Because the retina is often considered an extension of the brain—sharing common features such as anatomical structure, vascular supply and the blood/tissue barrier—retinal imaging offers a unique opportunity to detect and monitor progression of certain neurodegenerative diseases in vivo.1-8 For example, frequency-doubling perimetry has been shown to detect early changes in ganglion cells and higher visual cortical compromise in neurodegenerative diseases with high sensitivity and specificity.5,9 Whether the retinal findings in neurodegenerative diseases reflect a local pathogenesis of the disease process or a secondary result of retrograde loss from cortical neurons remains unknown.10 This article discusses the early signs of neurodegenerative disease in the eye to aid in early diagnosis and prompt management.
| Disease Impact Neurodegenerative disorders are a significant socioeconomic burden and a major health care concern.3 They are typically seen in adulthood, tend to be progressive and share similar mechanisms of pathogenesis.3,51 Due to the impact of neurodegenerative diseases on society, researchers are evaluating methods to identify high-risk individuals by screening for biomarkers.52 To be effective in a primary care setting, biomarker testing must be inexpensive, readily available, rapid, reproducible and well tolerated by the patient.1 The screening test must have a high sensitivity and specificity for the disease in question, particularly at its earliest stages. It must also have the ability to identify progression prior to significant cognitive loss.1,11,13,28 |
Alzheimer’s Disease
Dementia, a term for conditions representing a change in mental state due to brain disease or injury, had a global prevalence of 35.6 million in 2010 and is expected to double every 20 years.11 Damage to nerve cells can affect memory, behavior and the ability to perform routine tasks.12
The most common subtype of dementia is Alzheimer’s disease (AD), representing 60% to 80% of cases.12,13 Approximately 50% of patients diagnosed with presumed AD have solely AD, and 50% have mixed types of dementia.12 Examples of other forms include vascular dementia, frontotemporal lobar degeneration, Parkinson’s disease and dementia with Lewy bodies.12 Neurodegenerative diseases such as AD are exceedingly difficult to study because a definitive diagnosis cannot be made until postmortem histopathological evaluation. Therefore, clinical studies are difficult and often suffer from an increased rate of error.14
Alzheimer’s disease has a prevalence of 6.4% for North Americans age 60 or older.1 The prevalence increases with age, so that by age 85, 32% are afflicted with AD.15 It is incurable and leads to progressive neuronal cell death in the brain due to amyloid protein plaques and neurofibrillary tangles that abnormally accumulate in the central nervous system (CNS) and interfere with communication between neurons, resulting in cerebral and hippocampal atrophy.10,14 The disease progression is typically insidious, with estimates that neuronal damage may be present for up to 20 years prior to cognitive decline.8,10
The most common symptom in early disease is short-term memory loss. Symptoms can be grouped into cognitive dysfunction, including, but not limited to, memory loss and language deficits. The two remaining elements include non-cognitive and behavioral symptoms, which can include depression, hallucinations or delusions, among others. Vision and ocular abnormalities are also prevalent in AD (Table 1).
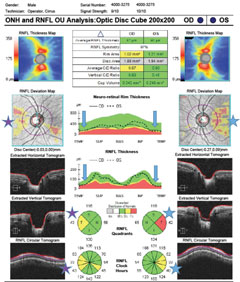 |
| Fig. 1. OCT peripapillary RNFL scan depicting temporal quadrant thinning OU compared with normative data, notated by the purple stars OD and blue stars OS on the deviation maps, quadrant thickness graphs and the clock hour thickness graphs of each eye. The blue arrows mark areas of thinning compared with normative data on the TSNIT graphs in the center of the OCT printout. |
A probable diagnosis of AD is made based on a thorough medical history, imaging tests and laboratory workup to rule out other neurodegenerative etiologies. Unfortunately, the diagnosis is often made after the disease has reached an advanced state and significant neuronal damage has occurred.16 Longitudinal studies postulate that, in early AD, cognitive impairment remains relatively stable.17 Therefore, early detection of probable AD provides the greatest opportunity for therapeutic intervention and management.
