Dig into Dry EyeThis month, Review of Optometry brings you its 24th annual dry eye report. Inside the May issue, multiple ocular surface experts offer their guidance on understanding and managing dry eye in contact lens wearers, the effect of eyelid health and MGD on dry eye and how to detect and treat lacrimal gland abnormalities. Check out the other articles featured in this issue:
|
In 2017, the Tear Film and Ocular Surface Society released the findings of the Dry Eye Workshop II. The goal was to update the definition of dry eye established by the original Dry Eye Workshop in 2007 and re-evaluate the pathophysiology, diagnosis and management of dry eye spectrum conditions.1 The report focused on the multifactorial nature of dry eye and evaluated the complex interconnections between the tear film, lacrimal glands, meibomian glands, cornea, conjunctiva and eyelids and alterations of these structures that lead to homeostasis disruption and symptomatic disease.2
This article will focus on two of these connections—eyelid abnormalities and meibomian gland dysfunction (MGD)—and discuss how to recognize these conditions, along with the role they play in the development of dry eye.
Eyelid Malpositions
Entropion and ectropion refer to eyelid malpositions that commonly affect the population, particularly geriatric patients. Entropion is an inward rotation of the eyelid margin which often results in misdirection of lashes and with time, the eyelid margin can become abnormally keratinized. Ectropion is the contrasting disorder in which the lid margin rotates outward. These most often affect the bottom lids but can impact the upper eyelids as well. The general etiologies for eyelid malpositions are:
• involutional
• cicatricial (inflammation, infection or trauma)
• spastic (orbicularis spasm)
• neurogenic (CN 5 palsy)3
Regardless of the cause, poor lid closure and incomplete blinks result in exposure of the cornea, inadequate distribution of the tear film and chronic irritation of the ocular surface, which have significant effects on patients’ quality of life. The result can be a myriad of symptoms, including blurred vision, epiphora, pain and foreign body sensation. Dry eye was the most common reason that patients sought treatment for their lids.4,5
When evaluating the lids, additional techniques can help determine severity and causation of the malpositions. For ectropion, perform a snap back test by pulling downward on the lower lid and upward on the upper lid and evaluate the speed at which the lid snaps back into its normal position. Anything other than a brisk return to normal position is considered abnormal and severity can be assessed by the speed and whether lid blink is necessary to resume normal positioning. Presence and degree of eversion of the puncta away from the ocular surface also is indicative of severity.
For entropion, perform a digital eversion test by manually rotating the lid margin outward to its normal position. Involutional entropion can be manually everted but cicatricial cannot due to the scarring and shortening of lid tissues.4 Another common diagnostic test is the pinch test where the central eyelid is pulled away from the globe. This test is considered positive for horizontal laxity if the lid can be pulled more than 8mm away from the globe.6
Yang et al. demonstrated alterations to the meibomian glands from marginal entropion. Keratinization of the lid margin commonly occurs with entropion due to chronic mechanical irritation of the lid tissues with blink. This keratinization gradually dislocates the meibomian gland orifices. Chronic contact between the meibomian gland orifices and the ocular surface causes shedding of conjunctival cells which obstructs the ducts. Dammed meibomian secretions over time cause inflammatory cell infiltration, dilatation of the ducts and finally, atrophy.
Entropic lids were found to have higher meibomian gland loss and thinner lipid layer thickness (LLT) than normal lids.7 The more extensive the entropion, the greater was the degree of meibomian gland loss, but LLT did not decrease accordingly, possibly due to compensatory upregulation of production of lipids in the remaining glands. Patients were followed post entropion repair and were found to have an unchanged meibomian gland loss, but an increased LLT, supporting the notion that entropion is involved in the cause and/or worsening of MGD. Decreased LLT has been shown to closely correlate with signs and symptoms and ocular comfort improves when LLT is increased.8,9
While Yang et al. proposed that entropion causes or exacerbates MGD, Siah described the opposite scenario, in which entropion is a result of MGD. Rather than a distinct clinical entity, entropion is a spectrum of conditions that begins with minor meibomian gland distortions and progresses to full-blown entropion of the lid.
