 |
Restasis (cyclosporine ophthalmic emulsion, 0.05%, Allergan) is classified as an immunomodulatory drug indicated for the treatment of keratoconjunctivitis sicca. Cyclosporine A (CsA), the active ingredient in Restasis, works by inhibiting the activation and proliferation of T-cells, which are the key players in the ocular inflammatory response.1 Yet, by virtue of its pharmacologic properties, this drug may have a wealth of additional applications in ocular inflammatory disease, particularly in situations in which corticosteroids are ineffective or contraindicated.
 |
|
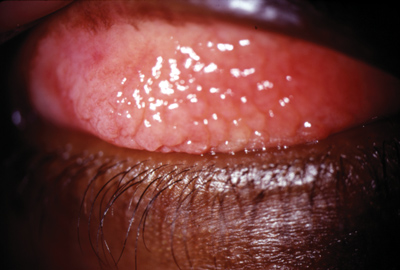
Topical cyclosporine has been shown to be beneficial in cases of vernal keratoconjunctivitis, as seen here. |
Vernal (VKC) and atopic keratoconjunctivitis (AKC). VKC and AKC are rare and extreme manifestations of the allergic cascade. VKC presents with a pronounced cobblestone papillary response, limbal infiltration by eosinophils and, in some cases, corneal erosion (shield ulcers). Symptoms include severe itching, lacrimation and photophobia. The most common therapy for VKC involves a mast cell stabilizing agent in combination with a topical antihistamine. Corticosteroids are usually required to suppress the late-phase, eosinophilmediated allergic response. The risks of long-term steroid useincluding cataractogenesis, ocular hypertension and delayed wound healingare, however, well known.
Several studies have explored topical CsA as an alternative therapy for VKC and have yielded positive results.2-6 Typically, improvement occurred after two weeks of q.i.d. therapy with 1% or 2% CsA in oil solvents. While Restasis is only 0.05% CsA, its emulsion vehicle helps to impart greater efficacy than expected for the drug concentration.7
AKC is also a severe, inflammatory ocular condition that presents with atopic lid dermatitis as well as conjunctival inflammation. More than any other form of ocular allergy, AKC has the potential to manifest corneal complications that can result in irrevocable vision loss. Corticosteroids are the mainstay of treatment in AKC, but the use of topical CsA, either alone or in combination with topical steroids, has shown beneficial results in several clinical trials.8-10 One study showed distinct benefits with the commercial 0.05% formulation of CsA used four to six times daily for four weeks in steroid-resistant AKC.10
Thygesons superficial punctate keratitis (SPK). Thygesons SPK is an idiopathic keratitis characterized by recurrent epithelial infiltration in an otherwise white and quiet eye. Patients with Thygesons SPK are typically highly symptomatic, reporting foreign body sensation, photophobia, blurred vision and excessive tearing, despite the eyes innocuous appearance. Treatment of this inflammatory disorder may involve ocular lubricants and/or bandage contact lenses. Most cases, however, require topical corticosteroidsa concern for practitioners since these patients usually require long-term therapy.
Research shows that topical CsA may also benefit patients who have Thygesons SPK.11-13 In two studies, nearly 70% of patients showed complete resolution of corneal infiltrates as long as therapy with 2% CsA continued.11,12 This is particularly significant for individuals who suffer from recalcitrant Thygesons SPK and must therefore be on chronic anti-inflammatory therapy.
Superior limbic keratoconjunctivitis (SLK). SLK is a recurrent, idiopathic perturbation of the superior palpebral and bulbar conjunctiva as well as the limbal cornea. It presents with hyperemia of the superior aspect of the conjunctiva and symptoms of burning, foreign body sensation and photophobia. Interestingly, SLK is not technically an inflammation; both corticosteroids and non-steroidal anti-inflammatory agents are ineffective.
Current treatment may involve mild interventions such as lubrication, bandage contact lenses or punctal occlusion; more invasive treatment consists of conjunctival chemocautery (0.5% to 1.0% silver nitrate) or surgical resection.14 Recently, however, a study showed that topical 0.5% CsA helped ameliorate the signs and symptoms of SLK when used as either a primary or adjunctive therapy.15
Neurotrophic keratitis. Neurotrophic keratitis is an immune-mediated corneal inflammation that results in epithelial disruption and, in some cases, sterile corneal ulceration. It is seen in patients who have severely compromised corneal innervation. These include patients with prior herpetic infections, tri-geminal palsies, systemic disease (e.g., diabetes or multiple sclerosis) or pharmacologic toxicity. Conventional therapy involves maximizing corneal hydration and providing prophylactic antibiosis. Corticosteroids are avoided in this disorder because they have the capacity to enhance collagenase activity, thereby contributing to corneal breakdown.
