 |
A 34-year-old man presented to the office with a chief complaint of a sudden-onset redness affecting one eye. He said he was not aware of it until someone pointed out to him that “his eye was bleeding.”
He did not report any pain or vision loss. He also denied trauma, systemic disease or allergies of any kind.
Diagnostic Data
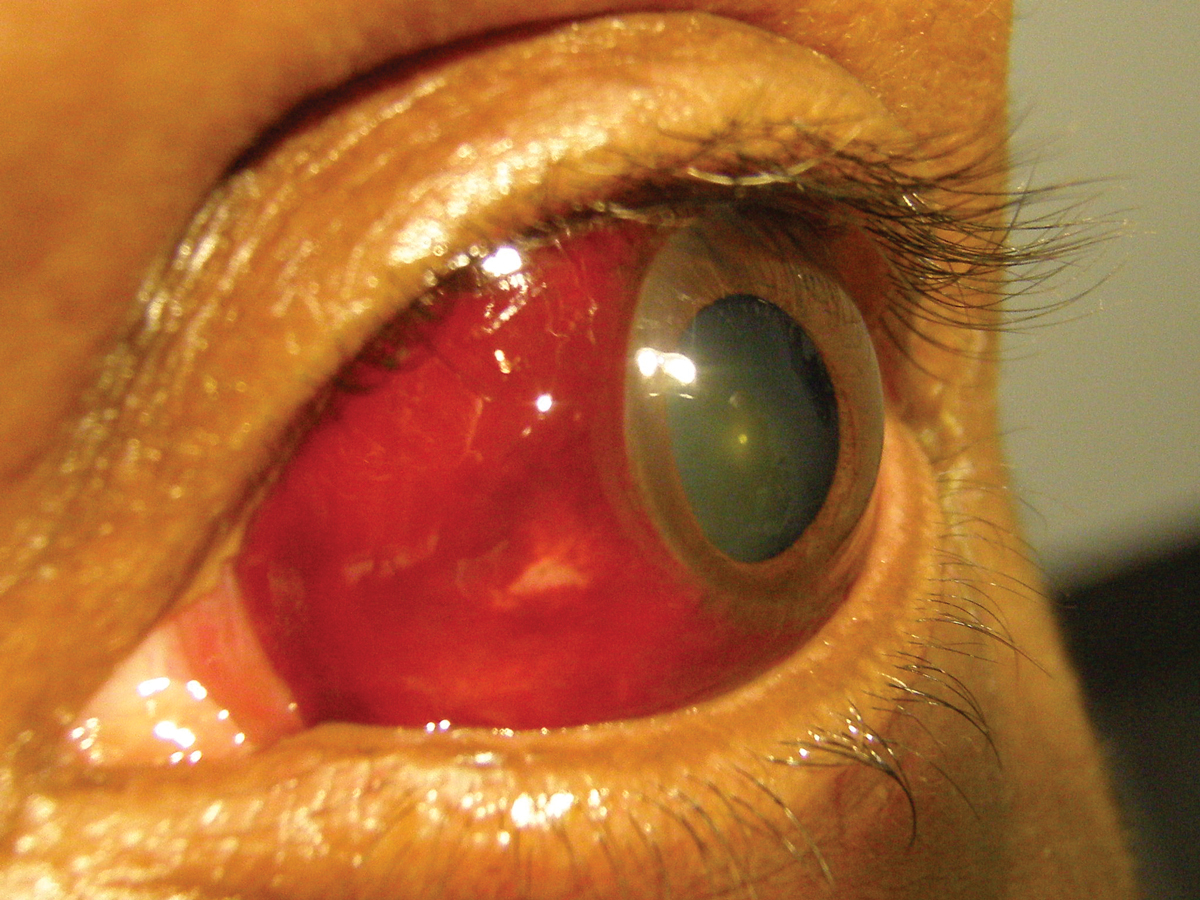
His best-corrected acuities were 20/20 OD and OS. The pertinent component of his external examination is demonstrated in the photo. There was no evidence of afferent pupillary defect. The remainder of his anterior, middle and posterior segment exam was normal. Goldmann tonometry measured 17mm Hg OU.
Additional Testing
Efforts to obtain more information included asking about possible recent trauma (e.g., sports, car accident), violent coughing, weight lifting, constipation and even any recent episodes of yelling.
