmology Diagnosis and Management, Second Edition. Philadelphia, PA, Saunders/Elsevier, 2010: 611-652. 12. Lutz, Shannon, OD, Anderson, Sheila F., OD, et. al. Non-Hodgkin’s Orbital Lymphoma. Optometry and Vision Science 2001; 78(9):639-645. 13. Moslehi R, Devesa SS, Schairer C, Fraumeni JF Jr. Rapidly increasing incidence of ocular non-hodgkin lymphoma. J Natl Cancer Inst. 2006;98(13):936-9. 14. Ng, Eileen and Ilsen, Pauline F. Orbital Metastases. Optometry 2010: 81, 647-657. 15. Nutting, Christopher M., Jenkins, Christopher D. et. Al. The Hematology Journal. 2002; 3:14-16. 16. Roe, Richard H., MD, Finger, Paul T., MD, et. al. Whole-Body Positron Emission Tomography/Computed Tomography Imaging and Staging of Orbital Lymphoma. Ophthalmology 2006;113: 1854-58. 17. Rosado, Manuel F., Byrne, Gerald E. Jr. et al. Ocular adnexal lymphoma: a clinicopathologic study of a large cohort of patients with no evidence for an association with Chlamydia psittaci. Blood 2006;107:467-472. 18. Sjo, Lene Dissing. Ophthalmic Lymphoma: Epidemiology and Pathogenesis. Acta Ophthalmologica Thesis. 2009: 1-24. 19. Stefanovic, Alexandra and Lossos, Izidore S. Extranodal marginal zone lymphoma of the ocular adnexa. Blood 2009:114:501-510. 20. Tranfa, Fausto, DiMatteo, Germano, et. al. Primary Orbital Lymphoma. Orbit 2001;20:119-124. 21. Yadav, B S and Sharma, SC. Orbital lymphoma: Role of radiation. Indian Journal of Ophthalmology 2009;57(2):91-97. |

