 |
Although the market may be saturated with dry eye disease (DED) treatments, there is a new medication with the potential to separate itself from the rest. Last month the FDA accepted a New Drug Application (NDA) for CyclASol, a proposed treatment for DED, with a potential FDA approval of June 2023. This means, based on an initial review, the FDA considers the NDA complete for full review.
While calcineurin inhibitor immunosuppressants have been with us for more than 18 years, I believe this one is unique enough to make a difference and stand out for our patients.
CyclASol
This medication has unique components and clinical findings that are worth noting. While cyclosporine A is a known potent anti-inflammatory and selective immunomodulatory, it is extremely lipophilic, or non-water-soluble. Some companies have been successful using nanocellular spheres (Cequa, Sun Pharmaceutials) to engulf the cyclosporine molecule and deliver it to the proper tissue’s sites, but others, like generic cyclosporine in Canada, have failed due to this highly lipophilic issue. In the case of CyclASol, it is completely soluble in a solution known as perfluorobutylpentane, a novel, water-free carrier, which allows for bioavailability at the target tissue and improves efficacy. This novel carrier of CyclASol contains no oils or surfactants and is preservative-free. Research shows that it improves tolerability and decreases visual disturbances.1
 |
|
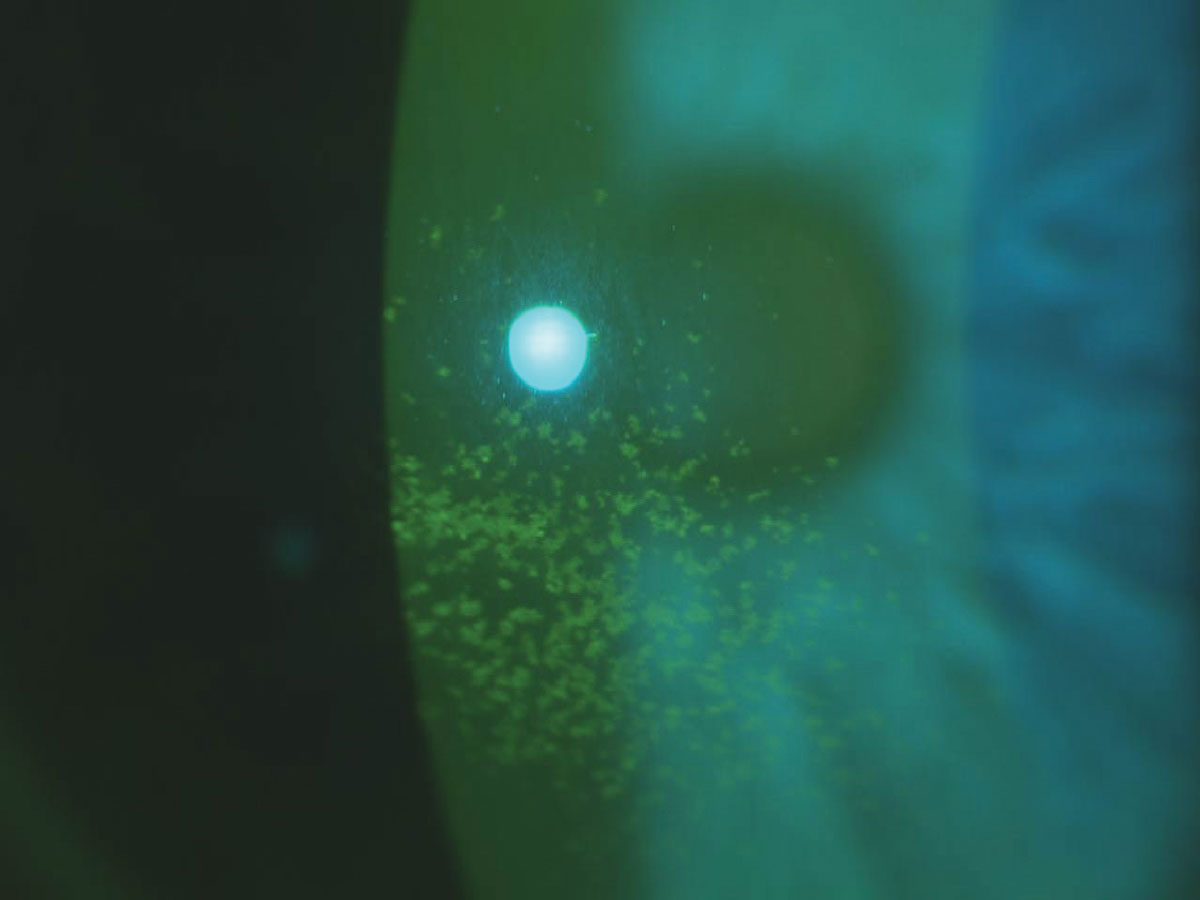
Patients with central corneal staining, as seen here, benefitted most from CyclASol treatment in the drug’s clinical trials. Click image to enlarge. |
Presentation
The NDA is supported by safety and efficacy results in over 1,000 DED patients from four clinical studies: a Phase II dose-finding study, the ESSENCE-1 and ESSENCE-2 trials and an open label extension study lasting over one year. The patient population investigated had significant ocular surface damage or staining, low Schirmer’s test scores and high symptom scores, characterizing them as moderate to severe, predominantly aqueous-deficient, DED patients.
