 |
A 63-year-old Hispanic female presented with a two-day history of blurred vision in her left eye. She reported that, while working on an Excel spreadsheet, parts of the line of text were missing. She said her right eye was “perfect,” but, when she covered it, she could immediately see her central vision was blurred. Her last eye exam was three to four years ago. She wore a hyperopic correction with a progressive lens. Medical history was significant for hypertension and osteoporosis. She is currently on hydrochlorothiazide and losartan.
On examination, entering distance acuities measured 20/20 OD and 20/40 OS. Her extraocular motility testing was normal. Confrontation visual fields were full-to-careful finger counting OU. The pupils were equally round and strongly reactive; there was no afferent pupillary defect. Amsler grid testing in the right eye was normal. The left eye showed central metamorphopsia. The anterior segment examination was remarkable for trace nuclear sclerosis OU.
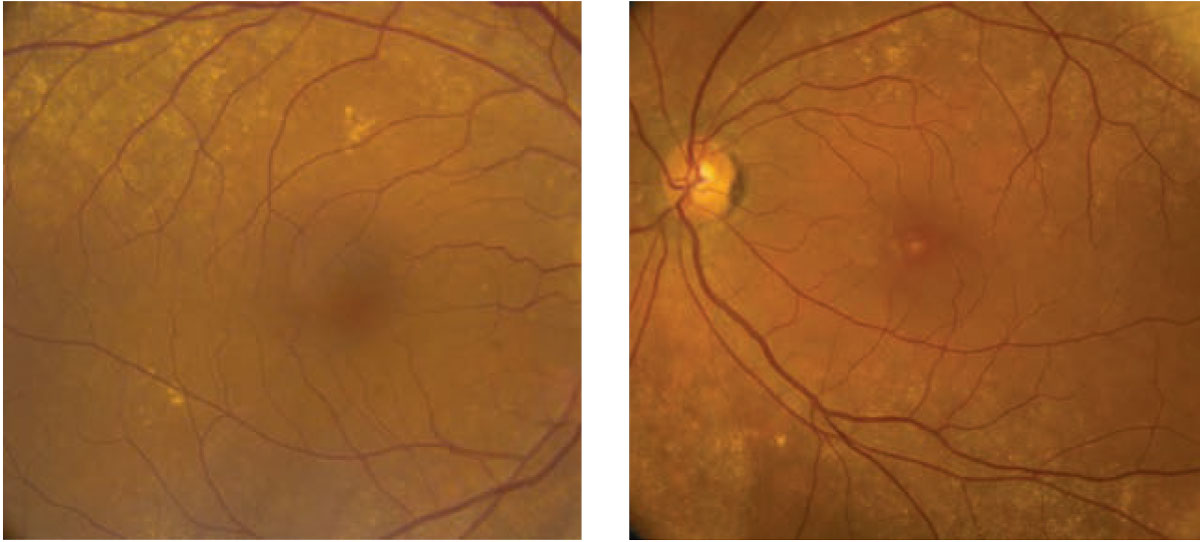
On dilated fundus exam of the right eye, there were peripheral drusen along the arcades. The macula appeared normal. We noted similar peripheral drusen in the left eye. In the macula, there was a deep yellow-white lesion (Figure 1). There did not appear to be any subretinal fluid. OCT and fluorescein angiography (FA) were performed and are available for review (Figures 2 and 3).
|
| Fig. 1. This is a fundus photograph of the right and left eyes of our patient. Note the macular appearance of the left eye. Click image to enlarge. |
Take the Retina Quiz
1. How would you describe the OCT appearance?
a. Hyperreflective lesion, likely CNV
b. Focal disruption of the outer retina
c. Outer retinal placoid lesion
d. Focal pigment epithelial detachment
2. What is the most likely diagnosis?
a. Solar maculopathy
b. Wet age-related macular degeneration (AMD)
c. Multiple evanescent white dot syndrome (MEWDS)
d. Unilateral acute idiopathic maculopathy (UAIM)
3. How should this patient be managed?
a. Observation
b. Anti-VEGF injection
c. Prednisone PO
d. Acyclovir PO
4. What would you expect her clinical prognosis to be?
a. Reasonably good central vision with treatment
b. Slow steady improvement of her vision over time
c. Rapid decline in her central vision without treatment
d. Impossible to know
For answers, see below.
Discussion
Based on the patient’s history of sudden blurred vision in the left eye and the presence of drusen along the arcades, our initial thought was that she had macular degeneration and had likely developed a choroidal neovascularization in her left eye. The problem is that she didn’t have any drusen in the macula of her right eye, which you would expect to see if she had AMD, as the dry form is usually a bilateral symmetric disease.
There was a deep yellow-appearing lesion in the macula that could possibly be a drusen, but the other clinical findings in the left eye don’t fit with CNV. The macula appeared flat, and there was no subretinal fluid or hemorrhage present. Instead, we saw some retinal pigment epithelium (RPE) disruption and this peculiar yellow lesion.