Meibomian gland inversion (MGI) is the lowest severity on the spectrum and begins with tarsal contraction due to the subclinical inflammation involved with MGD. The contraction/curling causes intermittent inversion of the gland orifices, particularly with blinking, and patients experience discomfort despite an absence of obvious entropion. Orifices of the glands gradually fill with epithelium and thick meibum fills the ducts. As the condition progresses, inward rotation of the lid increases and lid ptosis, trichiasis and entropion can occur with time.
MGI can be observed with a careful slit lamp exam, noting that the meibomian gland orifices of the upper lid are not visible in up gaze but can be brought into view with slight manipulation of the lid with a cotton tipped applicator. Patients often note improvement of symptoms with repositioning and following minor surgical corrections for MGI.10
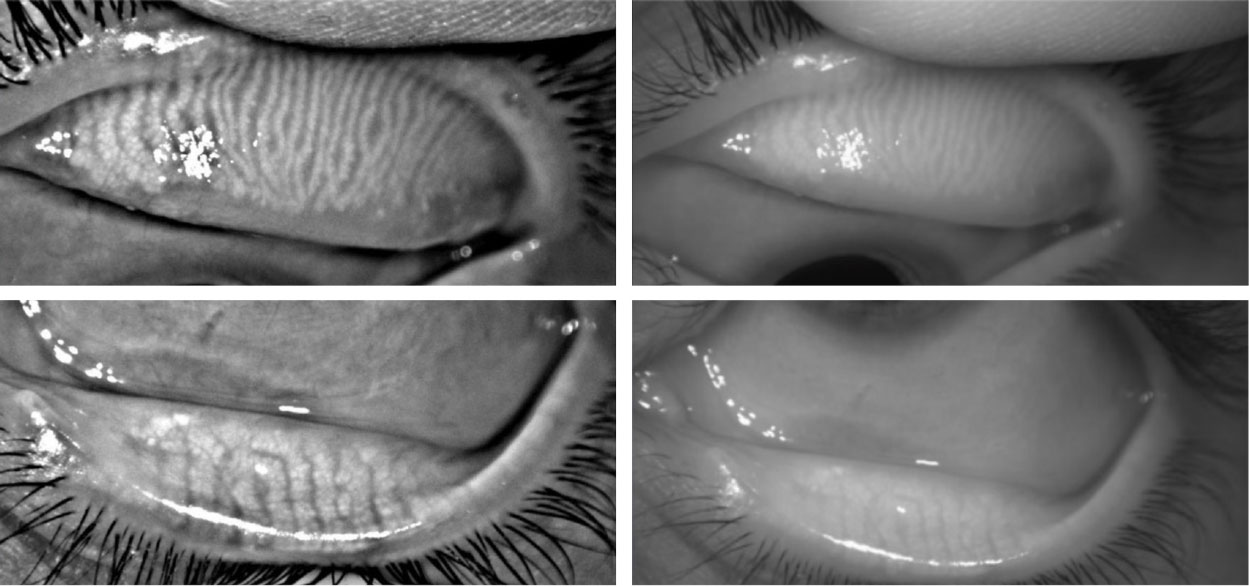 |
| Meibography images from the Oculus keratograph showing normal meibomian gland number and morphology within the upper and lower lids. Click image to enlarge. |
Surgical Procedures
These are often necessary to address gross lid malpositions and can greatly improve symptoms of dry eye and ocular irritation that patients experience. In other instances, however, surgery may be the cause of, rather than the treatment for, patients’ dry eye.
Dry eye is a relatively minor complication of blepharoplasty, but it can significantly impact patients’ quality of life and surgical satisfaction. Upper eyelid ectropion and lagophthalmos are the more common of the blepharoplasty complications. Excessive pulling or excessive removal of upper eyelid skin can cause the lid margin to evert away from the ocular surface, and dry eye ensues due to ductal hyperkeratinization and lipid deficiency.3,11,12 Pre-referral assessment for blepharoplasty should include Schirmer testing, tear break-up time (TBUT) testing and monitoring for proptosis or exophthalmos, which increase the risk of developing dry eye postoperatively.