Testing for AD includes magnetic resonance imaging (MRI) of the head and a cerebrospinal fluid (CSF) evaluation; the latter tends to be accurate in identifying specific biomarkers for AD.18 However, CSF testing can be painful and is invasive, which decreases its usefulness as a screening method. Functional MRI has been employed in testing for AD, but variable presentations of the normal aging brain anatomy presents challenges.19,20 Position emission tomography (PET) scans are currently being evaluated for the detection of AD using compounds that bind amyloid beta peptides known to accumulate in plaques.21 In combination with genetic testing, PET scans can be highly predictive for the development of AD.21
Currently, the only therapies available for AD are palliative. Standard treatment typically consists of neurotransmitter modulators for acetylcholine or glutamate.22 Other medicinal therapies target behavioral symptoms and may include antidepressants, anxiolytics, anti-Parkinson’s medications, beta-blockers, anti-epileptics and neuroleptics.22,23
The Eye in Alzheimer’s Disease
Alzheimer’s disease can affect not only the brain but also the eye. Research on humans and mice indicate that AD pathology occurs in the retina as well as the CNS.10 Some findings include:
• Retinal nerve fiber layer (RNFL). Investigators have noted a significant reduction in RNFL thickness in individuals with AD, as well as those with earlier, prodromal stages of AD with mild cognitive impairment (MCI), compared with normals.13 Decreased RNFL thickness quantified by optical coherence tomography (OCT) in patients with MCI and AD correlates to loss of retinal neurons and axons.13 This suggests that the retina may be an early site of damage prior to significant cognitive decline.13 Investigators noted thinning of the RNFL in all quadrants of AD patients using OCT, which was confirmed with abnormal retinal function testing using pattern electroretinograms.24 Other researchers evaluated MCI and early AD patients compared with moderate and late-stage AD patients and found RNFL thinning in all quadrants with each group.10,25,26 Although RNFL thinning occurs in AD, it is non-specific for a particular disease process, and whether it can be used to predict those at high risk for developing AD remains to be seen.27 Recent studies have focused on longitudinal RNFL thinning in combination with memory measurements to help further define and predict future progression.28
| Table 1. Common Ocular Symptoms/Findings of Alzheimer’s Disease1,2,59 | ||
Eye Function | Symptoms | Testing |
| Visual acuity (VA) | • Blurring | • Snellen VA chart |
Color vision | • Abnormal appearance of colors | • Color vision plates |
Contrast sensitivity | • Unable to distinguish objects based on changes in light | • Pelli Robson contrast sensitivity chart |
Dark adaptation | • Increased time needed to adapt to changes in light level | • Macular photostress test |
Visual perceptual abnormality | • Illusions | • Visual perceptual workup |
| Pupils | • Increased pupil response to cholinergic drops | • Pharmacological evaluation |
| Binocularity | • Double vision | • Extraocular motility testing |
Visual field defects | • No symptoms | • Confrontational fields |
• Macula. Researchers have found varying results in macular thickness and volume measurements with MCI and AD patients and hypothesize the differences are related to the stage of the disease.13 Studies report increased macular thickness and volume in some MCI patients, which may be related to the early development of AD.13,29 In contrast, other researchers found reduced macular thickness and volume measurements in AD patients, with the severity of macular findings related to the degree of cognitive impairment.4,10,30 Yet others obtained similar results but with no correlation between findings and dementia severity.4
Looking more closely at cellular changes in individual retinal layers, research shows significant retinal ganglion cell (RGC) degeneration in AD patients.10,31 No plaques or tangles were found in the retinal or optic nerve tissue. This supports known evidence that plaques and tangles primarily occur in the hippocampus and limbic areas of the brain in AD, sparing the visual and motor regions of the CNS.31Therefore, the researchers hypothesized that RGC loss was most likely related to neurodegenerative disease and not the result of a secondary retrograde process from the CNS.10,31 The findings showed a 25% reduction in ganglion cell density in the fovea and parafoveal regions, with greatest reduction in the temporal sector of patients with AD.10,31 Anatomically, these follow the distribution of ganglion cell fibers in the optic nerve forming the papillomacular bundle.
