 |
History
A 39-year-old black male presented to the emergency department complaining of worsening blurred vision in both eyes over the course of one week.He also noted weight loss, palpitations and night sweats. A cursory work up uncovered pancytopenia (deficiency of red blood cells, white blood cells and platelets) and splenomegaly. The patient was referred to an ophthalmologist to investigate the ocular issues.
 |
On the initial presentation, entering visual acuity without correction was 20/200 OD with no improvement upon pinhole testing and counting fingers at 10 feet with pinhole improvement to 20/200 OS.
There was a 15% red cap color desaturation present in the patient’s right eye.
Pupils were round, equal, reactive, and there was no presence of a relative afferent pupillary defect.
Confrontation fields were blurry, but full-to-finger counting in each eye and extraocular muscle movements were full and smooth in both of his eyes.
Diagnostic Data
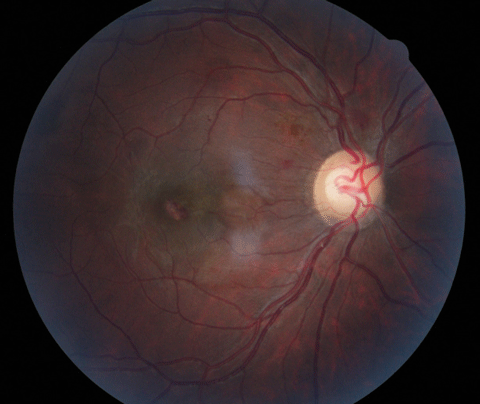
Biomicroscopic anterior segment examination found normal structures with deep anterior chambers, no evidence of inflammation, open angles and intraocular pressures measuring 8mm Hg OD and 10mm Hg OS by Goldmann applanation tonometry.The pertinent fundus findings are demonstrated in the photographs.
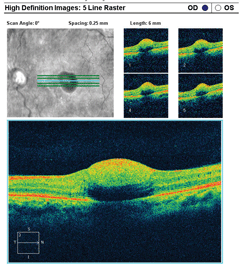 | ||
| Can the above fundus photos and these OCT images help explain our 39-year-old patient’s worsening vision blur? What can his systemic changes tell you about his diagnosis? Click images to enlarge. | ||
Your Diagnosis
Does this case require additional tests? What is your diagnosis? How would you manage this patient? What’s the likely prognosis?Answers
Additional testing included a titer for human immunodeficiency virus, testing for sepsis, hepatitis, lymphoma and leukemia. Photography was done and optical coherence tomography (OCT) was completed for the purposes of understanding the extent of retinal involvement and for monitoring resolution. The iris was also scrutinized for neovascularization.The diagnosis in this issue is leukemic retinopathy. The laboratory tests came back positive for hairy cell leukemia. Leukemia is cancer of the blood’s stem cells causing replacement of normal bone marrow tissue with malignant cells. Leukemia can result in two kinds of ophthalmic involvement: direct leukemic infiltration and indirect involvement.1 The latter can result in leukemic retinopathy often characterized by white-centered hemorrhages termed Roth spots, dilated and tortuous vessels and cotton wool spots.1
Leukemias are often classified as acute or chronic and then further divided into lymphoid or myeloid.1-3 In acute leukemia, cells are immature. In chronic leukemia, cells are well-differentiated.1-3 Lymphoid progenitors give rise to T-cells, B-cells, and natural killer cells, while the myeloid progenitors give rise to red blood cells, mast cells, basophils, eosinophils, neutrophils, macrophages and thrombocytes. In leukemia, this process is pathologically interrupted. Hairy cell leukemia (HCL) is a form of chronic lymphocytic leukemia.1 Hairy cell leukemia is named for the morphology of the cells when viewed histologically.1
HCL is treated with purine analogues with great success; with patients often achieving complete remission in 77% of cases.4 Long term follow-up shows 40% of patients relapse, however, they can be retreated with the same or different purine analogue (the expectation for complete remission decreases with each relapse).4 The use of rituximab after or with purine analogues have shown to perhaps increase complete remission rates.4
Leukemia can affect many parts of the eye and the adnexa. There can be infiltration into the conjunctiva, sclera, limbus, iris, ciliary body, and orbit. The trabecular meshwork may also be infiltrated causing acute glaucoma. Infiltration of the choroid causes thickening resulting in retinal pigment epithelium (RPE) changes resulting in atrophy, or hypertrophy. Drusen or serous detachment have been documented as well. The most common ocular tissue to be affected by leukemia is the retina.1 The main findings are dilated and tortuous vessels, intraretinal hemorrhage within any layer of the retina and Roth spots.1 Roth spots may be produced by conditions other than leukemia such as anemia, subacute bacterial endocarditis and carbon monoxide poisoning. The white center is created by a fibrin and platelet aggregate.5
A common cause of decreased acuity in cases such as these is premacular sub-internal-limiting (ILM) membrane hemorrhage.6 While these are mostly self-limiting, absorption back into the system can take months. In order to provide the patient with better vision sooner, pars plana vitrectomy with ILM peeling and aspiration of the blood can be performed.6 If patients cannot undergo surgery, Nd:YAG laser can be used to disrupt the posterior hyaloid allowing the blood to drain into the inferior vitreous thereby clearing the macula. Reports of complications such as macular hole formation and retinal detachments are side effects.3
Treatment for leukemia involves hemopoietic stem cell transplantation, chemotherapy, irradiation, and radioimmunotherapy.2 There is no treatment for leukemic retinopathy. Typically, it resolves spontaneously as the blood parameters normalize following the induction of systemic treatment.1-3
In this patient, it was decided to refer the patient to retinology to determine if a clearing procedure was a possibility. While that was being done, the patient was regularly seen by their oncologist. It was decided the best action would be to wait for spontaneous clearing which occurred in 5-12 months’ time.
Five months after the initial presentation, almost all of the macular sub-ILM hemorrhage had resolved with minor residual fluid. The best corrected vision returned to pre-event levels; 20/30+ OD and 20/25+ OS. Color vision returned to 7/7 OD and OS with Ishihara color plates and there was no red desaturation.
Finally, because the cup-disc ratios were enlarged the patient was scheduled for baseline glaucoma testing which included a 24-2 automated visual field, optic nerve OCT, gonioscopy and pachymetry.
Dr. Gurwood thanks Chris Brennan, OD, and Jeremy Han, OD, for contributing this case.
| 1. Charma T, Grewal J, Gupta S, et al. Ophthalmic Manifestations of Acute Leukaemias: The Ophthalmologist's Role. Eye 2004;18(7):663-72. 2. Kotzerke J, Bunjes D and Scheinberd DA. Radioimmunoconjugates in Acute Leukemia Treatment: The Future is Radiant. Bone Marrow Transplantation 2005;36(12):1021-6. 3. Gelston CD and Ciardella AP. Neodymium:YAG Laser Treatment for a Premacular Haemorrhage in a T Cell Lymphoblastic Lymphoma Patient. The British Journal of Ophthalmology 2006;90(10):1329-30. 4. Zinzani PL, Pellegrini C, Stegoni V, et al. Hairy Cell Leukemia: Evaluation of the Long-Term Outcome in 121 Patients. Cancer 2010;116(20):4788-92. 5. Ling R and James B. White-Centered Retinal Haemorrhages (Roth Spots). Postgraduate Medical Journal 1998;74(876):581-2. 6. De Maeyer K, Van Ginderdeuren R, Postelmans L, et al. Sub-Inner Limiting Membrane Haemorrhage: Causes and Treatment with Vitrectomy. British Journal of Ophthalmology 2007;92(7):869-72. |