The decision to perform blepharoplasty on an existing dry eye patient should not be taken lightly and ODs should educate dry eye patients carefully and refer thoughtfully.
Lax Eyelid Conditions
Uncertainty remains as to the exact mechanism of FES development, but theories include direct pressure on the lids due to these patients often sleeping prone or on their sides to minimize snoring, as well as intermittent systemic hypoxia during apnea.14 Connective tissues within the lid are damaged over time and result in laxity, reduced goblet cell counts and gel-forming mucins, MGD and dry eye symptoms.15,16 FES patients will demonstrate MGI and posterior migration of the gland orifices which negatively affects the tear film and causes chronic irritation with blink.17
Dry eye will additionally be worsened by eversion of lids during sleep, allowing greater tear evaporation. Symptoms will be worse in the morning upon waking and laterality can vary depending on prone, supine or side sleeping due to mechanical eversion of the lids against the pillow during sleep. Slit lamp evaluation of the upper lids will clearly identify these patients in that the upper lids will distract easily from the globe when pulled superiorly and laterally. Distraction of greater than 5mm of the upper lid is considered a diagnostically positive test. Conjunctival changes will be visible upon evaluation of the superior conjunctiva and a possible lash ptosis.15
A history of OSA may be elicited in undiagnosed patients by asking about daytime somnolence, loud snoring or nocturnal gasping or choking.18 While continuous positive airway pressure (CPAP) therapy is the gold standard treatment for OSA, it has been shown to have mixed results related to FES. Many patients reported no significant improvement to dry eye symptoms and intermittently experienced heightened instances of ocular irritation due to air leakage from poorly sealed CPAP masks. A smaller number of patients experienced improvement of symptoms and demonstrated increased TBUT values likely related to the supine sleeping position necessitated by CPAP mask wear and diminished mechanical trauma.19 For this reason, evaluation of symptomatic FES patients should always include a careful history regarding CPAP use, mask type, proper mask fitting and episodic leakage of air.
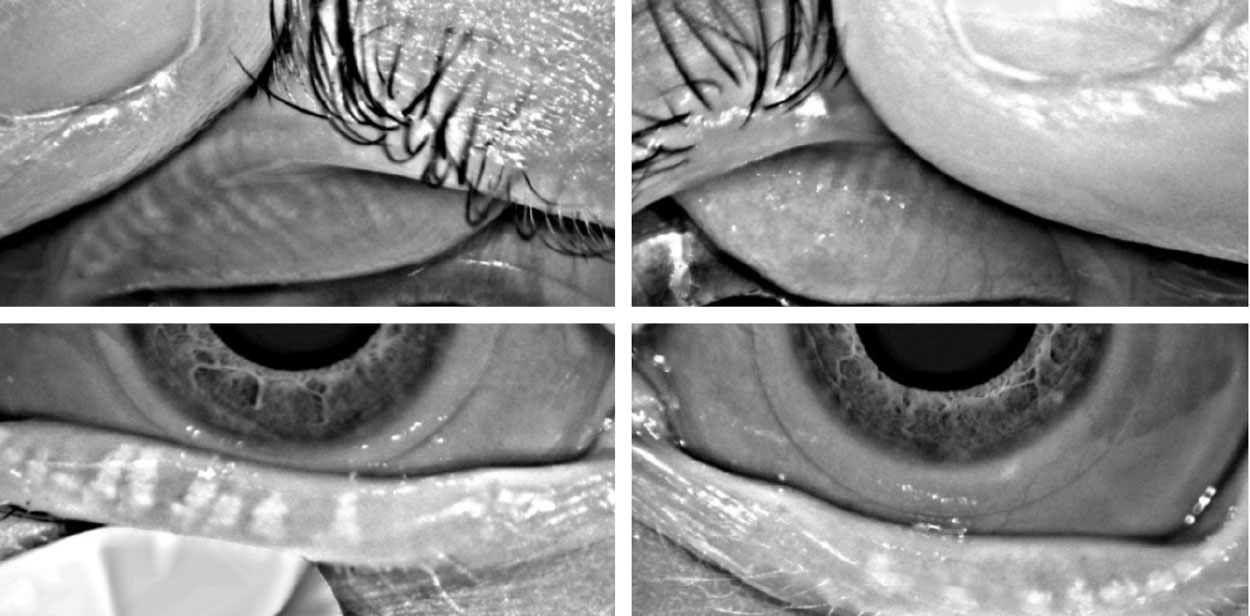 |
|
Meibography images from a long-term contact lens wearer, demonstrating mild attenuation of the meibomian glands within the upper lid and marked gland atrophy within the lower lid. Click image to enlarge. |
Sleep-related Issues
On average, a third of a person’s life is spent asleep; therefore, it stands to reason the significant impact sleep-related issues can have on ocular health. Eye closure in sleep is involuntary and requires coordinated action from the ocular muscle groups, namely relaxation of the levator palpebrae and active contraction of the orbicularis oculi.18,20 Any disruption of this coordination can negatively affect the eye’s ability to achieve full closure during sleep.