Current research is inconclusive regarding the extent to which changes in macular anatomy and the use of macular imaging can clearly define neurodegenerative pathology in the CNS. Due to the concentration of RGCs in the macula, researchers believe this may represent an area to evaluate both neuronal and axonal degeneration by assessing ganglion cell loss and RNFL thinning, respectively.10
• Optic nerve cupping. Research suggests retinal ganglion cell loss in AD mimics loss seen in glaucoma at a biochemical level due to neurotoxicity from amyloid deposition.10 Investigators demonstrated a fivefold greater risk of visual field defects and optic disc cupping in patients with AD, as well as a higher prevalence of glaucoma in this population.32 Researchers also found accelerated visual field loss in glaucoma patients with AD.10 Differentiating optic nerve pathologies with respect to neurodegenerative diseases continues to present a challenge to clinicians.
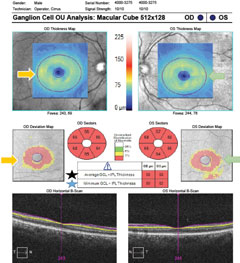 |
| Fig. 2. OCT ganglion cell analysis demonstrating overall ganglion cell/inner plexiform layer thinning compared with normative data. The orange arrows OD and green arrows OS indicate thinning in the thickness and deviation maps for each eye. The black and blue stars notate the data box in the center of the printout showing significant reduction in average and minimum ganglion cell/inner plexiform layer thickness OU. |
Figures 1 and 2 depict a presentation not typical of a glaucomatous process, which mainly affects the inferior and superior neuroretinal rim first, but is more likely associated with a non-glaucomatous neuropathy. However, the exact etiology of the temporal thinning cannot be determined by OCT imaging alone. Visual field testing, serial nerve photography, a thorough history and clinical examination can be adjunctive, but not exclusive, in diagnosing ocular or CNS disease processes. Advancements in disease-specific OCT software will likely improve our ability to identify and differentiate ocular pathologies and correlate findings with neurodegenerative disease.
• Microvascular abnormalities. Researchers have evaluated retinal photos of patients with probable AD and found retinal blood vessel alterations associated with plaque deposits in the brain.3 These variations consist of venous branching pattern asymmetry, as well as increased arteriolar length to diameter ratio values in AD. Researchers evaluated other measurements such as vascular attenuation, complexity of branching pattern and vessel tortuosity and hypothesized that retinal vasculature morphological changes consist of amyloid deposition extending from the CNS to the retina, resulting in vessel wall destruction.3 Therefore, detailed funduscopic evaluation and serial retinal photography may present a screening mechanism for earlier AD detection.3 Using a mouse model, investigators demonstrated microvascular impairment for AD due to amyloid accumulation in small vessels leading to tortuosity and reduction in vessel caliber.33
• Pupillary abnormalities. Currently, investigators are evaluating pupillary abnormalities as potential markers for AD. One particular area of interest is hypersensitivity to pupillary dilation with cholinergic antagonists and agonist medications, such as diluted tropicamide and pilocarpine, respectively.10 Other research is looking at pupil flash response, which evaluates the pupil’s reaction to light of varying intensities and different durations, which is found to be compromised in AD patients.10
Parkinson’s Disease
A neurodegenerative disorder affecting the basal ganglia of the brain, Parkinson’s disease (PD) describes a process that leads to dopamine-producing cell loss and abnormal deposition of protein inside nerve cells, termed Lewy bodies.8 Other regions of the brain affected by PD include the hypothalamus and the nuclei of the thalamus, as well as the cerebral cortex, amygdala and the hippocampus in advanced disease.34 A key hormone for signal transmission in the CNS, dopamine’s depletion with loss of associated neurons leads to impairment of cognitive, motor and sensory function.6,35
| Testing Limitations Because neurodegenerative disease typically occurs in a diffuse fashion throughout the CNS, the retina—and retinal ganglion cells in particular—only represents a small subset of the CNS and may not fully reflect the extent of neurological damage.53 Other challenges include normal changes in the retina and optic nerve anatomy with age; namely, thinning of RNFL and loss of ganglion cells.53 Studies have shown the normal rate of RNFL thinning is approximately 0.2μm per year, and yet the magnitude of RNFL decline with age may be higher in eyes with larger baseline RNFL thickness.54,55 Furthermore, there is a distinct variability in structural characteristics, which will continue to be an obstacle with current OCT software and normative databases.56 Not only is there anatomical variability between normal eyes of different individuals, but the normal findings between the right and left eyes of each patient may differ as well. We must define