 |
Q:
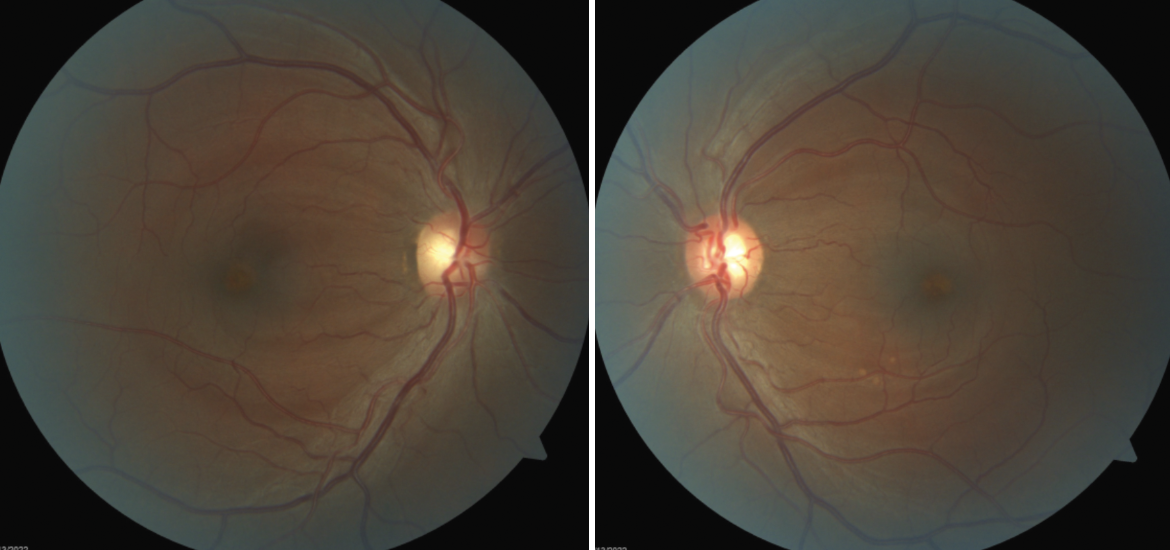
I have a 51-year-old African American male who presents with symmetrical bilateral macular lesions and 20/20 acuity. What could be causing them?
A:
“When we see bilateral macular lesions, our first thought is congenital dystrophy or age-related macular degeneration (AMD),” says Anney Joseph, OD, of Westchester Medical Group in Rye, NY. “However, we must also rule out infection and drug toxicity.”
Differentials
Pattern dystrophies are a group of dominantly inherited macular changes at the retinal pigment epithelium (RPE) level characterized by differing patterns of pigment deposits. These dystrophies occur due to various mutations in the human retinal degeneration slow/peripherin gene.1
Adult vitelliform macular dystrophy is an autosomal dominant disease that results in bilateral “egg-like” lesions in the macula. These lesions are made up of lipofuscin deposits in the RPE layer and usually manifest between 30 and 50 years of age. Visual potential is typically good and treatment is only warranted if choroidal neovascular membrane forms.2 Fluorescein angiography (FA) shows hypofluorescence in the area of the vitelliform lesion with a ring of hyperfluorescence that intensifies in the late phases. OCT reveals the hyperreflective accumulation below the sensory retina at the fovea and above the RPE as well as thinning of the overlying outer retinal layers.3 In late stage with progression (collapse of the vitelliform or the “scrambled egg” stage), disruption of the inner/outer segment junction layer interface and external limiting membrane will lead to visual impairment.
 |
|
Bilateral macular lesions can arise from myriad conditions that an OD must rule out. Click image to enlarge. |
Butterfly-shaped pattern dystrophy is a rare, bilateral condition characterized by a buildup of yellow, orange or gray pigmented material in a butterfly-shaped pattern within the RPE of the macula. The butterfly shape may also be described as linear, stellate, branching or shaped like a letter. FA will help differentiate this condition from others, Dr. Joseph says.
On FA, a large hypofluorescent, butterfly-shaped macular lesion will appear. OCT will show hyper-reflective material at the RPE level. The yellow or pigmented material is usually present in the second or third decade of life and patients are typically asymptomatic. However, the disease can progress with age, causing decreased visual acuity in a patient’s fifties and beyond.4
Multifocal pattern dystrophy simulating Stargardt’s disease or fundus flavimaculatus is characterized by irregular yellow-white flecks scattered throughout the posterior pole. The flecks are usually seen around the retinal vascular arcades as well as the macular area. Onset of this dystrophy is typically in the fifth decade and visual acuity is relatively good and stable. On FA, the flecks are hyperfluorescent in the early and late phase. To distinguish this from Stargardt’s, one would note the absence of the dark choroid on FA. OCT will show highly reflective focal thickening of the hyper-reflective outer red line.5
Fundus pulverulentus is the rarest of all the pattern dystrophies. It is characterized by coarse pigment mottling of the RPE layer in the macula. This dystrophy is commonly confused with AMD. On FA, the pigment mottling shows as hypofluorescent areas. This condition’s typical onset is in the fourth to fifth decade of life and visual acuity is relatively good and stable unless choroidal neovascularization forms.6
Consider Degeneration
AMD typically occurs in older adults when drusen accumulate in the RPE layer. These deposits can be viewed on OCT in between the RPE and Bruch’s membrane. It can be unilateral or bilateral and typically vision loss occurs in later stages (intermediate and exudative/wet).
