Although amniotic membrane use isn’t new to medicine, it’s a relatively recent non-surgical modality within our discipline to assist in wound healing. The versatility of the product and the ability to use a non-pharmaceutical option to stimulate tissue repair and regeneration can provide a significant advantage to patient care at any level.
With eye care practitioners increasingly embracing medical care of the anterior segment and more companies providing a form of amnion to the marketplace, use of amniotic tissue in optometric practice is becoming more common. But with this increased use comes a few inevitable complications and challenges, including patient selection, application and removal techniques, understanding the patient’s physiology and setting appropriate patient expectations. These will all affect how your patient fares in both their actual and perceived response to the therapy and its eventual outcome—expedited ocular surface healing—as well the nature and severity of complications.
Below are a few tips for engaging in successful amniotic membrane therapy and managing common complications you may encounter along the way. Let’s take some time to delve into the root causes of the complications we may occasionally face and discuss how to avoid or correct them.
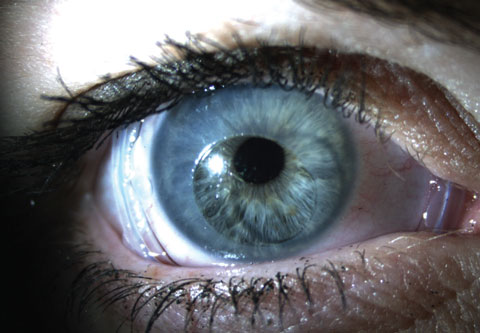 |
| Virtually any corneal or conjunctival surface disease that involves inflammation may benefit from the use of an amniotic membrane. Click to enlarge. |
Poor Insertion/Removal
Properly preparing for any allograft placement procedure can help avoid many of the complications associated with the modality. Poor preparation encourages a rushed technique and may lead to these post-procedural complications.
Corneal abrasions. Abrasions to the corneal epithelium may arise from the lid speculum, forceps, a very aggressive application or removal of an older version of the cryopreserved product containing a bare symblepharon (PMMA) ring (in more recent versions, the ring is wrapped in amnion, providing more cushion) on an unstable surface, or from eye rubbing by the patient.
If induced at insertion, patients will likely report increased foreign body sensation and discomfort, which may range from mild to severe. Generally, the abrasions induced during insertion should not cause much concern, as the properties of the membrane are intended to accelerate epithelial healing. More troubling are those occurring during removal, perhaps due to unintended contact between a bare symblepharon ring or lid speculum and a newly healing epithelium that may still be inadequately bound to the basement membrane complex and unstable.
To avoid this, make sure most supplies are close at hand and you have enough room to maneuver the patient for insertion. Positioning the patient on their back for straightforward insertion of a lid speculum and slowly draping the tissue over the cornea can help with proper graft placement and resultant retention. Directing patients to look away from the contact point of the speculum also decreases the chance of corneal contact. Rehydration of the membrane can be accomplished next (one drop at a time), and placement of the bandage contact lens should be accomplished prior to removal of the lid speculum.
Educate the patient that eye rubbing is strictly forbidden. Although irritation, discomfort and itching are all possible following insertion of the membrane, it is important to remind the patient that eye rubbing may potentially disrupt the very surface we are attempting to heal—their corneal epithelium. In addition, rubbing the eye can lead to destabilization or premature ejection of the graft, which will lessen the beneficial properties of contact with the membrane.
Poor cornea-contact lens relationship. With application of dehydrated membranes, it is imperative that a contact lens covers the membrane to hold it in place. Selecting the right contact lens is critical to the overall function and retention of the membrane. Too tight a contact lens fit will cause impingement of the limbal stem cell region, which can exacerbate issues with a healing corneal epithelium. Fit too loose and the lens can slide around, leading to premature loss of the membrane and potential ejection. I tend to select a bandage contact lens close to the curvature of the cornea, but in many cases will use an oversized soft contact lens such as a Kontur for improved retention.