normal physiological variance between eyes to better define pathological asymmetry. Understanding the variance of normal OCT findings is helpful, considering researchers found that foveal thinning was more pronounced in the eye opposite the side of the body most affected by tremor in PD subjects as well as patients with essential tremor.42 The organic etiology for asymmetrical laterality in neurodegenerative disease manifestations in some presentations is still poorly understood and further investigation is warranted. Technological advances in OCT software for cell distinction and disease specificity will determine the role OCT plays in neurological disease diagnosis and monitoring in the future.42 Evaluating the correlation of RNFL thinning and RGC loss in association with MRI of the brain will help determine the exact association of retinal findings in correlation with CNS manifestations in neurodegenerative disease.11 Further investigation into the combination of RNFL parameters, macular thickness and macular volume findings may give the highest sensitivity and specificity as a potential biomarker for neurodegenerative states. |
This disorder affects more than one million people in the United States, with a peak incidence in the fifth and sixth decade and a prevalence of approximately 1% in the elderly.36 It is the second most common neurodegenerative disease in the western world; however, its incidence is estimated to be only 1/10 that of AD.12,37 Symptoms of the disease include, but are not limited to, bradykinesia (slow movement), muscle rigidity on movement, resting tremor, posterior instability and altered gait.7 Additional motor symptoms can include apathy, anxiety, depression, fatigue, memory disturbance, sensory impairment, sleep disorders and autonomic disturbance.7 PD can have a number of visual symptoms and ocular signs (Table 2).
The diagnosis of PD is typically based on motor function symptoms; however, non-motor deficiencies—such as constipation, sleep disturbances, bladder problems and depression—are common in the disease process as well.35,38 Early diagnosis and prompt therapeutic intervention can increase the likelihood progression of the disease can be slowed.34 As in AD, no specific test exists to diagnose PD. Typically, a diagnosis is made based on clinical history, symptoms and a neurological examination. Imaging tests such as MRI and PET scans are ordered along with laboratory testing to rule out neurological conditions such as essential tremor, dementia with Lewy bodies, chorea, embolic stroke and others, as differentials.38,39
| Table 2. Common Ocular Symptoms/Findings of Parkinson’s Disease60,61 | ||
| Eye Function | Symptoms | Testing |
| Visual acuity | • Reduced VA
| • Snellen VA |
| Dry eye | • Reduced tear break-up time | Dry eye workup: |
| Binocularity/motility | • Convergence insufficiency | Extraocular motility testing: • Vergance testing • Saccades • Pursuits • Fixation testing • Optokinetic nystagmus testing |
| Visual perceptual abnormality | • Visual hallucinations | • History |
| Eyelid | • Blepharospasm
| • Upgaze test to assess fatigability and hypometric movement |
| Contrast sensitivity | • Abnormal sensitivity with intermediate to high frequency | • Pelli Robson contrast sensitivity chart |
| Color vision | • Visual blur with colors | • Color vision plates |
| Visual field | • Field defects may be increased in glaucoma patients | • Confrontational fields |
Although the disease is considered incurable, certain medications may help with symptoms and aid in quality of life. Because PD targets dopamine-producing cells, drug therapy is aimed at replacing the neurotransmitter dopamine, since dopamine itself cannot directly penetrate the blood/brain barrier. A common medication prescribed in PD is levodopa, which is able to cross the blood/brain barrier where it is converted to dopamine. Often, levodopa is combined with carbidopa to prevent premature conversion to dopamine, thereby reducing side effects of the drug. In 2015, a carbidopa-levodopa infusion called Duopa (AbbVie) was approved by the FDA. Duopa is administered continuously by a pump directly to the small intestine, which keeps levels constant in the blood stream.39 Other classes of drugs either mimic dopamine or help prevent the breakdown of dopamine, thereby prolonging the effect.40,41
In advanced disease, surgical implantation of a deep brain stimulation device that sends small electric shocks to areas of the brain such as the thalamus and globus pallidus has proven effective in reducing symptoms of tremor, rigidity and stiffness.38,41 This procedure blocks electrical impulses to targeted CNS regions, but does not damage brain tissue.38
The Eye in Parkinson’s Disease
Researchers have demonstrated that dopamine, which is commonly found in the retina of normal subjects, is decreased in PD patients.7 Dopamine deficiency specifically can affect many cells of the retina, including horizontal, amacrine, bipolar and ganglion.7 These cells may provide a biomarker to detect early disease onset as well as follow its progression.
