 Which is better: a $100 bottle of brand-name Xalatan or a $25 bottle of generic latanoprost? Seems like an easy question with an obvious answer, right? Your patient and the patient’s pharmacist may certainly think so, and have no qualms choosing the much cheaper generic. However, should you be as cost-conscious? After all, if the generic does not work properly, then even the $25 is a waste—and the patient’s vision may be at risk.
Which is better: a $100 bottle of brand-name Xalatan or a $25 bottle of generic latanoprost? Seems like an easy question with an obvious answer, right? Your patient and the patient’s pharmacist may certainly think so, and have no qualms choosing the much cheaper generic. However, should you be as cost-conscious? After all, if the generic does not work properly, then even the $25 is a waste—and the patient’s vision may be at risk.
Sometimes, you do indeed get what you pay for.
On the other hand, generic drugs work well enough most of the time and they offer very real cost savings—not only to individual patients, but also to the health care system as a whole.
When is it best to prescribe the generic or the brand-name drug? It’s a confounding question. But with a little more information—which we’ll cover in this article—hopefully you will have a better grasp on how to answer it for individual patients.
Based on Bioequivalence
While the initial brand-name company must submit to rigorous drug testing and lengthy clinical trials when applying to the Food and Drug Administration (FDA), companies that produce generic medications merely submit to an abbreviated form of the approval process. Safety and efficacy do not have to be proven or established with generics because thorough testing on the branded medication has presumably already been conducted. Companies merely have to show the FDA that their drugs are bioequivalent to the brand.1 Currently, in order to prove bioequivalence, scientists need to prove that the generic is in an acceptable +/- range of labeled concentration. Typically, this is accomplished by ensuring that the generic version releases its active ingredient in the bloodstream at the same speed and in the same amount as the branded drug.1
While this is relatively easy to measure with systemic medications, bioequivalence of ophthalmic drugs cannot reliably be tested in the same way. Moreover, bloodstream concentrations of an ophthalmic medication applied directly to the eye are likely not important in assessing its efficacy. So, the FDA’s only stipulations for ophthalmic generic medications is that they contain the same concentration of the active ingredient, dosage and route of administration.2
Without true bioequivalence testing for ophthalmic medications, eye care practitioners must assume that they work equally well; however, we all know this is not always the case. Most eye care providers likely remember the series of patients in the early 2000s who experienced corneal melts associated with the use of generic diclofenac ophthalmic solution (as opposed to the brand-name Voltaren) after ocular surgery. In one article, three in five patients who experienced a corneal melt were taking generic diclofenac, and four patients eventually progressed to corneal perforation that required a transplant.3
Potential Problems with Generics
Doctors and health policy analysts largely agree that generic medications can bring significant savings to health care costs. A 2002 study by the Schneider Institute for Health Policy at Brandeis University indicated that if generic usage was increased by Medicare recipients, there could be theoretical savings in billions of health care dollars.4 A 2005 report estimated that for every 1% increase in generic drug use, overall spending on prescription drugs would be reduced between $1.3 to $4 billion annually.5
On the other hand, it is possible that doctor follow-up visits may increase in number and/or frequency if the insurance company or the patient makes the decision to switch to generic medications. This could effectively nullify the health care savings.
Especially with glaucoma medications, some practitioners may be more inclined to bring patients back sooner to check the efficacy and tolerability of the generic medication than they normally would if no switch had occurred. Due to the increasing number of generic manufacturers and the potential for differing efficacy between generics, one glaucoma specialist has gone as far as to recommend tracking each type of generic by having their patients bring in the bottles to each visit. By doing this, he can track each type of generic and watch for patterns of efficacy and tolerability.6
While the FDA sets definitive guidelines for generic drugs produced in the United States, overseas manufacturing factories may not be monitored as closely. Although a firm’s application to the FDA must include a full description of the facilities it uses for manufacturing, testing and packaging, there may be budgetary restrictions that prevent every facility overseas from being inspected on the same schedule as those in the U.S.7
There is also concern over the ingredients in generic medications coming from possible sources with less oversight, such as India and China. For example, a 2005 study found that 20% of generic ciprofloxacin eye drops purchased in India were under-potent, and some preparations of the antibiotic content were low enough to negatively affect treatment outcomes.8
With generic medications, an increase in allergy symptoms from one month to the next can occur if there is a difference among manufacturers in the preservatives and/or inactive ingredients they use. If the pharmacy receives the medication from differing companies on a month to month basis, this could potentially result in more ocular surface toxicity, poor tolerability and patients returning for a “red eye” visit.