Patient Intolerance
Intolerance of various portions of the amniotic membrane complex is a common yet highly patient-dependent variable. In my experience, the most important issue is setting expectations prior to insertion. Common issues surrounding intolerance are pain or discomfort with either the membrane or the symblepharon ring, improper rinsing and preparation of the membrane or excessive inflammation of the lids or conjunctival surface prior to insertion, which can lead to issues with the fit and movement of the symblepharon ring or bandage contact lens.
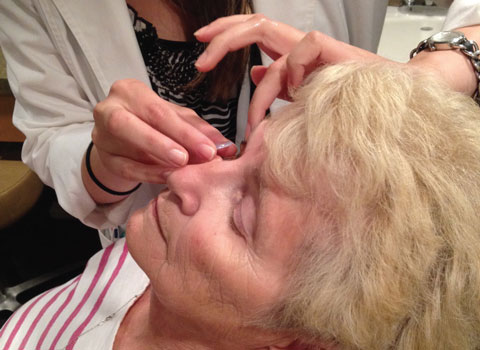 |
| Some of the biggest impediments to success happen during insertion of the membrane. Careful preparation and thorough patient education are critical. |
Product/Formulation Issues
Some potential complications are related to the specific formulation of amniotic membrane and product you have chosen.
Self-retaining cryopreserved products. With this modality, tolerability of the PMMA ring is generally the largest hurdle to overcome for the majority of practitioners. In my experience, verbalizing to the patient that the first 24 hours will be uncomfortable helps them better understand that there is some adaptation to the ring—it is normal and to be expected, and that comfort will generally improve following the first 24 hours. With the exception of patients who also undergo epithelial debridement procedures, it is rare for patients to need additional pain medications. For those who do have increased pain response, occasional use of topical bromfenac or oral ibuprofen may be used to moderate the discomfort. If needed, the amnion tissue can also be excised from the symblepharon ring complex and then draped over the cornea with an overlying bandage contact lens. This is, however, more technically challenging and time consuming, as the cryopreserved membranes are flaccid and do not position easily.
Stinging on instillation is an issue for patients only when there has not been adequate rinsing done prior to insertion. Cryopreserved amniotic membranes (Prokera specifically) are stored in Dulbecco’s modified Eagle medium, which contains amphotericin B and ciprofloxacin, as well as a lower pH, which can cause stinging if not rinsed properly. In our office, we typically will use five 15ml bottles of sterile balanced saline solution for rinsing prior to insertion. This has essentially eliminated this complaint from patients.
Ejection of the ring is a possibility. To minimize this risk, assess the fit of the ring under the slit lamp after insertion. Have the patient look in various gazes to gauge how the edge of the ring is positioned relative to the lid margin. If the upper lid is quite tight, a slightly higher risk exists that the lower portion of the ring will ride up and “teeter-totter,” similar to a rigid gas permeable lens. Applying tape to the temporal and/or nasal aspects of the lower lid in this situation to “raise” the lower lid may be useful in reducing ejection. In addition, an upper lid tape tarsorrhaphy, consisting of a single piece of tape positioned laterally to bolster the rigidity of the upper lid, will also improve comfort in many patients, as well as reduce air exposure. However, this needs to be assessed and applied immediately after insertion and before patient discharge to be successful.
If the membrane dissolves quickly, this is likely a sign of either aggressive inflammation or exposure to air, both of which can accelerate breakdown of the membrane. Amniotic membranes sequester inflammatory cells from the ocular surface in their matrix, which may contribute to breakdown of the membrane over time.1 There is no global period for amniotic membrane insertion, so if the membrane is dissolving rapidly and the patient’s condition warrants it, I recommend replacing it with a new one.
Occasionally, patients will complain of increased white discharge after membrane insertion; this seems to be more common in patients with the self-retained product. It is likely associated with irritation to the conjunctiva by the ring and is not harmful to the ocular surface in the vast majority of cases. Most amniotic membranes available on the market are either stripped of epithelium or contain devitalized tissue, which is biologically inert and does not carry complete HLA antigens responsible for an aggressive immunologic attack. In cases where some inflammation of the conjunctiva is present along with a white discharge, a small amount of topical anti-inflammatory is helpful; in my clinic, we typically use loteprednol twice daily, which reduces the amount of discharge and superficial inflammation.