• Retinal nerve fiber layer. Researchers first described peripapillary RNFL thinning in PD patients using OCT in 2004.37,42,43 More recent research has demonstrated variable results showing thinning in the RNFL of PD patients with predominant loss in the temporal peripapillary retina in some studies, while other research is inconclusive.28 One study found that both RNFL thinning and RGC loss did occur in a cohort of PD patients with correlation to functional reductions in visual acuity, contrast sensitivity, visual fields, color vision and electrodiagnostic tests. They concluded, however, that macular measurements of the RGC may be more reliable than RNFL thinning to define retinal structural alterations in PD.44 With no consensus in repeatable longitudinal study findings, RNFL changes associated with PD cannot currently be applied to a screening process.
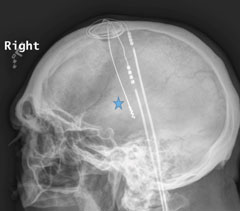 |
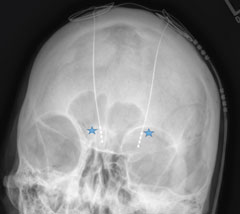 |
| Figs. 3 and 4. Radiographs of the skull with two deep brain stimulators within the cranium notated adjacent to the blue stars. |
• Macula. A recent study demonstrated a reduction in central macular thickness and macular volume as well as thinner inner retinal layers in PD patients correlating with reduced motor score testing.28 Others found a negative correlation between central macular thickness (CMT) and motor scores. These results suggest that depleted dopaminergic cells are not able to communicate with the cone receptors within the fovea, resulting in macular thinning.28,45 Other investigators found retinal thinning at the edges of the foveal pit in a study cohort of PD patients. They hypothesize this reconstruction of the parafoveal zone may be unique in PD patients and may be related to oxidative stress that occurs to retinal neurons during the disease process.42,46,47
Advanced OCT software has depicted morphological changes in the individual retinal layers of PD patients. The maximal resolution of 3µm to 5µm with SD-OCT limits its ability to define specific cell types.42 Several studies have found retinal thinning in the nasal and inferior paramacular regions related to PD.7 These findings are supported by autopsy findings of protein aggregates typical of PD in the inner retinal layers of subjects.8 Specific evaluation of the ganglion cell, inner nuclear, inner plexiform and outer nuclear layers has also demonstrated thinning related to loss of dopaminergic cells in PD subjects.8
Increased volume of the outer plexiform layer has been reported in PD patients using OCT, supported by histopathological evidence of increased autoimmune reactivity to proteins within this layer.8 Future studies may focus on the parafoveal inner retinal layers, specifically the inner plexiform layer that houses the dopaminergic amacrine cells and innerconnections.8,36 Specific OCT algorithms for PD are not commercially available, but literature suggests that macula scans may be more accurate than RNFL for defining neurodegenerative loss in PD.42
If an accurate ocular biomarker can be established for routine exams, it could aid in possible neuroprotective strategies, disease targeting, anti-inflammatory, immunosuppressive or antioxidative therapies to slow the progression of the neurodegenerative processes.14 OCT and retinal photography may provide a fast, inexpensive, noninvasive and reproducible means of detecting neurological disease in high-risk patients in the primary optometric setting.28 Further longitudinal studies will be needed to determine if retinal findings represent a primary retinal degeneration as a part of the neurodegenerative CNS disease process or retrograde manifestation from the CNS.37
Drs. Mazzarella and Cole are staff optometrists in the Salisbury VA Health Care System, Salisbury, NC.