|
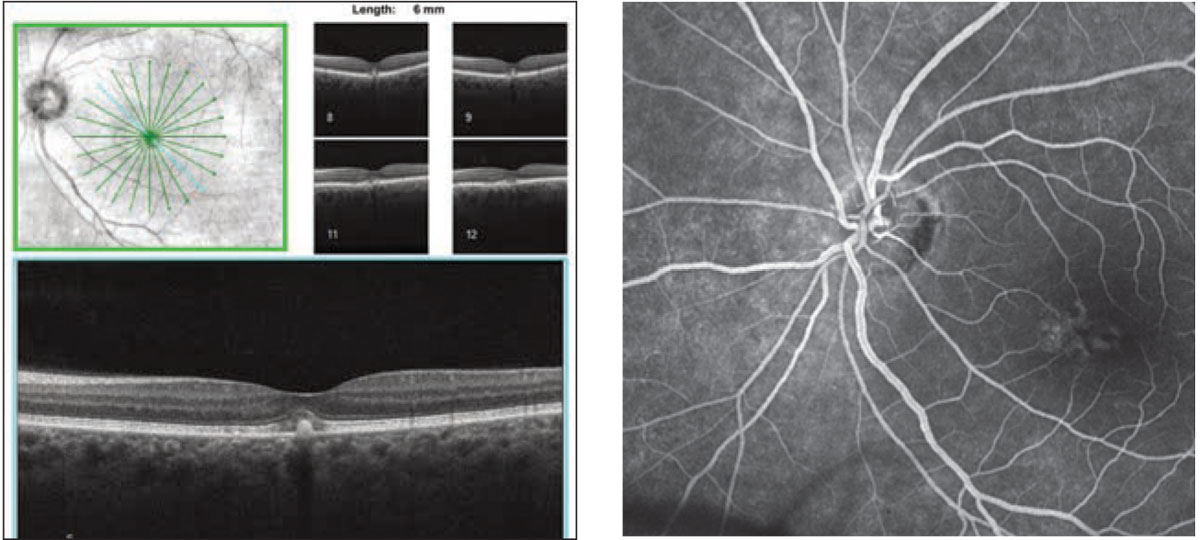
| Figs. 2 and 3. How do you interpret this SD-OCT of the left eye (left)? What do the macular changes represent in this late-phase FA of the left eye (right)? Click image to enlarge. |
The OCT showed a focal hyperreflective lesion in the outer retina corresponding to the yellow lesion with central IS/OS junction disruption. There was no subretinal or intraretinal fluid, which was consistent with our clinical exam. The FA showed a central area of hypofluorescence with surrounding hyperfluorescent staining but no leakage of the fluorescein dye, which is not consistent with CNV. Interestingly, there was a larger irregular area nasal to the fovea where there was also staining. So, what’s going on with our patient?
This appeared to be an acute, unilateral process affecting the outer retina. Based on the clinical appearance and OCT findings, this is likely a post-viral process, such as UAIM.
Atypical Findings
UAIM was originally described as sudden vision loss following a flu-like illness.1 All the participants with UAIM in one study had an exudative macular detachment, spontaneous resolution of the macular changes and near-complete recovery of their vision.
The researchers felt the clinical course and macular appearance were suggestive of an inflammatory disease of the RPE.1 They were not able to determine a causative etiology, but some case reports suggest possible infection from coxsackievirus.2
Our patient did not report having a preceding viral-like infection nor did she have an exudated macular detachment. However, since the initial description, other case reports of UAIM show that not all patients present with neurosensory retinal detachment. In one published series, neurosensory detachment was seen within 48 hours of the onset of symptoms and improved over the first week.3
Despite not having an exudative retinal detachment, other notable characteristics included the circumscribed central granular yellow lesion as well as OCT findings that described a focal defect involving the outer retina and ellipsoid layer.
Interestingly, patients with UAIM also have a thickened choroid. We did not do enhanced depth imaging on our patient, so we don’t know if she had a thickened choroid. The FA findings are also consistent with what has been described. FA shows early central hypofluorescence and parafoveal patchy hyperfluorescence without leakage, which was exactly the same case with our patient.
The relationship between UAIM and coxsackievirus is interesting, but the virus has not been clearly established to be a causative agent in all cases. Coxsackievirus is a viral infection that may cause hand, foot and mouth disease, orchitis and epididymitis. It is usually seen in children, but adults can also get it. There is such a close relationship between UAIM and coxsackievirus that some authors wonder if UAIM should be renamed “coxsackievirus maculopathy.”2 Our patient did not have a preceding viral-like illness, so it’s unlikely she had coxsackievirus.
Another atypical feature of our patient was her age. Most reports of UAIM describe patients as young adults, no older than mid- to late-30s.1,4 However, a more recent case report described a 59-year-old patient who developed UAIM 30 days after developing yellow fever, which was much closer to our patient who was 63.5
We elected to observe our patient without treatment. She returned for follow-up two weeks later. Her vision had improved to 20/30, and she had less metamorphopsia. She did not return for follow-up after that.
Retina Quiz Answers
1) b; 2) d; 3) a; 4) a.
| 1. Yannuzzi LA, Jampol LM, Rabb MF, et al. Unilateral acute idiopathic maculopathy. Arch Ophthalmol. 1991;109(10):1411-6. 2. Beck AP, Jampol LM, Glaser DA, Pollack JS. Is coxsackievirus the cause of unilateral acute idiopathic maculopathy? Arch Ophthalmol. 2004;122(1):121-3. 3. Nicolo M, Rosa R, Musetti D, et al. Early swept-source optical coherence tomography angiography findings in unilateral acute idiopathic maculopathy. Ophthalmic Surg Lasers Imaging Retina. 2016;47(2):180-2. 4. Freund KB, Yannuzzi LA, Barile GR, et al. The expanding clinical spectrum of unilateral acute idiopathic maculopathy. Arch Ophthalmol. 1996;114(5):555-9. 5. Dompieri RC, Manzano RPA, Frazão MAM, et al. Unilateral acute idiopathic maculopathy secondary to yellow fever disease: a case report. Am J Ophthalmol Case Rep. 2019;15:100464. |