We only need to look as far back as the issues related to Travatan (travoprost, Alcon) preserved with benzalkonium chloride (BAK) to see the possibility for problems with certain preservatives.9 As previously noted, generic medications are only required to have the same concentration of the active ingredient, meaning that the inactive ingredients and the preservatives are often different. Consequently, an increased number of office visits to check efficacy and tolerability of a generic medication could theoretically cause eye care costs to increase, and offset the savings from the drug’s lower price.6
The growing number of generic manufacturers may lead to inconsistent bottle appearances and increased patient uncertainty about proper compliance. Producers of generic medications must follow manufacturing standards and comply with labeling regulations, which prohibit generic medication bottles from looking similar to their brand-name counterparts.10 This may ultimately cause confusion among patients if there is a different cap color, different label color or varying bottle size from month to month. One study found that 36% of individuals said they would be upset or worried if the color or appearance of their medication bottle changed in any form.11
Some generic medications have also been found to have different dropper sizes, which may vary the drop size, number of doses in the container, and ultimately the cost of a month’s supply.12 Additionally, concern has been expressed about the plastics used in medication bottles and quality control and what this means in terms of efficacy.6
We need to recognize the significance of our role in patient education regarding these potential differences and make sure our patients contact us if they have any questions about their treatment.
Studies on Generics
 • Glaucoma drops. Currently, the generic ophthalmic medication making the most headlines is latanoprost 0.005%, the trade version of Xalatan (Pfizer). Currently, “The Orange Book,” the FDA’s text containing “Approved Drug Products with Therapeutic Equivalence,” lists seven approved manufacturers of generic latanoprost.13
• Glaucoma drops. Currently, the generic ophthalmic medication making the most headlines is latanoprost 0.005%, the trade version of Xalatan (Pfizer). Currently, “The Orange Book,” the FDA’s text containing “Approved Drug Products with Therapeutic Equivalence,” lists seven approved manufacturers of generic latanoprost.13
The efficacy of generic glaucoma medications is of particular interest in the ophthalmic community. Although the generic may be less expensive and may provide significant savings, there is some concern that generic glaucoma medications do not have the same effect as their trade versions, and the result could be progression of the disease and loss of vision.14
However, a recent study that compared generic latanoprost to Xalatan found that both treatments demonstrated comparable safety and tolerability among patients.15 Take note, though, that 99.6% of the patients in the study were white, so these results may not be the same in African Americans, Asians and Hispanics. Also, one could argue that there may have been investigator bias because Bausch + Lomb, the producer of the generic, conducted the study.
Previously, there had been only one other study conducted on a generic formulation of latanoprost in 2007.16 The study consisted of 30 patients who were randomized into two groups; they received either Xalatan or a generic latanoprost (Latoprost [Sun Pharmaceuticals], produced in India) for the first 12 weeks, then switched to the other drug for the last 12 weeks. The investigators found that patients who were crossed over from Latoprost to Xalatan experienced a further decrease in IOP. In contrast, when subjects were crossed over from Xalatan to Latoprost, IOP rose more than 1mm Hg on average. Overall, the generic, which is formulated at a higher pH than the branded product, was found to be less efficacious at lowering IOP.16
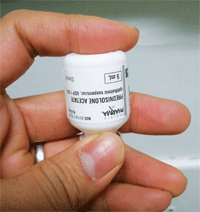
At least one study found that generic prednisolone acetate has much larger particles, which settle out of the suspension at a faster rate, sink to the bottom, and require greater shaking by the patient.
Concerns have also been raised regarding the prescribing of the generic version of timolol gel-forming solution. Researchers have shown that retention time on the ocular surface differs between various extended-release gel vehicles. Specifically, the gellan gum found in Timoptic-XE (timolol maleate ophthalmic gel-forming solution, Merck) may allow the brand-name medication to have longer surface contact, better absorption and beta-blocker activity intraocularly.17 In a 2002 study, the branded version of timolol gel-forming solution based in gellan gum was shown to lower IOP better than its generic counterpart based in xanthan gum.18 Consequently, some individuals in the medical field have proposed that considerations, such as particle size and other properties of a suspension must be evaluated for generic equivalence.19
• Steroid drops. Several studies and cases in recent years have also illustrated the need to exercise caution and vigilance when treating inflammation with ophthalmic steroids. Allergan has noted in its memorandums to doctors that the micro-fine suspension in its Pred Forte (prednisolone acetate) is more uniform, remains longer in the conjunctival sac and minimizes mechanical irritation to the eye compared to generic prednisolone acetate.20
A 2007 study looked at the differences in particle size between Pred Forte, EconoPred Plus and generic prednisolone acetate 1%.21 The assumption was that larger particles would settle out of the suspension at a faster rate, sink to the bottom, and require greater shaking by the patient. The results of the study found that the generic form of prednisolone acetate had a greater tendency for particles to agglomerate, which subsequently led to inconsistent dosage concentration.21 In addition, the authors wrote, the larger particle sizes may potentially clog the tip of the medication bottle during instillation and further alter dosage consistency and concentration. In contrast, the particle sizes in Pred Forte were smaller and more uniform, which allowed them to stay in suspension longer and gave a more precise dosage of the drop.21
Optometrists should consider that products with poorly suspended ingredients can potentially compromise the treatment of highly inflamed eyes.
