 |
A 56-year-old Hispanic female presented with new-onset hazy vision and floaters in the left eye for the past week. She has had similar episodes in that eye before; the last one occurred six years earlier. In fact, she lost central vision in the left eye almost 20 years ago when the problem first started. The right eye was good, and she reported no problems. She reported being in good health and does not take any medications.
Upon examination, her best-corrected visual acuity was 20/20 OD and 20/400 OS with eccentric viewing. Confrontation visual fields were full-to-careful finger counting OU, but she did have a central scotoma in the left eye. Pupils were equal, round and reactive to light, but there was a trace afferent pupillary defect in the left eye. The anterior segment exam was unremarkable, and there was no cell or flare in either eye.
On dilated fundus exam, the posterior pole of the right eye was unremarkable. She did have a significant finding superonasally, which can be seen in the fundus photos provided. The view in the left eye was hazy. Other changes are seen in Figure 1.
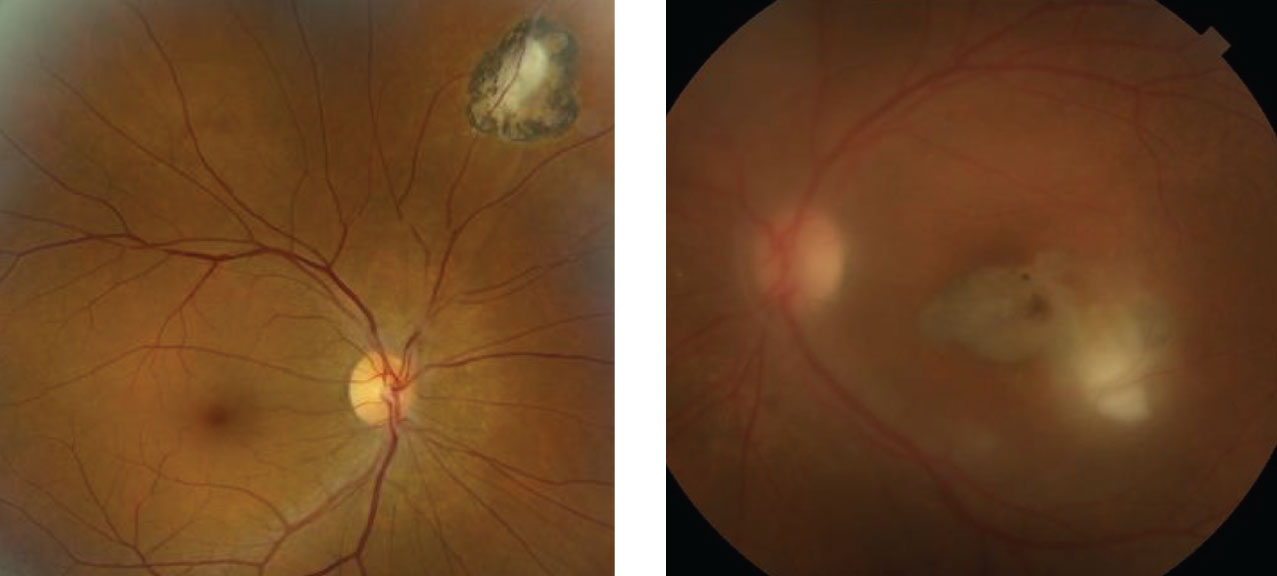 |
| Fig. 1. These are the right and left eyes of our patient. What does the hazy view in the left eye represent? Click image to enlarge. |
Take the Retina Quiz
1. How do you account for the hazy view into the left eye?
a. Anterior chamber inflammation
b. Cataract
c. Vitreous cells
d. Just a poor photograph
2. What does the white lesion adjacent to the macula represent?
a. Active retinochoroiditis
b. Placoid lesion from syphilis
c. Cotton wool spot
d. Fungal lesion
3. What is the correct diagnosis for the fundus lesion?
a. Toxocara canis
b. Toxoplasmosis
c. Active syphilis
d. Histoplasmosis
4. What is not a medication used to treat this condition?
a. Clindamycin PO
b. Azithromycin PO
c. Penicillin
d. Primethamine
5. Which statement best characterizes her condition?
a. Likely acquired
b. Likely congenital
c. Autoimmune
d. Reactivation
For answers, see below.