Dehydrated products. The biggest issue here is ejection of the membrane from underneath the bandage contact lens. In my experience, a larger contact lens such as a Kontur 16mm diameter lens can be very helpful at retaining the membrane for the entirety of the treatment. With larger bandage contact lens use, it is imperative that limbal compression be avoided.
Pain can be an issue with dehydrated products, although it is less common in dehydrated products than with cryopreserved products due to the presence of the symblepharon ring in the cryopreserved products. Anecdotally, pain is most frequently found in patients with large epithelial defects.
On the Horizon: Amniotic DropsSoon, patients may have the benefits of amniotic tissue without the potential complicating factors of tissue placement. Drops made from amniotic fluid, reconstituted dehydrated amnion or morselized amniotic tissue will soon be available to practitioners. However, while they may sound similar in terms of therapy, they have their own list of drawbacks and limitations.In the case of amniotic fluid for ocular surface treatments, very little peer-reviewed evidence exists, with the exception of mouse models of dry eye. But with further research, this therapy may one day provide significant benefits, as one comparative study examining human amniotic fluid vs. human serum in a mouse model shows favorable outcomes. However, in cultured human epithelial cell lines, another study’s data favored serum. Morselized amniotic tissue has demonstrated efficacy in the treatment of non-healing epithelial defects. To make this product, human amniotic tissue is pulverized into a gel-like consistency, which closely approximates the consistency of a cryopreserved product. Thus, researchers hope to find it as effective as the amniotic membrane itself without the added risk and discomfort of the symblepharon ring. |
Improving Odds and Outcomes
While amniotic membranes can be a powerful medium for improving ocular surface health, the key to improving outcomes is choosing your product based on the condition and goals for therapy.
In dry eye patients, amniotic membranes can improve symptoms and ocular surface health for an average of more than four months following application and with an optimal retention time of five days.2 This should not be considered a stand-alone therapy—for best outcomes, membranes should always be used in conjunction with other forms of ocular surface therapy. While amniotic membranes provide an immunologic “boost” to the surface, there are many other issues typically in play, especially in dry eye, which should be addressed simultaneously for maximum patient benefit.
Amniotic membranes have been touted to have significant anti-inflammatory, anti-scarring, anti-neovascular, anti-pain and regenerative healing properties; however, patient response is highly variable and not all such advantages can be expected in every patient.
Recently, much of the regenerative properties of amniotic tissue have been attributed to a molecular complex called HC-HA/PTX3, a heavy-chain hyaluronic acid bound to pentraxin-3.3 Though fresh amniotic tissue has the highest amounts of HC-HA/PTX3, it is almost never used in clinical practice. Cryopreserved amniotic membrane has the next highest amount.4 Dehydrated products, due to higher levels of processing, have the lowest amounts of HC-HA/PTX3—among the reasons FDA labeling indicates dehydrated products for wound coverage rather than accelerated wound healing. One study examining the healing results following strabismus surgery showed no difference in scar formation in patients using dehydrated tissue vs. none at all.5 However, other researchers have found dehydrated amniotic membrane tissue to have greater bioavailability and sustained biochemical factor release time than cryopreserved tissue, although the results in this study do not reflect the tissues available commercially in the United States.6
Because acute inflammation from certain pathologies may be higher than what the membranes may be able to handle, in certain cases it may be prudent to consider prescribing topical steroids for one week preoperatively to reduce the inflammatory load on the ocular surface.