Patient Perspective
• Cost and compliance. Theoretically, generic medications should improve management and patient compliance because the obstacle of cost has been removed. With a more affordable prostaglandin analog now available, patients previously on generic timolol, generic dorzolamide or those who had refused treatment may elect to take generic latanoprost. This kind of switch between classes of glaucoma mediations should also lead to significantly greater compliance due to the prostaglandin’s once-daily dosage schedule.
• Inferiority complex. Another potential issue is that patients may be resistant to going on a generic medication because they perceive them as inferior to their branded counterpart.22 Generics are often considered second-rate medications due to perceived poor compliance with standard manufacturing practices, lack of patient knowledge about generics, and influence of the brand-name company.22,23 According to a study in New Zealand, less than half of participants interviewed perceived generic medications to be safe, effective and equal in quality compared to their branded counterpart.11
Furthermore, the New Zealand study found that younger patients who had a better knowledge and understanding of drug differences were more likely than those with poor understanding of the drugs to say they would use a generic drug in both major and minor illnesses.11 We need to educate our patients so they can make an informed decision about their treatment and be willing to try a generic medication if the situation is appropriate. This is particularly true in the elderly who may be on multiple medications, as well as those with low socioeconomic status who could benefit from the savings.
• Patient preferences. We often need to consider our patient’s personality and past experience when deciding between generic and brand drugs as well. One important question to ask our patients is whether they have taken generics before and what the outcome was. If the patient had a poor experience, they may be biased towards branded medications and willing to pay the higher price.
The severity of the disease can also influence how willing a patient is to take a generic medication. According to the same study in New Zealand, individuals surveyed were more prepared to change to a generic for a minor illness than for a major illness (79% vs. 58.7%).11 In a severe disease like glaucoma, which has the potential for permanent vision loss, patients may be less willing to switch to a generic.
Which Story to Believe?
The true safety and effectiveness of generic medications cannot be certain without head-to-head clinical trials comparing topical ophthalmic brand-name drugs to their generic counterparts, but currently the data is limited.19 For now, here’s what we do:
In patients with ocular inflammation secondary to iritis, given the aforementioned research, we prefer to prescribe Pred Forte over a generic whenever possible.
A patient with advanced or end-stage glaucoma, who is at high risk for progression and vision loss, should probably stick with the brand-name medication to keep IOP under good control.
Similarly, due to the lack of information regarding the efficacy of generic glaucoma medications, in patients with advanced or end-stage glaucoma who are at high risk for progression and vision loss, we give strong consideration to staying with the brand-name medication that has the patient’s IOPs under good control. We do not want to risk losing control of the disease or cause ocular toxicity by switching from brand to generic.
Preparing a stock letter for the insurance carrier, which states that your patient has “advanced or end-stage vision-threatening glaucoma” and has been stable on his or her current medications, will be helpful if and when this situation arises. Another option is to switch patients to a branded drug for which there is no generic available at the time (i.e., switch the patient from Xalatan to a different brand-name prostaglandin), as long as similar efficacy is achieved.
Overall, we need to educate our patients on the potential for allergy and poor efficacy with generic medications. Also, more office visits may be required to evaluate the IOP-lowering effect between generic and branded glaucoma medications, as well as other ophthalmic drugs.
In addition, we need to tell patients to contact us if they experience any adverse effects after switching to a generic medication. As long as efficacy is comparable and there are no allergic reactions, generics provide an affordable alternative treatment option that should be utilized and prescribed to our patients.
As eye care providers, we need to remember that when choosing between generics and brand medications, there are two sides to the story.
Dr. Chu and Dr. Hamp see large populations of patients with advanced disease. They each lecture on glaucoma and other clinical topics. They have no financial or proprietary interests in any of the aforementioned medications and/or companies.
1. Cantor LB. Ophthalmic generic drug approval process: implications for efficacy and safety. J Glaucoma. 1997 Oct;6(5):344-9.
2. Fiscella RG. Weighing the choice between generic and branded drugs. Insert to Cat Refr Surg Today. 2010 June. Available at: http://bmctoday.net/crstoday/2010/06/insert/article.asp?f=weighing-the-choice-between-generic-and-branded-drugs (accessed January 2012).
3. Lin JC, Rapuano CJ, Laibson PR, et al. Corneal melting associated with use of topical nonsteroidal anti-inflammatory drugs after ocular surgery. Arch Ophthalmol. 2000 Aug;118(8):1129-32.