Inadequate lid seal (ILS) is the general term for incomplete eyelid closure during sleep and is a common phenomenon. Causes include thyroid eye disease, facial palsies, trauma and cosmetic procedures, but can also be found in patients with ‘normal’ lid anatomy and function. The most common factor in these patients without lid closure issues is alcohol intoxication the night before due to sympathetic muscle activity to Müller’s muscle and altered eye movements. Use of hypnotics such as Ambien, Lunesta or Sonata were also found to be contributory.18,20,21
Physiologically, tear production has a circadian rhythm and is diminished during sleep, leaving the eye in a relatively dry state, and protection from desiccation is typically provided by lid closure.22 Coupled with hypoxia due to closed lids and tear composition changes resulting in a subclinical inflammatory state, the ocular surface is at significant risk during sleep.18 This can greatly exacerbate existing dry eye disease, treatment for which can be difficult if the ILS is not addressed.
Korb et al. demonstrated that while 80% of asymptomatic patients have normal lid closure with no signs of compromised lid seal, 61% of symptomatic dry eye patients have some degree of compromise. This leads to nocturnal evaporative stress and correlates closely with the presence of moderate to severe symptoms of dry eye.23 These patients will typically have symptoms most severe upon waking, similar to FES.
While some patients present with a history of observing incomplete lid closure during sleep, others have to be evaluated in-office using the snap back test described earlier and the Korb-Blackie light test. To perform this test, the patient is placed in a semi-reclined exam chair in a fully darkened room and asked to close their eyes as if they were falling asleep. While the lids are closed, a transilluminator is placed directly against the outside of the closed upper eyelid with only enough pressure to ensure contact and avoid pulling of the lid. The practitioner then observes the presence or absence of light emanating from between the lashes of the closed lids.