My Take on Treating RCEAmniotic membranes are often used for cases of recurrent corneal erosion (RCE). My treatment for patients with RCE includes epithelial debridement and gentle diamond burr polishing, along with cryopreserved amniotic membrane. Retrospective analysis reveals that the recurrence of erosions in patients receiving the combination of diamond burr polishing and cryopreserved amniotic membrane was roughly half the recurrence in patients receiving the diamond burr polishing with a bandage contact lens. |
Lifestyle choices and the overall health of the individual patient will impact the wound healing response. To optimize the environment for ocular surface healing, ask patients about the following in your preoperative discussion of benefits and risks:
- Age and sex hormones
- Dietary habits
- Diabetes and other inflammatory or autoimmune diseases
- Stress, obesity, atopic diseases
- Medications such as NSAIDs, aspirin, steroids, chemotherapy
- Alcohol use and smoking
- Healing from previous surgeries or trauma
All of these factors can negatively impact the ocular surface and the healing response. Patients who have several of these items may actually benefit to a greater degree from amniotic membrane use in healing their ocular surface more quickly than without.
In general, amniotic membrane therapy is a boon for treating ocular surface disease. For patients with moderate to severe ocular surface disease, amniotic membrane therapy can act as a great catalyst to the healing process. It is a synergistic adjunct to pharmacologic treatments and can be beneficial for a broad range of clinical entities and severities of virtually any corneal or conjunctival surface disease. Being able to identify potential issues for patients ahead of time also empowers both the doctor and patient in ensuring compliance and better outcomes. Armed with the right information, you can be confident in providing amniotic membrane therapy to patients in need.
Dr. Hauswirth is a practicing optometrist at Minnesota Eye Consultants and an adjunct clinical faculty at Southern California College of Optometry. He is an active industry consultant and speaker. Relevant disclosures: Allergan, Bausch+Lomb, BioTissue, Shire, Sun, TearScience.
|
1. Shimmura S, Shimazaki J, Ohashi Y, Tsubota K. Antiinflammatory effects of amniotic membrane transplantation in ocular surface disorders. Cornea. 2001;20:408-413. 2. Cheng AM, Zhao D, Chen R, Yin HY, Tighe S, Sheha H, Casas V, Tseng SC. Accelerated restoration of ocular surface health in dry eye disease by self-retained cryopreserved amniotic membrane Ocul Surf. 2016 Jan;14(1):56-63 3. Tseng SC. HC-HA/PTX3 purified from amniotic membrane as novel regenerative matrix: insight into relationship between inflammation and regeneration. IOVS 2016, Seattle, WA. 4. Cooke M, Tan EK, Mandrycky C, et al. Comparison of cryopreserved amniotic membrane and umbilical cord tissue with dehydrated amniotic membrane/chorion tissue. J Wound Care. 2014 Oct;23(10):465-74. 5. Chun BY, Kim HK, Shin JP. Dried human amniotic membrane does not alleviate inflammation and fibrosis in experimental strabismus surgery. J Ophthalmol. June 23, 2013. [Epub]. 6. Allen CL, Clare G, Stewart EA, et al. Augmented dried versus cryopreserved amniotic membrane as an ocular surface dressing. PLoS One. 2013 Oct;8:e78441. 7. Quinto GG, Castro-Combs J, Li L, et al. Outcomes of different concentrations of human amniotic fluid in a keratoconjunctivitis sicca-induced mouse model. Int Ophthalmol. 2016 Oct;36(5):643-50. 8. Quinto GG, Camacho W, Castro-Combs J, et al. Effects of topical human amniotic fluid and human serum in a mouse model of keratoconjunctivitis sica. Cornea. 2012 Apr;31(4):424-30. 9. Kasper K, Frank B, Geerling G, et al. Amniotic fluid eyedrops versus serum eyedrops – an in vitro study. IOVS. 2008 May;49:2353. 10. Cheng AMS, Chua L, Casas V, Tseng SC. Morselized amniotic membrane tissue for refractory corneal epithelial defects in ciccatricial ocular surface diseases. Transl Vis Sci Technol. 2016 May;5(3):9. 11. Hauswirth SG, Hom MM. Cryopreserved amniotic membrane after epithelial debridement for recurrent corneal erosion. Abstract #2453745, #4356-A0159. ARVO 2016, Seattle, WA. |