4. U.S. Food and Drug Administration. Greater Access to Generic Drugs: New FDA Initiatives to Improve the Drug Review Process and Reduce Legal Loopholes. January 2006. Available at: www.fda.gov/drugs/resourcesforyou/consumers/ucm143545.htm (accessed January 2012).
5. Subcommittee on Health of the Committee on Energy and Commerce Hearing, House of Representatives, 109th Congress. Increasing Generic Drug Utilization: Saving Money for Patients. May 18, 2005. Available at: www.gpo.gov/fdsys/pkg/CHRG-109hhrg21639/pdf/CHRG-109hhrg21639.pdf (accessed January 2012).
6. Loewen NA, Realini T, Ruderman JM, Smit B. Xalatan goes generic: a landmark event in glaucoma. Glaucoma Today. Early Summer 2011:55-8.
7. U.S. Food and Drug Administration. FDA Ensures Equivalence of Generic Drugs. Available at: www.fda.gov/Drugs/EmergencyPreparedness/BioterrorismandDrugPreparedness/ucm134444.htm (accessed January 2012).
8. Weir RE, Zaidi FH, Charteris DG, et al. Variability in the content of Indian generic ciprofloxacin eye drops. Br J Ophthalmol. 2005 Sep;89(9):1094-6.
9. Baudouin C, Riancho L, Warnet JM, et al. In vitro studies of antiglaucomatous prostaglandin analogues: travoprost with and without benzalkonium chloride and preserved latanoprost. Invest Ophthalmol Vis Sci. 2007 Sep;48(9):4123-8.
10. Gallardo MJ, Fiscella RG, Whitson JT. Generic glaucoma medications. Glaucoma Today. 2006 Jan-Feb;4(1):32-5.
11. Babar ZU, Stewart J, Reddy S, et al. An evaluation of consumers’ knowledge, perceptions and attitudes regarding generic medicines in Auckland. Pharm World Sci. 2010 Aug;32(4):440-8.
12. Kirkner RM. Myths and realities of generic latanoprost. Ophthalmic Office. 2011 June.
13. U.S. Food and Drug Administration. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Last updated: October 28, 2011. Available at: www.accessdata.fda.gov/scripts/cder/ob/default.cfm (accessed January 2012).
14. Bell NP, Ramos JL, Feldman RM. Safety, tolerability, and efficacy of fixed combination therapy with dorzolamide hydrocholoride 2% and timolol maleate 0.5% in glaucoma and ocular hypertension. Clin Ophthalmol. 2010 Nov 22;4:1331-46.
15. Allaire C, Dietrich A, Allmeier H, et al. Latanoprost 0.005% test formulation is as effective as Xalatan in patients with ocular hypertension and primary open-angle glaucoma. Eur J Ophthalmol. 2012 Jan;22(1):19-27.
16. Narayanaswamy A, Neog A, Baskaran M, et al. A randomized, crossover, open label pilot study to evaluate the efficacy and safety of Xalatan in comparison with generic latanoprost (Latoprost) in subjects with primary open angle glaucoma or ocular hypertension. Indian J Ophthalmol. 2007 Mar-Apr;55(2):127-31.
17. Meseguer G, Buri P, Plazonnet B, et al. Gamma scintigraphic comparison of eyedrops containing pilocarpine in healthy volunteers. J Ocul Pharmacol Ther. 1996 Winter;12(4):481-8.
18. Stewart WC, Sharpe ED, Stewart JA, Hott CE. The safety and efficacy of timolol 0.5% in xanthan gum versus timolol gel forming solution 0.5%. Curr Eye Res. 2002 May;24(5):387-91.
19. Fiscella RG, Gaynes BI, Jensen M. Equivalence of generic and brand-name ophthalmic products. Am J Health Syst Pharm. 2001 Apr 1;58(7):616-7.
20. Allergan Medical Affairs. Prednisolone Forte Ophthalmic Drops Medical Information. Irvine, CA, communication dated December 19, 2011.
21. Roberts CW, Nelson PL. Comparative analysis of prednisolone acetate suspensions. J Ocul Pharmacol Ther. 2007 Apr;23(2):182-7.
22. King DR, Kanavos P. Encouraging the use of generic medicines: implications for transition economies. Croat Med J. 2002 Aug;43(4):462-9.
23. Minutes of the PHARMAC Consumer Advisory Committee (CAC) Meeting, July 13, 2007; Wellington, New Zealand. Available at: www.pharmac.govt.nz/pdf/021007a.pdf (accessed January 2012).
24. Pharmaceutical Research and Manufacturers of America. Press release: R&D Investment by U.S. Biopharmaceutical Companies Reached Record Levels in 2010. March 15, 2011; Washington, D.C. Available at: www.phrma.org/media/releases/rd-investment-us-biopharmaceutical-companies-reached-record-levels-2010 (accessed January 2012).

