 |
A careful retinal examination is critical in order to evaluate each patient for various pathologies, some of which yield an underlying systemic cause. One of these potential retinal findings is the cotton wool spot (CWS). A CWS appears as a white and fluffy superficial lesion 0.1mm to 1.0mm in diameter that obscures the underlying retinal detail.1,2 This small but important finding can be a marker for potentially life-threatening conditions, making it of great clinical utility. To understand the health implications that cotton wool spots may carry, it is necessary to explore the process behind their formation.
Deep Dive
A CWS is thought to originate in cases where there is reduced perfusion, leading to focal or diffuse retinal ischemia. The retina is comprised of several kinds of neurons, including the retinal ganglion cells (RGCs), which are located within the superficial retinal nerve fiber layer (RNFL). In the ischemic retina, both orthograde and retrograde axonal transport of RGCs is disrupted. The CWS is a manifestation of this interruption of flow and is an accumulation of axoplasmic debris within the unmyelinated axons.1,3
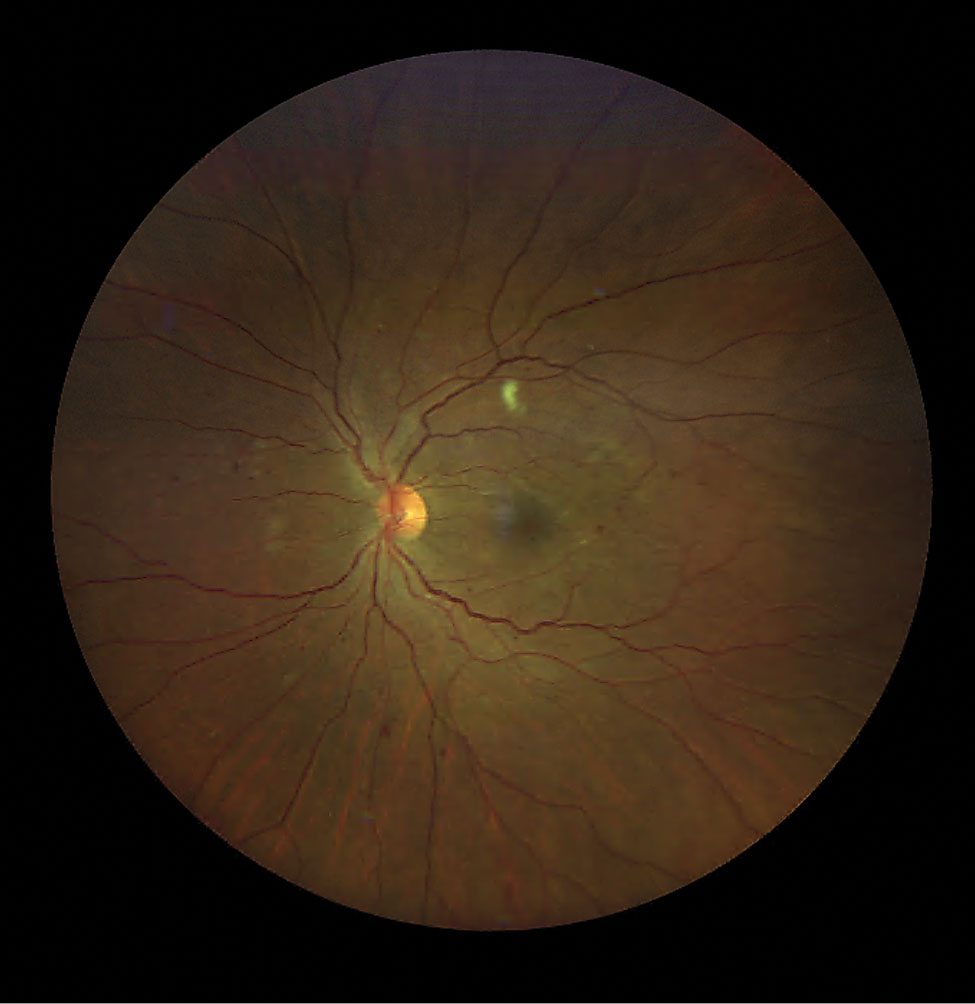
|
Diabetic retinopathy and a CWS along the superior temporal RNFL arcade. Click image to enlarge. |
The “cytoid body,” or end bulb of Cajal, is the histological landmark of the CWS and represents the terminal swelling of an RGC axon from the disruption of flow and hypoperfusion of the underlying retina.4 Because the RNFL thickens as we move toward the periphery of the retina, a cotton wool spot is most evident within the post-equatorial region.1 Although patients are typically asymptomatic, some reports indicate either local or arcuate scotomas at the site of the CWS.3
Besides the typical funduscopic appearance, a CWS can also be identified and monitored using optical coherence tomography (OCT). Since these lesions accumulate within the RNFL, the abnormality on OCT is typically found within this inner retinal layer. A scan over the area of the CWS will reveal marked thickening and hyper-reflectivity of the RNFL and inner neurosensory retina. Following clinical resolution of the CWS, retinal thinning may be evident in the corresponding area.1
In contrast to OCT, fluorescein angiography (FA) of a CWS manifests as a focal area of hypofluorescence that corresponds with the lesion. The reason for the darkened area is twofold. First, the presence of the CWS, to some extent, masks the underlying fluorescence. Second, the disruption of flow is presumed to be from an obstructed arteriole, in some cases, thus impairing normal vessel filling.4
DISTINGUISHING FEATURES OF THE TWO MOST COMMON CAUSES OF COTTON WOOL SPOTS | ||
| Hypertension | Diabetes | |
| Source of CWS Formation | arteriolar obstruction | capillary non-perfusion |
| Systemic Control | poor | mild to severe |
| Accompanying Retinal Findings | arteriolar narrowing, arteriovenous nicking, flame hemorrhages, microaneurysms, disc edema | microaneurysms, dot and blot hemorrhages, intraretinal microvascular abnormalities, neovascularization of the disc or retina, venous beading |
| Duration of CWS | eight to 12 weeks | up to 17 months |
Clinical Correlates
A CWS may present as a solitary retinal finding or be accompanied by additional pathology that can contribute to the overall clinical picture. In most cases, the finding is related to systemic disease.1-7 At first, it was believed to be solely of hypertensive origin; however, this was later refuted when diabetic patients without hypertension also presented with a CWS.