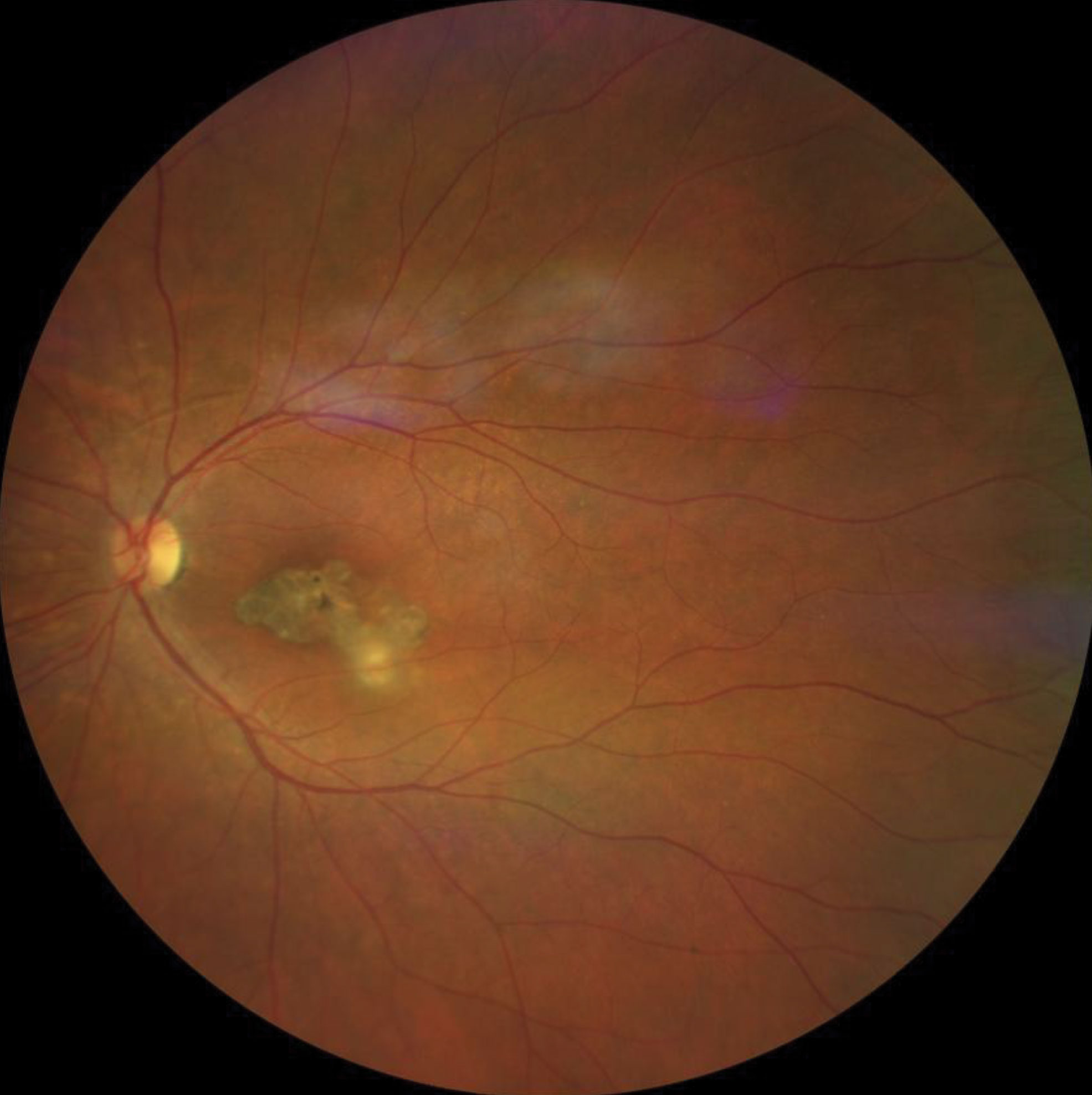 |
| Fig. 2. This patient three months later. Click image to enlarge. |
Discussion
The reason we have such a hazy view into the retina of the left eye is because the patient has significant vitreous cells that obscure the view. We can see chorioretinal scarring involving the macula of the left eye, which explains why her acuity is 20/400. The presence of the macula scar in the left eye in addition to the peripheral scar in the right eye should automatically make you suspicious for toxoplasmosis.
Unfortunately, it appears she has reactivation. In addition to her onset of floaters, there is a fluffy white lesion adjacent to the macular scar, which represents an area of active retinochoroiditis. This is classic for reactivation of the toxoplasmosis.
Toxoplasma gondii is a widespread parasite that infects almost all mammals and birds across the world. It is estimated that 25% to 30% of the human population is infected with Toxoplasma gondii.1,2 It is considered to be the most common cause of posterior uveitis and accounts for approximately 90% of focal necrotizing retinitis.