In ESSENCE-1 and ESSENCE-2, CyclASol demonstrated a clinically meaningful and statistically significant improvement in numerous DED findings.1 It showed a statistically significant reduction in total corneal fluorescein staining scores over vehicle as early as day 15, which was the first post dosing study visit. Differences in total corneal fluorescein staining were also significantly superior over vehicle at the primary endpoint visit on day 29. The central region of the cornea benefitted most from CyclASol treatment. Conjunctival staining scores also improved statistically over vehicle at day 29. The previous Phase II study showed that the effects on the ocular surface are greater and onset was faster compared with Restasis, which was used as comparator.2
A novel aspect of this study is that it included responder analyses for total corneal fluorescein staining to evaluate whether the results were clinically meaningful for the patient. A responder was defined as an improvement of three or more grades on the NEI scale. Such a difference is considered immediately noticeable and clinically relevant, according to experts in the field. A total of 71.6% of the patients responded within four weeks with an improvement of three or more grades in total corneal staining. The proportion of responders was statistically significantly higher compared with vehicle-treated patients in both studies. Notably, responders also showed improvements in almost all symptoms compared with non-responders at day 29, underlining the clinical importance and correlations to symptom improvement at this level of cornea staining reduction.2
Ocular surface damage identified via corneal staining is an important DED indicator and requires treatment. Corneal staining has been recognized by some cornea specialists as the single most important clinical sign of DED, as it indicates the level of epithelial damage and visual impairment.3
 |
|
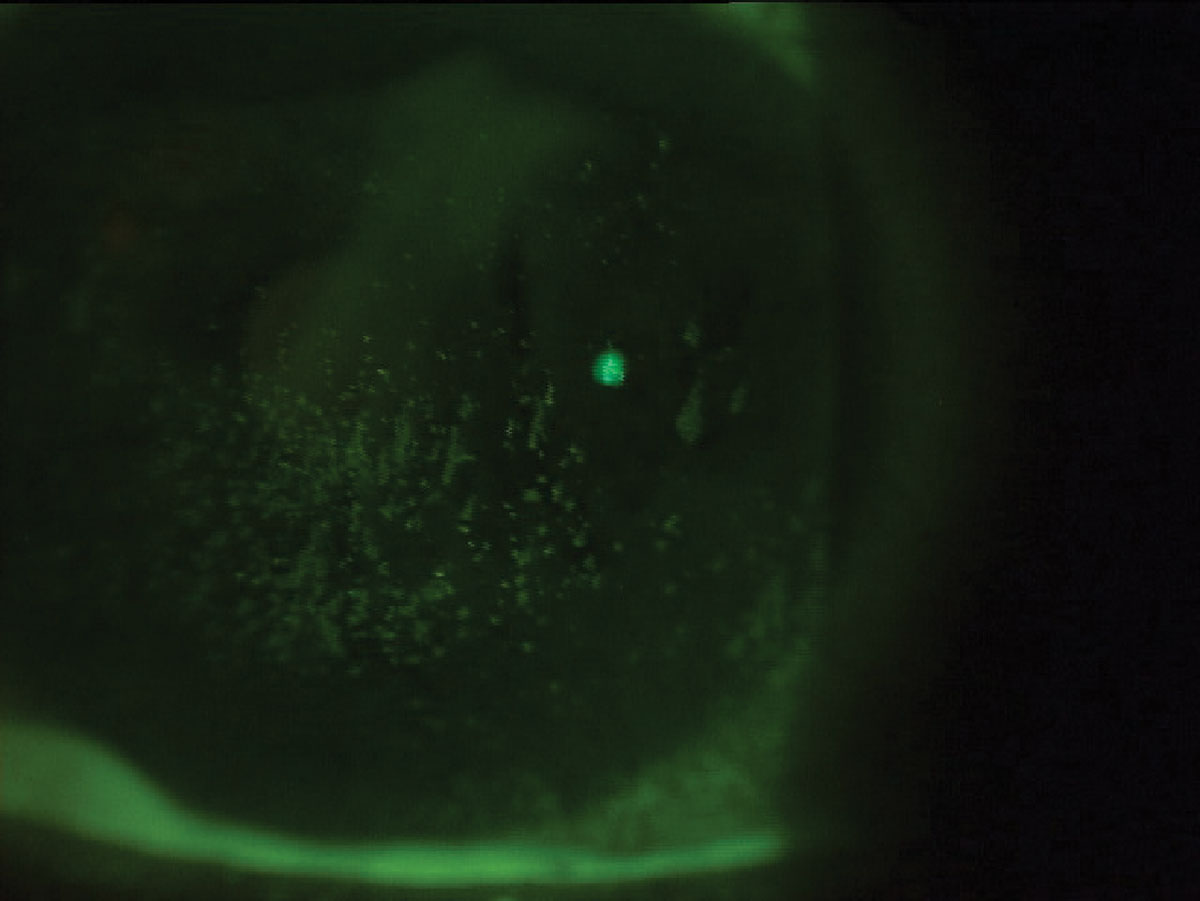
Nearly 72% of CyclASol patients presenting like this could expect three or more grades of improvement on the NEI scale. Click image to enlarge. |
The impact of corneal staining on visual function was confirmed in the CyclASol clinical trial program. In both studies, subjects with high central corneal staining at baseline benefited from CyclASol with a statistically significant improvement in their blurred vision score. CyclASol also showed positive effects on tear production. In both studies, statistically significant more patients using CyclASol showed an increase of ≥10 mm from baseline in Schirmer’s tear test score (Schirmer responder analysis), confirming a known effect of cyclosporine on the ocular surface.