In the absence of a mechanical vector elicited through discussion of history, lab work must be considered to rule out systemic diseases that could produce blood coagulopathy or hyperviscosity. Testing might include:
- complete blood count with platelets (CBC with differential and platelets)
- prothrombin time (PTT)
- activated partial thromboplastin time (APTT) and an international ratio (INR) to rule out over-anticogulation in patients taking blood thinners
- homocysteine level, particularly in young men
- assay for natural anticoagulants such as protein S and protein C
Repeat events, evidence of additional bruising observed elsewhere or multiple events in a child or young adult must be considered suspicious for a shaking injury.
 |
|
The patient was fairly unfazed by his appearance. Should the clinician be as well? Click image to enlarge. |
Diagnosis
The diagnosis is large subconjunctival hemorrhage (SCH). In this frequently seen ocular condition, patients typically present acutely, often with great anxiety regarding a “blood-red eye.” A history of antecedent trauma is often, but not always, present. Pain and/or photophobia are atypical unless the condition is associated with a blunt-force injury. Typically, visual acuity is not impacted unless there is an associated anterior or posterior segment injury form trauma.
The clinical presentation involves a well-defined, circumscribed area of visible, coalesced blood between the bulbar conjunctiva and episclera.1-5 The hemorrhage may be flat or elevated, depending upon the volume of blood involved. Biomicroscopic examination can reveal a widely variable clinical picture with mild cases demonstrating only a focal blotch to severe cases covering the full palpebral aperture completely obscuring the underlying sclera.1
SCH can be encountered at any age, in individuals of any race. There is no gender predilection. However, traumatic SCH tends to occur more commonly in younger men.4,5 In cases not associated with trauma, patients tend to be older, typically over 60 years of age.2,4 In such individuals, the systemic history often reveals associated risk factors such as hypertension, arteriolosclerosis, diabetes, hematologic disorders and medical therapy with anticoagulant or antiplatelet drugs.1,2,4-7
The patient may also confirm a recent Valsalva maneuver. By definition, this involves either an intentional or involuntary expiration of air against a closed glottis, causing rapidly and often severely increased pressure in both the chest and head.1,8,9 Episodes of violent sneezing, coughing, vomiting, forced defecation, weight lifting or other forms of straining (even yelling) can produce this phenomenon and a SCH.1,8,9 Hemorrhage may also occur in association with or subsequent to other primary ocular disorders with repeated injuries worthy of being considered as a manifestation of shaking.
Numerous studies have suggested that individuals with conjunctivochalasis are predisposed to more frequent and chronic instances of SCH, likely due to the frictional forces of a loosely adherent, redundant conjunctiva.10-12 Infectious conjunctivitides, most notably epidemic keratoconjunctivitis and acute hemorrhagic conjunctivitis, but also those caused by systemic infection such as typhoid, dengue fever and Ebola may induce SCH as well.13-17 Ophthalmic procedures can also produce SCH as an unintentional sequela, including foreign body removal, LASIK, cataract extraction and intravitreal injections.1,4,18 Injury associated with insertion or removal of contact lenses is another common source of SCH.3,4,19
Causes and Consequences
Histologically, SCH is defined as hemorrhage in the subconjunctival space, localized between the conjunctiva and Tenon’s capsule, superficial to the episclera and sclera.1,5 It is associated with rupture or leakage of subconjunctival capillaries. External mechanical forces are responsible for a good portion of cases seen clinically. In one study, nearly 78% of patients under the age of 40 had SCH associated with some type of traumatic event.4 Potential etiologies in this category include penetrating injury, iatrogenic trauma, blunt trauma, contact lens–related injury or vigorous eye rubbing.1,4,5 Rapid acceleration or deceleration (as might be encountered in a motor vehicle accident or amusement park ride) and barotrauma associated with scuba diving (i.e., “mask squeeze”) would also fall into this category.20
Internal mechanical forces, such as the aforementioned Valsalva maneuver, can also induce SCH.9 This pressure is also transmitted along the vasculature as well, primarily affecting the non-muscular walls of the venous system. The jugular, orbital and choroidal veins are subject to rapidly expansive forces; smaller tributaries of these blood vessels within the conjunctiva or retina may be susceptible to rupture.1,8,9,21 Conditions that further increase abdominal pressure, such as elevated body mass index or pregnancy, may enhance this susceptibility.22
Most cases of SCH in patients over 60 years of age are associated with small-vessel disease, primarily hypertension and diabetes.2,4 Hypertension causes increased pressure on arterial and venous walls making them more prone to structural damage, while diabetes induces macrovessel dilation and capillary dropout.23 A hallmark of these diseases is vascular leakage in end-organs, such as the eye.
