History
A 27-year-old white male reported to the office with a chief
complaint of reduced vision in both eyes that had persisted for one month. Specifically,
the patient explained that he noticed progressively worsening cloudiness and
reduced visual clarity OU. His ocular and systemic histories were unremarkable.
He reported no known allergies.
Diagnostic Data
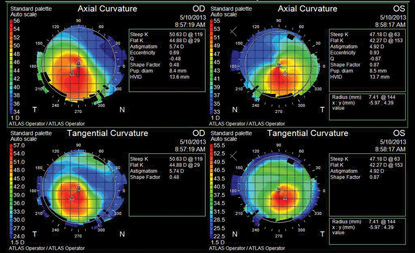 |
|
| Topography scan of our 27-year-old patient who complained of reduced vision OU.
|
|
Discussion
Additional testing included careful retinoscopy, indirect stereo-biomicroscopic examination of the fundus, color and brightness testing to rule out optic nerve involvement, Amsler grid testing, photodocumentation and optical coherence tomography of the fundus. The diagnosis in this issue is keratoconus––a disease of non-inflammatory bilateral corneal ectasia that progressively reduces patients’ visual acuity and quality of life.1-3 Clinical symptoms of keratoconus include a protruding cornea, with high amounts of irregular myopic astigmatism.1 Patients may present with a partial or complete iron deposit line surrounding the cone (Fleischer ring), and/or small folds of Descemet’s membrane and deep stroma (Vogt’s striae).1-4 It is important to note that apical corneal scaring also is possible, which can occur even in the absence of any contact lens use.4The incidence of keratoconus in the general American population is estimated to be one in 2,000 individuals, with an equal male-female predilction.1 Males are more likely to be diagnosed with keratoconus at a younger age than females.2 The cause of keratoconus is multifactorial in nature.4 Within normal corneal tissue, the stroma consists of a orthogonal lamellar matrix.3,6,7 However, in keratoconus patients, regions of highly aligned collagen intermixed with areas of poorly aligned collagen create an architectural anomaly.3,6,7
The structural alterations cause asymmetrical thinning of the cornea secondary to applied pressure (i.e., increased intraocular pressure, or as the result of mechanical eye rubbing in atopic disease).1,3 Lysyl oxidase (LOX) is responsible for biological crosslinking of collagens and elastin in a variety of tissues, including the central cornea.5,8 Recent studies suggest a strong link between keratoconus and a gene mutation at the locus 5q23.2, which codes for the LOX enzyme.9
Significant disruption of LOX regulation could yield decreased corneal rigidity and consequent keratoconic progression. Corneal crosslinking (CXL) with riboflavin is still being evaluated as a potential treatment for keratoconous and post-LASIK ectasia.10 In CXL, the treating clinician combines ultraviolet-A light pulses and liquid riboflavin drops to form additional covalent bonds between collagen molecules. This procedure increases corneal rigidity, which may slow or halt disease progression.
In the future, this may be combined with contact lens therapy. After we confirmed the diagnosis of keratoconus, we referred the patient for a specialty contact lens fitting. Topographical analysis revealed moderate focal steepening, with elevation located inferotemporally OU. Many systems can be used to halt disease progression. In this instance a standard Dyna Intra-Limbal 11.2mm diameter contact lens was used in both eyes. Applying the first definite apical clearance lens (FDACL) approach, the following parameters were indicated: 7.4/1 Flat/-4.00/11.2 OD and 7.78/ 1 Flat/-3.00/11.2 OS. After proper fitting, the contact lenses displayed lid attachment with adequate centering and 1mm of movement.
The fluorescein pattern revealed a central feathered touch with positive flush, mid-peripheral pooling and 1mm of edge lift––an optimal physiologic fit. The patient’s final best-corrected visual acuity measured 20/20 OU. Once the lens is fit and functioning properly, we will schedule him for a one-month follow-up. If his acuity remains stable, we’ll schedule another follow-up in three months. After that, longer follow-up periods may be sustainable.
Thanks to Chantel Garcia, OD, of Chevy Chase Md., and Todd Dimmick, OD, of Melbourne, Fla., for their contributions to this case.
1. Rabinowitz YS. Keratoconus. Surv Ophthalmol. 1998 Jan-Feb;42(4):297-319. 2. Léoni-Mesplié S, Mortemousque B, Mesplié N, et al. Epidemiological aspects of keratoconus in children. J Fr Ophtalmol. 2012 Oct;35(10):776-85. 3. Piñero DP, Alio JL, Barraquer RI, Michael R. Corneal biomechanical changes after intracorneal ring segment implantation in keratoconus. Cornea. 2012 May;31(5):491-9. 4. Wagner H, Barr JT, Zadnik K. Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study: methods and findings to date. Cont Lens Anterior Eye. 2007 Sep;30(4):223-32. 5. Sugar J, Macsai MS. What causes keratoconus? Cornea. 2012 Jun;31(6):716-9. 6. Meek KM, Tuft SJ, Huang Y, et al. Changes in collagen orientation and distribution in keratoconus corneas. Invest Ophthalmol Vis Sci. 2005 Jun;46(6):1948-56. 7. Daxer A, Fratzl P. Collagen fibril orientation in the human corneal stroma and its implication in keratoconus. Invest Ophthalmol Vis Sci. 1997 Jan;38(1):121-9. 8. Hamalainen ER, Jones TA, Sheer D, et al. Molecular cloning of human lysyl oxidase and assignment of the gene to chromosome 5q23.3-31.2. Genomics. 1991 Nov;11(3):508-16. 9. Bykhovskaya Y, Li X, Epifantseva I, et al. Variation in the lysyl oxidase (LOX) gene is associated with keratoconus in family-based and case-control studies. Invest Ophthalmol Vis Sci. 2012 Jun 28;53(7):4152-7. 10. Raiskup F, Spoerl E. Corneal crosslinking with riboflavin and ultraviolet A. Part II. Clinical indications and results. Ocul Surf. 2013 Apr;11(2):93-108.