Over the 52-week treatment period, all measurable effects of CyclASol were maintained and even improved for the majority of endpoints. Notably, all symptom scores reached their minimum at the last visit after one year of treatment with CyclASol, indicating continuous symptom improvement and the patient benefit of a chronic treatment.
Patient acceptance and comfort is often a challenge using DED therapies, including generic forms of cyclosporine. This may be one of the larger differentiators of CyclASol. High tolerability of CyclASol was demonstrated in both studies using drop comfort patient ratings, which show comfort level similar to that of aqueous-based artificial tears. Additionally, more than 80% of all patients selected positive descriptors when describing how the medication feels on the eye, with the most frequent descriptors being “comfortable, smooth and soothing.” The most common adverse reaction or side effect observed was instillation site discomfort. All but one patient rated it as mild.
Patients looking for relief are likely to cheer the release of CyclASol, if the drug gets the green light from FDA. Based on this clinical data, CyclASol seems to be effective in treating both signs and symptoms of DED with an excellent tolerability profile. The rapid onset of effect, the magnitude of improvements on corneal epithelial damage, the comfort or symptom scores and relatively low adverse event profile are exciting potential differentiators to existing therapies.
Dr. Karpecki is the director of Cornea and External Disease for Kentucky Eye Institute, associate professor at KYCO and medical director for Dry Eye Institutes of Kentucky and Indiana. He is the Chief Clinical Editor for Review of Optometry and chairman of the affiliated New Technologies & Treatments conferences. A fixture in optometric clinical education, he provides consulting services to a wide array of ophthalmic clients. Dr. Karpecki’s full disclosure list can be found here.
1. Sheppard JD, Wirta DL, McLaurin E, et al. A water-free 0.1% Cyclosporine A solution for treatment of dry eye disease: results of the Randomized Phase II/III ESSENCE Study. Cornea. 2021;40:1290-7. 2. Wirta DL, Torkildsen GL, Moreira HR, et al. A clinical phase II study to assess efficacy, safety and tolerability of water-free Cyclosporine formulation for treatment of dry eye disease. Ophthalmology. 2019;126:793-800. 3. Starr CE, Gupta PK, Farid M, et al. An algorithm for the preoperative diagnosis and treatment of ocular surface disorders. J Cataract Refract Surg. 2019;45(5):669-84. |