“Prophylactic treatment, including vitamins, diet and UV protection, is indicated in early and intermediate stages,” Dr. Joseph says. “When choroidal neovascular membranes form, invasive treatment with intravitreal injection and/or laser is indicated.” OCT has become irreplaceable in early detection of choroidal neovascular membrane and subretinal fluid in exudative, or wet, AMD. OCT can also track patients’ response to treatment.
In younger patients with pigmented macular lesions, Dr. Joseph suggests considering TORCH syndrome, which refers to infection of a developing fetus or newborn by any of a group of infectious agents: toxoplasmosis, other agents (syphilis, parvovirus, varicella-zoster), rubella, cytomegalovirus or herpes virus.
“Given the patient’s HIV status, cytomegalovirus is a possibility, Dr. Joseph says. “However, CD4 count is not as low as we would see with cytomegalovirus retinitis.”
Beware of Medications
Dr. Joseph also takes note of the drugs the patient is currently taking. Of the listed medications, ritonavir has been reported to cause retinal toxicity. Ritonavir, an antiretroviral protease inhibitor used in the therapy and prevention of HIV and AIDS, can cause lesions in the macula, including retinal pigment epitheliopathy, macular telangiectasis and intraretinal crystal deposits, which cause vision loss as well as visual field defects.7 OCT will reveal thickened and irregular RPE layer with overlying loss of inner/outer segment junction layer. Visual acuity, function and retinal layers will improve after discontinuation of the medication.8
When thinking of medications that can cause bilateral retinal toxicity, Dr. Joseph believes one would be remiss not to mention hydroxychloroquine and the drugs known to contribute to talc retinopathy. Hydroxychloroquine toxicity’s early signs are macular edema and/or bilateral granular depigmentation of the RPE layer in the macula. With continued use, progression to an atrophic bull’s-eye maculopathy with concentric rings of hypo- and hyperpigmentation surrounding the fovea is observed clinically.
With further continued medication usage, widespread atrophy, as well as retinal arteriolar attenuation and optic disc pallor, may be noted. Risk of toxicity is dependent on the daily dose and duration of use. Screening recommendations include baseline fundus exam for pre-existing maculopathy and annual screening (including automated visual field and OCT) after five years for patients without major risk factors (high dose, long duration, concomitant renal disease or tamoxifen use).9
Talc is an insoluble filler material in oral medications such as methylphenidate but also seen in narcotics like heroin and cocaine. Talc retinopathy consists of refractile, irregularly shaped yellow deposits/crystals found inside small retinal vessels and within retinal layers. This retinopathy is seen in chronic intravenous drug users. OCT will show numerous highly reflective focal areas in the inner retina. Clinical findings can range from asymptomatic crystalline retinopathy to capillary nonperfusion and neovascularization.10,11
When you see a bilateral symmetrical macular pattern, be aware of all of these conditions. “Many can be simply watched, but when in doubt or if you suspect progression, get a friendly second opinion,” Dr. Joseph says.
Dr. Ajamian is the center director of Omni Eye Services of Atlanta. He currently serves as general chairman of the education committee for SECO International and is vice president of the Georgia State Board of Optometry. He has no financial interests to disclose.
1. Kohl S, Christ-Adler M, Apfelstedt-Sylla E, et al. RDS/peripherin gene mutations are frequent causes of central retinal dystrophies. J Med Genet. 1997;34(8):620-6. 2. Doan A, Stone EM. Best vitelliform macular dystrophy: 30-year-old female referred for bilateral macular lesions. EyeRounds. February 21, 2005. www.eyerounds.org/cases/case11.htm. Accessed May 31, 2022. 3. Benhamou N, Souied EH, Zolf R, et al. Adult-onset foveomacular vitelliform dystrophy: a study by optical coherence tomography. Am J Ophthalmol. 2003;135(3):362-7. 4. Deutman AF, van Blommestein JD, Henkes HE, et al. Butterfly-shaped pigment dystrophy of the fovea. Arch Ophthalmol. 1970;83(5):558-69. 5. Boon CJ, van Schooneveld MJ, den Hollander AI, et al. Mutations in the peripherin/RDS gene are an important cause cause of multifocal pattern dystrophy simulating STGD1/fundus flavimaculatus. Br J Ophthalmol. 2007;91(11):1504-11. 6. Parodi MB. Choroidal neovascularizaiton in fundus pulverulentus. Acta Ophthlmol Scand. 2002;80(5):559-60. 7. Roe RH, Jumper JM, Gualino V, et al. Retinal pigment epitheliopathy, macular telangiectasis, and intraretinal crystal deposits in HIV-positive patients receiving ritonavir. Retina. 2011;31(3):559-65. 8. Tu Y, Poblete RJ, Freilich BD, et al. Retinal toxicity with Ritonavir. Int J Ophthalmol. 2016;9(4):640-2. 9. Marmor MF, Kellner U, Lai TY, et al.; American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy. Ophthalmology. 2016;123(6):1386-94. 10. Shah VA, Cassell M, Poulose A, et al. Talc retinopathy.Ophthalmology. 2008;115(4):755-55.e2. 11. Soliman MK, Sarwar S, Hanout M, et al. High-resolution adaptive optics findings in talc retinopathy. Int J Retina Vitreous. 2015;1:10.1. |