Any visible light from between the lashes in the central, nasal or temporal regions of the lids is considered a positive finding for compromised lid seal. Severity can then be assessed by noting the amount of light present (0 = no light visible, 1 = minimal light, 2 = moderate light, 3 = severe light).24
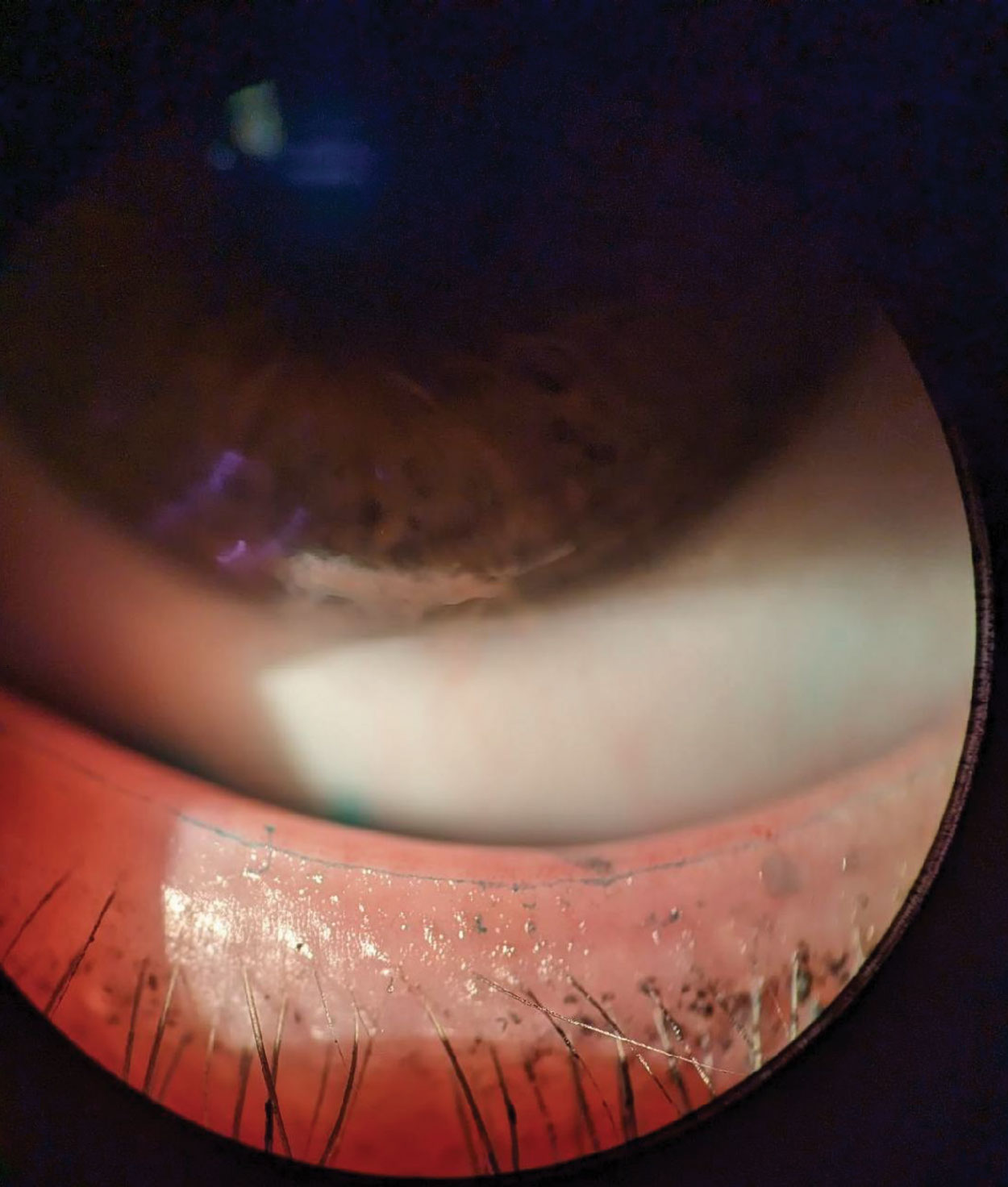 |
|
Slit lamp image of lissamine green staining of Marx’s Line on the lower lid, marking the mucocutaneous junction of the eyelid. Click image to enlarge. |
MGD: Cause and Consequence
Numerous references to the part MGD plays in dry eye and lid-related conditions have been made thus far, but now let us take a more direct look at MGD itself. In 2011, the International Workshop on Meibomian Gland Dysfunction published the results of an exhaustive literature and research review related to MGD and provided a summary of the definition, physiology and management.29 According to the group,“MGD is a chronic, diffuse abnormality of the meibomian glands, commonly characterized by terminal duct obstruction and/or qualitative/quantitative changes in the glandular secretion. This may result in alteration of the tear film, symptoms of eye irritation, clinically apparent inflammation and ocular surface disease.”30
The meibomian glands excrete lipids onto the ocular surface that form the outermost layer of the tear film overlying the aqueous and mucin layers.31,32 Meibum is a complex mix of various lipids including fats, free fatty acids and cholesterol, and serves several purposes.33 Meibum provides a smooth optical surface, reduces tear film evaporation, enhances tear film stability and spreading, forms a hydrophobic barrier to prevent tear film loss, prevents contamination of the tear film by sebum and seals the lid margins during sleep.34,12
Dysfunction. MGD can be classified into high delivery and low delivery states. High delivery or hypersecretory MGD is seen in seborrheic dermatitis, atopic disease and acne rosacea and plays a small role in overall incidence. Low delivery gland hyposecretion is seen with contact lens wear and meibomian gland atrophy. Low delivery gland obstruction is associated with retinoid use, systemic hormonal treatments and age, and is by far the most common category.31,35 65% of all dry eye patients demonstrate obstructive MGD.35 Regardless of classification, however, the net result is lipid abnormalities within the tear film leading to evaporative dry eye.34