Finally, hematologic abnormalities can produce the development of SCH. Anemias, blood dyscrasias and blood malignancies (e.g., polycythemia, hemochromatosis, hyperlipidemia, acute lymphoblastic leukemia, multiple myeloma) serve to increase blood volume and viscosity while diminishing blood flow. Bleeding disorders such as Von Willebrand’s disease, hemophilia and adverse effects of anticoagulation therapy (even the taking of aspirin) can diminish one’s ability to form clots and create an environment for the hemorrhagic condition associated with SCH.6,7,24-26
The etiologic factors underlying SCH may be reflected in its presentation, although individual variations certainly may occur. A prospective study of 151 consecutive patients with SCH reached the following conclusions: (1) Traumatic SCH had a smaller extent compared with SCH related to hypertension, diabetes, hyperlipidemia or those designated as idiopathic; (2) the extent of SCH was greater in older patients (>60 years), especially those who had previously undergone cataract surgery; and (3) overall, SCH was significantly more common in the inferior areas than the superior areas, but in patients with SCH secondary to trauma or diabetes, the temporal areas were affected more often than the nasal areas.3
For all patients, the management for SCH must begin with appropriate education and reassurance. In most instances—especially those involving spontaneous (i.e., non-traumatic) SCH—the chief concern is one of cosmesis. These events are typically self-limiting and resolve completely within a week or two in the vast majority of cases.27,28 As the hemorrhage dissipates, it is normal for the color of the blood to change, first from bright to dark red, then fading to yellow and ultimately clearing. To date, no specific therapy has been shown to expedite this process, although many practitioners continue to recommend artificial tears and warm or cool compresses in an effort to palliate the patient.
SCH associated with trauma warrants a comprehensive ophthalmic examination to identify any additional ocular compromise. Blunt injury carries a risk of concurrent uveitis, hyphema, angle recession, lens subluxation and retinal detachment. Lacerations and puncture wounds may penetrate deeper, whereby the SCH may conceal a possible perforation of the globe. Treatment of these comorbidities is case-specific and may necessitate referral for surgical consultation. SCH is not a contraindication itself to pupil dilation.
In those cases that are seemingly idiopathic—particularly recurrent or persistent cases—a systemic or shaking etiology should be considered and investigated. A comprehensive physical examination is prudent if the patient has not undergone one in the past year. Systemic hypertension, diabetes and hyperlipidemia are the most common associations.1 Additional laboratory testing may be directed toward identifying blood dyscrasias, clotting disorders and other hematological abnormalities. Suggested testing may include (but is not limited to) the following: a complete blood count (CBC) with differential and platelets, prothrombin time (PT), activated partial thromboplastin time (PTT), homocysteine levels, antiphospholipid antibodies, protein S, protein C, antithrombin III, factor V Leiden, beta-glycoprotein, sickle cell preparation and human immunodeficiency virus titers.
Patients taking warfarin (Coumadin, Bristol-Myers Squibb) or dabigatran (Pradaxa, Boehringer Ingelheim Pharmaceuticals) or aspirin should obtain international normalized ratio (INR) values to determine if therapy needs to be adjusted.29-31 However, before any systemic medications are discontinued or modified, communication and discussion with the internist is essential.
Patient Management
In discussing SCH with patients, we sometimes like to use the “ketchup under plastic wrap” or “blood blister” analogy. Even a small drop of ketchup (blood) can look menacing if it’s compressed over a large area by a tight layer of plastic wrap (conjunctiva). This description can help to make the patient less anxious about the presentation.
The word hyposphagma is sometimes used synonymously with subconjunctival hemorrhage. This term is considered obsolete by most and is rarely taught in today’s educational institutions; still, some authors persist in using this term (particularly in publications originating outside the United States).32
Additional though rarely encountered causes of SCH include strangulation injuries and conjunctival tumors. Patients who have survived asphyxiation may present with a triad of SCH, eyelid ecchymosis and petechial hemorrhages of the face.33 Vascular or malignant tumors of the conjunctiva may also be the source of SCH, although these diagnoses are typically evident well before the patient presents with hemorrhage.34,35 Patients with traumatic SCH may report discomfort if the SCH is associated with iritis. When considering an analgesic medication, those with anti-platelet activity such as aspirin and ibuprofen should be avoided. Acetaminophen, with or without narcotic or non-narcotic agents (e.g., codeine, hydrocodone, tramadol) is a better choice for these individuals.
Seemingly idiopathic cases that are recurrent or persistent in nature certainly warrant further investigation. One simple systemic investigation that can be readily performed on virtually all patients with SCH is blood pressure measurement. This is of particular value in patients over the age of 60, where systemic conditions are usually the causative etiology.
Finally, although it may be tempting to employ surgical evacuation for a large or extensive SCH, this technique is rarely used. It should only be considered in cases that present with severe pain and significant ophthalmic morbidity to adjacent structures, such as might be seen in a subconjunctival hematoma.36 Any 360-degree subconjunctival hemorrhage following trauma should invoke suspicion and prompt an investigation to rule out a ruptured globe.
Despite the “ugly” appearance of the patient presented here, this injury self-resolved over the course of six weeks with only cold compresses and lubrication for artificial tear drops and ointments. A traumatic vector was found (jujitsu competition) and the remainder of the ocular exam was normal. The patient was properly educated and informed with that sort of activity the injury could happen again.