Human infection can occur from a variety of causes, including ingestion of oocysts shed in the feces of cats (from contaminated soils or litter boxes) or ingestion of the tissue cysts (bradyzoites) in infected meat.1,2 Other forms of exposure include consuming unwashed vegetables and drinking contaminated water or unpasteurized bovine milk. Our patient laughed when we asked about consumption of undercooked meat. She said her family has always eaten their meat well-done—almost like shoe leather.
Congenital transmission of toxoplasmosis is quite common. Intrauterine transmission occurs in about one-third of pregnancies of women infected with toxoplasmosis.3 In the United States, congenital toxoplasmosis occurs in about 1/1000 to 10,000 live births.3 It generally presents in the eye as an inactive choroiretinal scar. The presence of bilateral macular scars is considered to be the hallmark of congenital toxoplamosis.
Reactivation of a congenital infection can occur. When this happens, reactivation occurs adjacent to an old scar, which was the case with our patient.
In a majority of cases, the diagnosis of toxoplasmosis, congenital or acquired, is based on the clinical presentation, and a serologic work-up is not required. In atypical cases, or when the diagnosis is in question, perform serologic blood studies, such as the Toxoplasma ELISA, to confirm the diagnosis. Detection of IgM antibody titers present within the first two weeks of infection and suggest a recently acquired infection. Positive IgG antibodies indicated the patient was exposed to the infection some time in their lifetime. IgG antibodies peak at two months and will be present for life.
Treatment
Toxoplasmosis retinochoroiditis will generally resolve on its own in four to eight weeks in immunocompetent patients. Patients with peripheral lesions that are not a threat to the macula or optic nerve are often observed without treatment, unless there is a severe vitritis that causes a significant reduction in visual acuity. Treatment is recommended for active lesions that threaten or involve the macula or optic nerve, though many studies have shown there is no significant difference in the visual outcome between treated eyes and observation in immunocompetent patients.
There seems to be no clear consensus among uveitis specialists on treating patients with active disease. The “classic therapy” for treating active toxoplasmosis retinochoroditis is oral pyrimethamine, (loading dose of 75mg to 100mg during the first day, followed by 25mg to 50mg on subsequent days), sulfadiazine (loading dose of 2g to 4g during the first 24 hours, followed by 1g QID and oral prednisone 40mg to 60mg per day, also referred to as triple therapy).4,5 Folinic acid is also given 3mg to 5mg twice a week to avoid developing thrombocytopenia and leucopenia.
Other medications that have been used are clindamycin 30mg PO TID and Bactrim DS DS (sulfamethoxazole-trimethoprim, Roche Pharmaceuticals) PO BID, combined with prednisolone 40mg PO QD. Some uveitis specialists will also give an intravitreal injection of dexamethasone 0.4mg/clindamycin 1mg.4,5 Azithromycin has also emerged as an alternative treatment for toxoplasmosis. Studies comparing traditional triple therapy with azithromycin have shown no significant difference in results.6
When our patient presented with reactivation of her toxoplasmosis, she already had a long and tumultuous history that started in 2000. The initial episode started in her left macula, but it was small, and with treatment, she recovered to 20/50. A toxoplasma ELISA was performed on that initial exam and came back positive.
She was stable until 2013, when she had another reactivation, and that episode robbed her of her central vision. She was stable until 2019, when a third reactivation occurred. Figure 2 represents how the patient looked three months after treatment. She has remained stable since her last episode.
Retina Quiz Answers
1) c; 2) a; 3) b; 4) c; 5) d.
| 1. Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965-76. 2. Stokkermans TJ, Havens SJ. Toxoplasma retinochoroiditis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing. Updated Jun 27, 2020. www.ncbi.nlm.nih.gov/books/NBK493182. Accessed October 12, 2020. 3. El Bissati K, Levigne P, Lykins J, et al. Global initiative for congenital toxoplasmosis: an observational and international comparative clinical analysis. Emerg Microbes Infect. 2018;27;7(1):165. 4. Harrell M, Carvounis PE. Current treatment of toxoplasma retinochoroiditis: an evidence-based review. J Ophthalmol. 2014;2014:273506. 5. Kim SJ, Scott IU, Brown GC, et al, Interventions for toxoplasma retinochoroiditis: a report by the American Academy of Ophthalmology. Ophthalmology. 2013;120(2):371-8 6. Bosch-Driessen LH, Verbraak FD, Suttorp-Schulten MS, et al. A prospective, randomized trial of pyrimethamine and azithromycin vs. pyrimethamine and sulfadiazine for the treatment of ocular toxoplasmosis. Am J Ophthalmol. 2002;134:34-40. |