Obstruction. Meibomian gland obstruction happens when the ductal epithelium becomes hyperkeratinized and meibum viscosity increases within the duct. With time, the obstruction leads to dilatation of the gland, gland atrophy and reduced secretions. With insufficient lipids on the ocular surface, evaporation and osmolarity increase, causing an unstable tear film and increased bacterial growth on the lid margin.30 Biofilm comprised of an over proliferation of normal Staphylococcal bacteria accumulates around the lash follicle and then progresses to the meibomian and accessory lacrimal glands of Wolfring and Krause due to decreased flushing of tears and diminished antibacterial proteins in the tear film. Bacteria increase enzyme activity within the ocular system and change the viscosity of meibum. This leads to further stasis and inflammation within the gland.31 Eventually, inflammation disrupts the structural integrity of connective tissues and glands within the lid.
At this point in the cycle, biofilm may recede as the ocular surface environment is so altered it is no longer conducive to bacterial growth.36
Evaluation. While evaluation tools such as symptom questionnaires, TBUT and Schirmer testing provide evidence of generic dry eye, additional, more focused evaluation of the glands is required to identify MGD. Lids should be viewed carefully under the slit lamp to look for MGI, gland migration and capping. Marx’s line, which occurs at the mucocutaneous junction of the eyelid, is usually visualized on the conjunctival side of the meibomian orifices when fluorescent dye is applied to the lid margin. It represents the barrier line between the lipids from the meibomian glands and the aqueous tear film and is found to shift outward to the cutaneous side of the orifices with MGD.12
Expressibility and quality of the secretions should be assessed through diagnostic expression of the glands with a cotton tipped application or digital pressure. Minimal pressure should be necessary to express a normally functioning meibomian gland and the resulting secretions should be clear and free-flowing.
Meibography and meiboscopy will demonstrate the number and morphology of the glands. Meiboscopy can be performed in-office simply by holding a transilluminator against the cutaneous side of the lid while everting the lids in a darkened exam room. The light will provide a limited view of the glands revealing any atrophy, dilatation or tortuosity. Meibographers such as the Keratograph 5M (Oculus) and LipiView II (Johnson & Johnson) capture detailed non-contact images of the meibomian glands and can be performed easily by skilled technicians.