Dr. Gurwood is a professor of clinical sciences at The Eye Institute of the Pennsylvania College of Optometry at Salus University. He is a co-chief of Primary Care Suite 3. He is attending medical staff in the department of ophthalmology at Albert Einstein Medical Center, Philadelphia. He has no financial interests to disclose.
1. Tarlan B, Kiratli H. Subconjunctival hemorrhage: risk factors and potential indicators. Clin Ophthalmol. 2013;7:1163-70. 2. Hu DN, Mou CH, Chao SC, et al. Incidence of non-traumatic subconjunctival hemorrhage in a nationwide study in Taiwan from 2000 to 2011. PLoS One. 2015;10(7):e0132762. 3. Mimura T, Yamagami S, Usui T, et al. Location and extent of subconjunctival hemorrhage. Ophthalmologica. 2010; 224(2):90-5. 4. Mimura T, Usui T, Yamagami S, et al. Recent causes of subconjunctival hemorrhage. Ophthalmologica. 2010;224(3):133-7. 5. Sahinoglu-Keskek N, Cevher S, Ergin A. Analysis of subconjunctival hemorrhage. Pak J Med Sci. 2013;29(1):132-4. 6. Taamallah-Malek I, Chebbi A, Bouladi M, et al. Massive bilateral subconjunctival hemorrhage revealing acute lymphoblastic leukemia. J Fr Ophtalmol. 2013;36(3):e45-8. 7. Felipe AF, Nottage JM, Rapuano CJ. Recurrent bilateral subconjunctival hemorrhage as an initial presentation of multiple myeloma. Oman J Ophthalmol. 2012;5(2):133-4. 8. Ricci S, Moro L, Minotti GC, et al. Valsalva maneuver in phlebologic practice. Phlebology. 2017 Jan 1:268355516678513. [Epub ahead of print]. 9. Kurultay-Ersan I, Emre S. Impact of Valsalva maneuver on central choroid, central macula, and disk fiber layer thickness among high myopic and hyperopic patients. Eur J Ophthalmol. 2016 Jul 18:0. [Epub ahead of print] 10. Di Pascuale MA, Espana EM, Kawakita T, Tseng SC. Clinical characteristics of conjunctivochalasis with or without aqueous tear deficiency. Br J Ophthalmol. 2004;88(3):388-92. 11. Mimura T, Usui T, Yamagami S, et al. Subconjunctival hemorrhage and conjunctivochalasis. Ophthalmology. 2009;116(10):1880-6. 12. Yamamoto Y, Yokoi N, Ogata M, et al. Correlation between recurrent subconjunctival hemorrhages and conjunctivochalasis by clinical profile and successful surgical outcome. Eye Contact Lens. 2015;41(6):367-72. 13. Harada K, Fujimoto T, Asato Y, Uchio E. Virological and epidemiological analysis of coxsackievirus A24 variant epidemic of acute hemorrhagic conjunctivitis in Okinawa, Japan, in 2011. Clin Ophthalmol. 2015;9:1085-92. 14. Jhanji V, Chan TC, Li EY, et al. Adenoviral keratoconjunctivitis. Surv Ophthalmol. 2015;60(5):435-43. 15. Thapa R, Banerjee P, Jain TS. Bilateral subconjunctival haemorrhage in childhood enteric fever. Singapore Med J. 2009;50(10):1038-9. 16. Jain S, Goswami A, Singh N, Kaur S. Bilateral eyelid ecchymosis and subconjunctival haemorrhage manifesting as presenting feature in a case of dengue haemorrhagic fever. Trop Doct. 2015;45(4):236-8. 17. Moshirfar M, Fenzl CR, Li Z. What we know about ocular manifestations of Ebola. Clin Ophthalmol. 2014;8:2355-7. 18. Fasih U, Shaikh N, Rahman A, et al. A one-year follow-up study of ocular and systemic complications of intravitreal injection of bevacizumab (Avastin). J Pak Med Assoc. 2013;63(6):707-10. 19. Mimura T, Yamagami S, Mori M, et al. Contact lens-induced subconjunctival hemorrhage. Am J Ophthalmol. 2010;150(5):656-665.e1 20. Yeoh R, Yeoh R, Singh M. Barotraumatic ocular haemorrhage sustained while scuba diving. Clin Exp Ophthalmol. 2008;36(6):581-2. 21. Goldman DR, Baumal CR. Natural history of valsalva retinopathy in an adolescent. J Pediatr Ophthalmol Strabismus. 2014;51(2):128. 22. Tara F, Sharifi M, Hoseini E. Valsalva retinopathy in pregnancy: a case report. BMC Res Notes. 2015;8:67. 1. |