While no controlled studies have been conducted to date, the use of topical CsA has been advocated in the treatment of neurotrophic keratitis because it may enhance tear production and diminish inflammatory mediators that contribute to sensory corneal denervation.16
The success of CsA on these five ocular conditions indicates that dry eye represents only the tip of the iceberg regarding this drugs potential in ophthalmic care. Every now and again we encounter an unusual patient who fails to respond to conventional therapy, so awareness of this drugs ability is essential in providing these patients with the best care possible.
Drs. Kabat and Sowka are members of Alcons speakers alliance. They have no financial interest in any of the products mentioned.
1. Nussenblatt RB, Palestine AG. Cyclosporin: immunology, pharmacology and therapeutic uses. Surv Ophthalmol 1986 Nov-Dec;31(3):159-69.
2. Whitcup SM, Chan CC, Luyo DA, et al. Topical cyclosporine inhibits mast cell-mediated conjunctivitis. Invest Ophthalmol Vis Sci 1996 Dec;37(13):2686-93.
3. Bleik JH, Tabbara KF. Topical cyclosporine in vernal keratoconjunctivitis. Ophthalmology 1991 Nov;98(11):1679-84.
4. Gupta V, Sahu PK. Topical cyclosporin A in the management of vernal keratoconjunctivitis. Eye 2001 Feb;15(Pt 1):39-41.
5. Avunduk AM, Avunduk MC, Erdol H, et al. Cyclosporine effects on clinical findings and impression cytology specimens in severe vernal keratoconjunctivitis. Ophthalmologica 2001 Jul-Aug;215(4)290-3.
6. Cetinkaya A, Akova YA, Dursun D, Pelit A. Topical cyclospor-ine in the management of shield ulcers. Cornea 2004 Mar;23(2): 194-200.
7. Sall K, Stevenson OD, Mundorf TK, Reis BL. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology 2000 Apr;107(4):631-9.
8. Hingorani M, Moodaley L, Calder VL, et al. A randomized, placebo-controlled trial of topical cyclosporin A in steroid-dependent atopic keratoconjunctivitis. Ophthalmology 1998 Sep;105(9): 1715-20.
9. Hingorani M, Calder VL, Buckley RJ, Lightman S. The immunomodulatory effect of topical cyclosporin A in atopic keratoconjunctivitis. Invest Ophthalmol Vis Sci 1999 Feb;40(2):392-9.
10. Akpek EK, Dart JK, Watson S, et al. A randomized trial of topical cyclosporin 0.05% in topical steroid-resistant atopic keratoconjunctivitis. Ophthalmology 2004 Mar;111(3):476-82.
11. Reinhard T, Sundmacher R. [Local cyclosporin A therapy in Thygeson superficial punctate keratitisa pilot study]. Klin Monatsbl Augenheilkd 1996 Oct;209(4):224-7.
12. Reinhard T, Sundmacher R. Topical cyclosporin A in Thygesons superficial punctate keratitis. Graefes Arch Clin Exp Ophthalmol 1999 Feb;237(2):109-12.
13. Del Castillo JM, Del Castillo JB, Garcia-Sanchez J. Effect of topical cyclosporin A on Thygesons superficial punctate keratitis. Doc Ophthalmol 1996-97;93(3):193-8.
14. Kabat AG. Lacrimal occlusion therapy for the treatment of superior limbic keratoconjunctivitis. Optom Vis Sci 1998 Oct;75(10):714-8.
15. Perry HD, Doshi-Carnevale S, Donnenfeld ED, Kornstein HS. Topical cyclosporine A 0.5% as a possible new treatment for superior limbic keratoconjunctivitis. Ophthalmology 2003 Aug;110 (8): 1578-81.
16. Kabat AG, Reynolds SA. The role of topical cyclosporine in managing HSV-related neurotrophic keratopathy. Paper presented at the American Academy of Optometry meeting. December 12, 2004; Tampa, Fla.