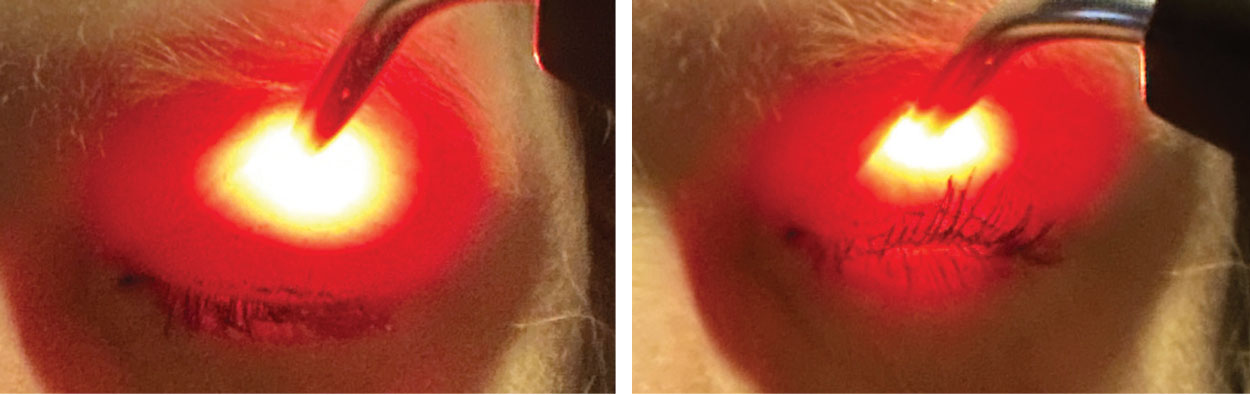 |
Korb-Blackie lid test to monitor for incomplete lid seal. Left image demonstrates artificially induced positive finding (visible light between lashes) when excessive pressure is applied to lid. Right image demonstrates proper testing procedure with no visible light between lashes. Click image to enlarge. |
Takeaways
Proper identification of the type of dry eye a patient has is the critical first step in determining an appropriate treatment plan. It’s important to screen carefully for eyelid abnormalities and MGD in all dry eye patients, keeping in mind that multiple categories and causes can occur simultaneously, and address each component separately to maximize the patient’s comfort and vision.Dr. Sanford is an attending optometrist at the Memphis VA Medical Center. She has no financial interests to disclose.
1. The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):75-92. 2. Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II Definition and Classification Report. Ocul Surf. 2017;15(3):276-83. 3. Cho IC, Kim BJ, You HJ, Tark WH. Surgical correction of upper eyelid ectropion presenting dry eye symptoms. Aesthet Surg J. 2021;41(1):NP1-9. 4. Hakim F, Phelps PO. Entropion and ectropion. Dis Mon. 2020;66(10):101039. 5. Doan S, Zagórski Z, Palmares J, et al. Eyelid disorders in ophthalmology practice: results from a large international epidemiological study in eleven countries. Ophthalmol Ther. 2020;9(3):597-608. 6. Marcet MM, Phelps PO, Lai JS. Involutional entropion: risk factors and surgical remedies. Curr Opin Ophthalmol. 2015;26(5):416-1. 7. Yang MK, Sa HS, Kim N, et al. Quantitative analysis of morphological and functional alterations of the meibomian glands in eyes with marginal entropion. PLoS One. 2022;17(4):e0267118. 8. Blackie CA, Solomon JD, Scaffidi RC, et al. The relationship between dry eye symptoms and lipid layer thickness. Cornea. 2009;28(7):789-94. 9. Finis D, Pischel N, Schrader S, Geerling G. Evaluation of lipid layer thickness measurement of the tear film as a diagnostic tool for meibomian gland dysfunction. Cornea. 2013;32(12):1549-53. 10. Siah WF, Boboridis K, Tan P, et al. Meibomian gland inversion: under-recognized entity. Acta Ophthalmol. 2019;97(8):e1116-e1122. 11. Pacella SJ, Codner MA. Minor complications after blepharoplasty: dry eyes, chemosis, granulomas, ptosis, and scleral show. Plastic and Reconstructive Surgery. 2010;125(2):709-18. 12. Yamaguchi M, Kutsuna M, Uno T, et al. Marx line: fluorescein staining line on the inner lid as indicator of meibomian gland function. Am J Ophthalmol. 2006;141(4):669-75. 13. Sward M, Kirk C, Kumar S, et al. Lax eyelid syndrome (LES), obstructive sleep apnea (OSA) and ocular surface inflammation. Ocul Surf. 2018;16(3):331-6. 14. Mastrota KM. Impact of floppy eyelid syndrome in ocular surface and dry eye disease. Optom Vis Sci. 2008;85(9):814-6. 15. Salinas R, Puig M, Fry CL, et al. Floppy eyelid syndrome: a comprehensive review. Ocul Surf. 2020;18(1):31-9. 16. Barbosa FL, Xiao Y, Bian F, et al. Goblet cells contribute to ocular surface immune tolerance-implications for dry eye disease. Int J Mol Sci. 2017;18(5):978. 