|
1. Ikram MK, Cheung CY, Wong TY, Chen CP. Retinal pathology as biomarker for cognitive impairment and Alzheimer’s disease. Journal of Neurology, Neurosurgery, and Psychiatry. 2012;83(9):917-22. 2. Mancil GL. Visual disorders associated with Alzheimer’s disease and optometric management. J Am Optom Ass. 1994;65(1):27-31. 3. Frost S, Kanagasingam Y, Sohrabi H, et al. Retinal vascular biomarkers for early detection and monitoring of Alzheimer’s disease. Translational Psychiatry. 2013;3:e233. 4. Gao L, Liu Y, Li X, et al. Abnormal retinal nerve fiber layer thickness and macula lutea in patients with mild cognitive impairment and Alzheimer’s disease. Archives of Gerontology and Geriatrics. 2015;60(1):162-7. 5. Valenti DA. Alzheimer’s disease: screening biomarkers using frequency doubling technology visual field. ISRN Neurology. 2013;2013:989583. 6. Roth NM, Saidha S, Zimmermann H, et al. Photoreceptor layer thinning in idiopathic Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society. 2014;29(9):1163-70. 7. Mailankody P, Battu R, Khanna A, et al. Optical coherence tomography as a tool to evaluate retinal changes in Parkinson’s disease. Parkinsonism & Related Disorders. 2015;21(10):1164-9. 8. Chorostecki J, Seraji-Bozorgzad N, Shah A, et al. Characterization of retinal architecture in Parkinson’s disease. Journal of the Neurological Sciences. 2015;355(1-2):44-8. 9. Thomas D, Thomas R, Muliyil JP, George R. Role of frequency doubling perimetry in detecting neuro-ophthalmic visual field defects. Am J Ophthalmol. 2001;131(6):734-41. 10. Jones-Odeh E, Hammond CJ. How strong is the relationship between glaucoma, the retinal nerve fibre layer, and neurodegenerative diseases such as Alzheimer’s disease and multiple sclerosis? Eye (London). 2015;29(10):1270-84. 11. Larrosa JM, Garcia-Martin E, Bambo MP, et al. Potential new diagnostic tool for Alzheimer’s disease using a linear discriminant function for Fourier domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2014;55(5):3043-51. 12. Alzheimer’s Association. 2014 Alzheimer’s Disease Facts and Figures. Alzheimer’s & Dementia. 2014;10(2). www.alz.org/downloads/Facts_Figures_2014.pdf. 13. Ascaso FJ, Cruz N, Modrego PJ, et al. Retinal alterations in mild cognitive impairment and Alzheimer’s disease: an optical coherence tomography study. Journal of Neurology. 2014;261(8):1522-30. 14. Sivak JM. The aging eye: common degenerative mechanisms between the Alzheimer’s brain and retinal disease. Invest Ophthalmol Vis Sci. 2013;54(1):871-80. 15. Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology. 2013;80(19):1778-83. 16. National Institute of Aging. 2014-2015 Alzheimers Disease Progress Report: Advancing Research Toward a Cure. 2015 December. 17. Lazarczyk MJ, Hof PR, Bouras C, Giannakopoulos P. Preclinical Alzheimer disease: identification of cases at risk among cognitively intact older individuals. BMC medicine. 2012;10:127. 18. Adamczuk K, Schaeverbeke J, Vanderstichele HM, et al. Diagnostic value of cerebrospinal fluid Abeta ratios in preclinical Alzheimer’s disease. Alzheimer’s Research & Therapy. 2015;7(1):75. 19. Zhan Y, Chen K, Wu X, et al. Identification of conversion from normal elderly cognition to Alzheimer’s disease using multimodal support vector machine. Journal of Alzheimer’s disease. 