 |
Q:
I have a patient with early geographic atrophy (GA) of the retinal pigment epithelium (RPE) that I have followed for years and told her there is no treatment. I am hearing that a new medication has been approved, and would like to know when to refer and what to expect when comanaging?
On February 17, Syfovre (pegcetacoplan), manufactured by Apellis Pharmaceuticals, was approved for treating GA, an advanced form of dry AMD. This is a progressive condition, but clinical trials found an intravitreal dose of Syfovre could slow the growth of GA lesions by an average of 20% when taken monthly, and 17.5% when taken every other month.1 The approval was based on positive results from the Phase III OAKS and DERBY studies at 24 months.2
“The key to reaping the benefits of this medication is early use,” says Tanuj P. Banker, MD, a vitreoretinal surgeon at Center For Sight in Sarasota, FL. Once the drug became available, he performed the first injection in Southwest Florida. “For patients with GA who qualify for treatment, it is our goal to start treatment as soon as possible, as there has not been a treatment option available for those suffering from this form of macular degeneration,” Dr. Banker adds.
Patients will benefit most with long-term use of the medication. They will need injection treatments every 25 to 60 days, but Dr. Banker assures that the procedure uses multiple rounds of topical anesthetic and only lasts a few seconds. The patient may experience mild pressure during the injection; however, there is typically no significant discomfort.
 |
|
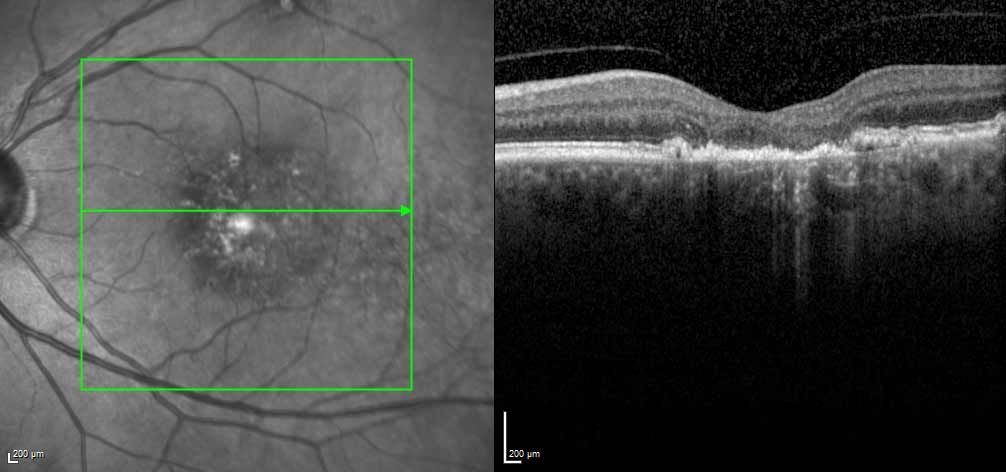
Diffuse loss of photoreceptors and RPE can be useful to detect GA. Click image to enlarge. |
Positive Effects
Apellis has warned that Syfovre could cause episodes of ocular inflammation elevated pressure, endophthalmitis, retinal detachment or development of wet AMD. In particular, there is a 12% chance of progression to wet AMD on a monthly injection schedule and a 7% chance with a bimonthly treatment plan.3
Dr. Banker’s patients have tolerated Syfovre very well thus far. His practice is currently using the medication only in patients with Medicare with a secondary insurance. However, he believes coverage will expand in the coming months to take care of those who are commercially insured.
Tackling GA Together: Comanagement is Critical
As retina specialists start using Syfovre in hopes of mitigating the retinal cell loss and visual impairment that often develops with dry AMD, they will heavily rely upon optometrists to educate and refer the proper patients. Dr. Banker believes that recognizing the early stages of the disease is critical before advanced atrophy sets in and that optometrists could closely monitor patients taking Syfovre throughout their treatment regimen.
“As you are following these patients, remember that GA correlates poorly with VA. In the study the average VA was 20/63, but treating patients with good vision provides the most potential long-term benefits,” Dr. Banker explains. “Other retina specialists are less conservative and inject patients with much better vision.”
He also recommends OCT and fundus autofluorescence (FAF) as excellent tools for detecting GA.
“On OCT, I tell my referring physicians to look specifically for (1) diffuse loss of RPE and photoreceptors, (2) loss of the external limiting membrane and (3) increased choroidal transmission,” he says. “On FAF, look for depigmented hypoautofluorescent areas.”
In addition to OCT, Dr. Banker finds red-free photography very helpful for high-risk markers to identify potential intermediate AMD patients who are at risk.
Dr. Ajamian is board certified by the American Board of Optometry and serves as Center Director of Omni Eye Services of Atlanta. He is vice president of the Georgia State Board of Optometry and general CE chairman of SECO International. He has no financial interests to disclose.
1. What is Syfovre? Syfovre. syfovre.com/about-syfovre/what-is-syfovre. Accessed March 26, 2023. 2. FDA approves Syfovre (pegcetacoplan injection) as first and only treatment for greographic atropy (GA), a leading cause of blindness [press release]. Apellis. investors.apellis.com/news-releases/news-release-details/fda-approves-syfovretm-pegcetacoplan-injection-first-and-only. February 17, 2023. Accessed March 26, 2023. 3. Side effects. Syfovre. syfovre.com/about-syfovre/side-effects. Accessed March 26, 2023. |