17. Yvon C, Hunt S, Malhotra R. The importance of identifying meibomian gland inversion in patients with floppy eyelid syndrome. Ophthalmic Plast Reconstr Surg. 2023;39(2):156-61. 18. McNab AA. The eye and sleep. Clin Exp Ophthalmol. 2005;33(2):117-25. 19. Kadyan A, Asghar J, Dowson L, Sandramouli S. Ocular findings in sleep apnoea patients using continuous positive airway pressure. Eye (Lond). 2010;24(5):843-50. 20. Takahashi A, Negishi K, Ayaki M, et al. Nocturnal lagophthalmos and sleep quality in patients with dry eye disease. Life (Basel). 2020;10(7):105. 21. Greenlund IM, Cunningham HA, Tikkanen AL, et al. Morning sympathetic activity after evening binge alcohol consumption. Am J Physiol Heart Circ Physiol. 2021;320(1):H305-15. 22. Kawashima M, Uchino M, Yokoi N, et al. The association of sleep quality with dry eye disease: the Osaka study. Clin Ophthalmol. 2016;10:1015-21. 23. Korb D, Blackie C, Nau A. Prevalence of compromised lid seal in symptomatic refractory dry eye patients and asymptomatic patients. Invest Ophthalmol Vis Sci. 2017; 58:2696. 24. Blackie CA, Korb DR. A novel lid seal evaluation: the Korb-Blackie light test. Eye Contact Lens. 2015;41(2):98-100. 25. Alevi D, Perry HD, Wedel A, et al. Effect of sleep position on the ocular surface. Cornea. 2017;36(5):567-71. 26. Gauba V, Curtis ZJ. Sleep position and the ocular surface in a high airflow environment. Saudi J Ophthalmol. 2014;28(1):66-8. 27. Ayaki M, Kawashima M, Negishi K, Tsubota K. High prevalence of sleep and mood disorders in dry eye patients: survey of 1,000 eye clinic visitors. Neuropsychiatr Dis Treat. 2015;11:889-94. 28. Ayaki M, Tsubota K, Kawashima M, et al. Sleep disorders are a prevalent and serious comorbidity in dry eye. Invest Ophthalmol Vis Sci. 2018;59(14):DES143-50. 29. Nichols KK. The international workshop on meibomian gland dysfunction: introduction. Invest Ophthalmol Vis Sci. 2011;52(4):1917-21. 30. Nichols KK, Foulks GN, Bron AJ, et al. The international workshop on meibomian gland dysfunction: executive summary. Invest Ophthalmol Vis Sci. 2011;52(4):1922-9. 31. Baudouin C, Messmer EM, Aragona P, et al. Revisiting the vicious circle of dry eye disease: a focus on the pathophysiology of meibomian gland dysfunction. Br J Ophthalmol. 2016;100(3):300-6. 32. Arita R, Morishige N, Koh S, et al. Increased tear fluid production as a compensatory response to meibomian gland loss: a multicenter cross-sectional study. Ophthalmology. 2015;122(5):925-33. 33. Butovich IA. The meibomian puzzle: combining pieces together. Prog Retin Eye Res. 2009;28(6):483-98. 34. Nelson JD, Shimazaki J, Benitez-del-Castillo JM, et al. The international workshop on meibomian gland dysfunction: report of the definition and classification subcommittee. Invest Ophthalmol Vis Sci. 2011;52(4):1930-7. 35. Knop E, Knop N, Millar T, et al. The international workshop on meibomian gland dysfunction: report of the subcommittee on anatomy, physiology, and pathophysiology of the meibomian gland. Invest Ophthalmol Vis Sci. 2011;52(4):1938-78. 36. Rynerson JM, Perry HD. DEBS—a unification theory for dry eye and blepharitis. Clin Ophthalmol. 2016;10:2455-67. |