2015;47(4):1057-67. 20. Ferreira LK, Busatto GF. Resting-state functional connectivity in normal brain aging. Neuroscience and Biobehavioral Reviews. 2013;37(3):384-400. 21. Dukart J, Sambataro F, Bertolino A. Accurate prediction of conversion to Alzheimer’s disease using imaging, genetic, and neuropsychological biomarkers. Journal of Alzheimer’s disease. 2016;49(4): 1143-1159. [Epub ahead of print]. 22. Winslow BT, Onysko MK, Stob CM, Hazlewood KA. Treatment of Alzheimer disease. American Family Physician. 2011;83(12):1403-12. 23. Massoud F, Leger GC. Pharmacological treatment of Alzheimer disease. Canadian Journal of Psychiatry. 2011;56(10):579-88. 24. Parisi V. Correlation between morphological and functional retinal impairment in patients affected by ocular hypertension, glaucoma, demyelinating optic neuritis and Alzheimer’s disease. Seminars in Ophthalmology. 2003;18(2):50-7. 25. Paquet C, Boissonnot M, Roger F, et al. Abnormal retinal thickness in patients with mild cognitive impairment and Alzheimer’s disease. Neuroscience Letters. 2007;420(2):97-9. 26. Kesler A, Vakhapova V, Korczyn AD, et al. Retinal thickness in patients with mild cognitive impairment and Alzheimer’s disease. Clin Neurol Neurosurg. 2011;113(7):523-6. 27. Shi Z, Wu Y, Wang M, et al. Greater attenuation of retinal nerve fiber layer thickness in Alzheimer’s disease patients. Journal of Alzheimer’s Disease. 2014;40(2):277-83. 28. Shi Z, Zhu Y, Wang M, et al. The utilization of retinal nerve fiber layer thickness to predict cognitive deterioration. Journal of Alzheimer’s disease. 2015;49:2:399-405. 29. Reichenbach A, Wurm A, Pannicke T, et al. Muller cells as players in retinal degeneration and edema. Graefe’s Arch Clin Exp Ophthalmol. 2007;245(5):627-36. 30. Iseri PK, Altinas O, Tokay T, Yuksel N. Relationship between cognitive impairment and retinal morphological and visual functional abnormalities in Alzheimer disease. Journal of Neuro-Ophthalmology. 2006;26(1):18-24. 31. Blanks JC, Hinton DR, Sadun AA, Miller CA. Retinal ganglion cell degeneration in Alzheimer’s disease. Brain Research. 1989;501(2):364-72. 32. Bayer AU, Ferrari F. Severe progression of glaucomatous optic neuropathy in patients with Alzheimer’s disease. Eye (London). 2002;16(2):209-12. 33. Dorr A, Sahota B, Chinta LV, et al. Amyloid-beta-dependent compromise of microvascular structure and function in a model of Alzheimer’s disease. Brain: a Journal of Neurology. 2012;135(Pt 10):3039-50. 34. Jimenez B, Ascaso FJ, Cristobal JA, Lopez del Val J. Development of a prediction formula of Parkinson disease severity by optical coherence tomography. Movement Disorders. 2014;29(1):68-74. 35. Bayhan HA, Aslan Bayhan S, Tanik N, Gurdal C. The association of spectral-domain optical coherence tomography determined ganglion cell complex parameters and disease severity in Parkinson’s disease. Current Eye Research. 2014;39(11):1117-22. 36. Adam CR, Shrier E, Ding Y, Glazman S, Bodis-Wollner I. Correlation of inner retinal thickness evaluated by spectral-domain optical coherence tomography and contrast sensitivity in Parkinson disease. Journal of Neuro-Ophthalmology. 2013;33(2):137-42. 