Furthermore, a CWS may be a retinal characteristic in patients with AIDS, anemia, collagen disease, bacterial endocarditis, radiation retinopathy, vascular occlusive disease, leukemia and systemic lupus erythematosus.1,2 While the systemic causes are many, the mechanism is the same, in that it is found predominantly in diseases where there are circulatory abnormalities.2
The two most common causes of a CWS, by far, are hypertension and diabetes. In hypertension, a CWS is often found in conjunction with a narrowed arteriovenous ratio, nicking, flame-shaped retinal hemorrhages and exudates. This stage of hypertensive retinopathy, stage three, signifies poor blood pressure control.5,6 In diabetic retinopathy, however, a cotton wool spot may be present even in milder forms and accompanied by dot and blot hemorrhages, microaneurysms, lipid exudates, edema and neovascularization.2,7
Though many of the retinal findings in these two etiologies overlap, the clinical course of a cotton wool spot differs in each. In hypertensive retinopathy, CWSs appear initially as a gray discoloration, days before developing their classic white and fluffy appearance. Studies that used FA demonstrated that nearby arterioles are leaky right before the CWS is evident, triggering a surrounding ring of capillary aneurysm formation during the course of the CWS. The arteriolar occlusion is the source of this ischemic cascade. However, following tight hypertensive control, resolution occurs within eight to 12 weeks. At this stage, the capillary bed returns to normal and few microaneurysms persist.2
In contrast, a CWS arising from diabetic retinopathy exhibits a similar clinical appearance but follows a different course. Capillary disease, especially microaneurysms, is the primary source of dysfunction triggering the ischemic events that lead to CWS formation. Since the capillary bed takes longer to recover, a CWS secondary to diabetes may last up to 17 months. This persistence may be attributed to the source of CWS clearance: macrophages. Macrophages are responsible for the clearing of cellular debris and arrive via the capillary circulation. As such, direct impairment to this transport system inhibits timely clearance, even with tight glucose control.2
Take-home Message
A CWS is a common retinal manifestation of many systemic diseases that affect circulation. Some, or many, of these conditions are life-threatening, necessitating prompt diagnosis and workup, or referral when applicable. The ability to identify this finding and its cause is an imperative skill, facilitated by the understanding of many of these aforementioned characteristic features.
Dr. Labib graduated from Pennsylvania College of Optometry, where she now works as an associate professor. She completed her residency in primary care/ocular disease and is a fellow of the American Academy of Optometry and a diplomate in the Comprehensive Eye Care section. She has no financial interests to disclose.
1. Gohari M, Soleimani AR, Manaviat MR, et al. Single cotton wool spot as a late manifestation of head trauma. J Ophthalmic Vis Res. 2014;9(2):272-5. 2. Dollery CT. Microcirculatory changes and the cotton-wool spot. Proc R Soc Med. 1969;62(12):1267-9. 3. Chui TY, Thibos LN, Bradley A, et al. The mechanisms of vision loss associated with a cotton wool spot. Vision Res. 2009;49(23):2826-34. 4. McLeod D. Why cotton wool spots should not be regarded as retinal nerve fibre layer infarcts. Br J Ophthalmol. 2005;89(2):229-37. 5. Modi P, Arsiwalla T. Hypertensive Retinopathy. Treasure Island (FL): StatPearls Publishing; 2020. 6. Tsukikawa M, Stacey AW. A review of hypertensive retinopathy and chorioretinopathy. Clin Optom (Auckl). 2020;12:67-73. 7. Lechner J, O’Leary OE, Stitt AW. The pathology associated with diabetic retinopathy. Vision Res. 2017;139:7-14. |