37. Stemplewitz B, Keseru M, Bittersohl D, et al. Scanning laser polarimetry and spectral domain optical coherence tomography for the detection of retinal changes in Parkinson’s disease. Acta Ophthalmologica. 2015. [Epub ahead of print]. 38. National Parkinson Foundation. Nonmotor Symptoms. Accessed December 18, 2015. www.parkinson.org/Understanding-Parkinsons/Non-Motor-Symptoms. 39. Mayo Clinic. Diseases and Conditions: Parkinson’s Disease. Accessed December 18, 2015. www.mayoclinic.org/diseases-conditions/parkinsons-disease/basics/symptoms/con-20028488. 40. Tarsly D. Pharmacologic treatment of Parkinson disease. UpToDate. 2015. www.uptodate.com/contents/pharmacologic-treatment-of-parkinson-disease. 41. Tarsly D. Nonpharmacologic management of Parkinson disease. UpToDate. www.uptodate.com/contents/nonpharmacologic-management-of-parkinson-disease. 42. Bodis-Wollner I, Miri S, Glazman S. Venturing into the no-man’s land of the retina in Parkinson’s disease. Movement Disorders. 2014;29(1):15-22. 43. Inzelberg R, Ramirez JA, Nisipeanu P, Ophir A. Retinal nerve fiber layer thinning in Parkinson disease. Vision Research. 2004;44(24):2793-7. 44. Kaur M, Saxena R, Singh D, et al. Correlation between structural and functional retinal changes in Parkinson disease. Journal of Neuro-Ophthalmology. 2015;35(3):254-8. 45. Altintas O, Iseri P, Ozkan B, Caglar Y. Correlation between retinal morphological and functional findings and clinical severity in Parkinson’s disease. Advances in Ophthalmology. 2008;116(2):137-46. 46. Spund B, Ding Y, Liu T, et al. Remodeling of the fovea in Parkinson disease. Journal of Neural Transmission. 2013;120(5):745-53. 47. Slotnick S, Ding Y, Glazman S, et al. A novel retinal biomarker for Parkinson’s disease: Quantifying the foveal pit with optical coherence tomography. Movement Disorders. 2015;30(12):1692-5. 48. Wills Eye Hospital. Test Procedures 2015. Accessed December 2, 2015. 49. Armstrong RA. Visual symptoms in Parkinson’s disease. Parkinson’s Disease. 2011;2011:908306. 50. Lenka A, Hegde S, Jhunjhunwala KR, Pal PK. Interactions of visual hallucinations, rapid eye movement sleep behavior disorder and cognitive impairment in Parkinson’s disease: A review. Parkinsonism & Related Disorders. 2015. [Epub ahead of print]. 51. Jentsch S, Schweitzer D, Schmidtke KU, et al. Retinal fluorescence lifetime imaging ophthalmoscopy measures depend on the severity of Alzheimer’s disease. Acta Ophthalmologica. 2015;93(4):e241-7. 52. National Cancer Institute. Biomarkers 2015. Accessed December 12, 2015. 53. Cheung CY, Ong YT, Hilal S, et al. Retinal ganglion cell analysis using high-definition optical coherence tomography in patients with mild cognitive impairment and Alzheimer’s disease. Journal of Alzheimer’s Disease. 2015;45(1):45-56. 54. Knight OJ, Girkin CA, Budenz DL, et al. Effect of race, age, and axial length on optic nerve head parameters and retinal nerve fiber layer thickness measured by Cirrus HD-OCT. Archives of Ophthalmology. 2012;130(3):312-8. 55. Leung CK, Yu M, Weinreb RN, et al. Retinal nerve fiber layer imaging with spectral-domain optical coherence tomography: patterns of retinal nerve fiber layer progression. Ophthalmology. 2012;119(9):1858-66. 56. Bussel, II, Wollstein G, Schuman JS. OCT for glaucoma diagnosis, screening and detection of glaucoma progression. Br J Ophthalmol. 2014;98 Suppl 2:ii15-9. |

