 |
Q:
I have a patient who was put on Rhopressa (netarsudil, Aerie Pharmaceuticals) after other medications did not work. Her pressure lowered significantly, but six weeks later her eyes turned very red, and she’s now mortified to go out in public. Lumify (brimonidine tartrate ophthalmic solution 0.025%, Bausch + Lomb) didn’t help quiet the eyes. Is this a common problem?A:
“I’ve personally had a fair amount of clinical success prescribing Rhopressa to treat glaucoma; however, ocular hyperemia does occur at some level in roughly 50% of patients who begin netarsudil 0.02% therapy,” says Chris Wroten, OD, of Bond-Wroten Eye Clinic in Louisiana. As many as one in five patients experience conjunctival hemorrhage, and 5% to 10% manifest eyelid and/or instillation site erythema, so red eyes are not uncommon.1
“Although a number of patients do experience side effects such as these, most are mild and tolerable, so netarsudil is certainly a welcome addition to our glaucoma treatment arsenal,” Dr. Wroten notes.
Two other side effects of note are corneal verticillata, which can occur in up to 20% of patients and is usually after four or more weeks of therapy, and blurred vision, which is reported in 5% to 10% of those taking netarsudil.1
Dr. Wroten has had three patients whose intraocular pressure (IOP) responded well to the medication but were forced to discontinue therapy due to significantly blurred vision.
However, he has also had two patients with uncontrolled IOP on maximum topical glaucoma therapy who experienced a nearly 40% reduction in pressure when netarsudil was added—atypical cases.
“It will be interesting to see if tachyphylaxis develops because netarsudil was found to be most effective when IOP was below 26mm Hg at initiation of therapy,” Dr. Wroten says.2
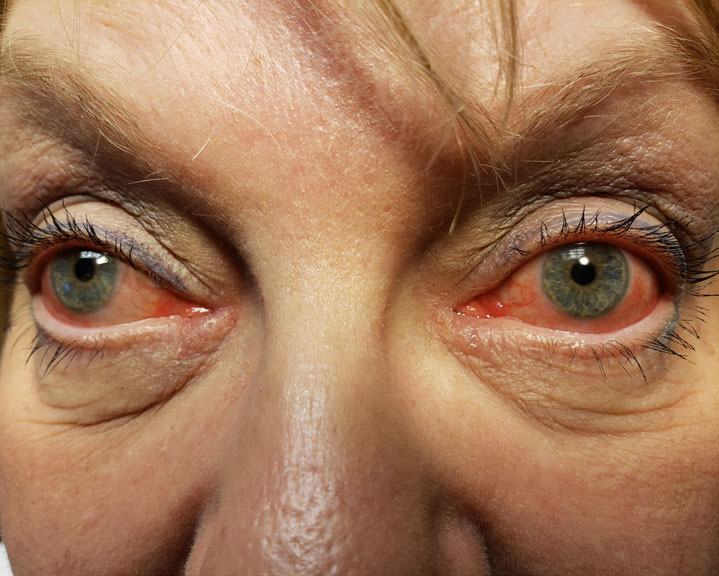 |
| While netarsudil therapy may be effective, patients can experience conjunctival hyperemia. |
A New Hope
Given the side effects, Dr. Wroten had no choice but to stop the patient’s medication. A week later, her eyes were dramatically quieter, but it will take time for the corneal changes to resolve. Because the patient also presented with cataracts, he determined that she would be a great candidate for minimally-invasive glaucoma surgery (MIGS) to manage her IOP in conjunction with cataract surgery.
MIGS devices and procedures that shunt and/or reduce aqueous production are becoming increasingly attractive options for patients. “MIGS should be considered as adjunctive therapy, especially when topical therapies have failed or are poorly tolerated,” Dr. Wroten says.
In the hands of an experienced surgeon, they are quick and effective, with device instillation occurring immediately after the intraocular lens is inserted in the capsular bag.
If this patient was not a candidate for cataract surgery, then selective laser trabeculoplasty would be another attractive option. This procedure is well-tolerated and effective for about 80% of patients, lowering IOP by 20% to 30% for about 24 months on average.2 It is also repeatable once the effect wears off, with similar therapeutic effect expected. More invasive filtering and valve surgeries were probably not warranted given her mild stage of glaucoma.
With our patient, Dr. Wroten had tried just about every medication, so the laser and surgical options were all that were left. He controlled her IOP at 12mm Hg after the iStent Inject (Glaukos) was put in, and she is only on a beta blocker now.
“Fortunately, with today’s diagnostic technologies and treatment options, patients with glaucoma have a far better prognosis than ever before,” Dr. Wroten says.
| 1. Rhopressa. Prescribing Information. rhopressa.com/assets/pdf/RHOPRESSA-Prescribing-Information.pdf. Accessed July 3, 2019. 2. Khouri AS, Lari HB, Berezina TL, et al. Long term efficacy of repeat selective laser trabeculoplasty. J Ophthalmic Vis Res. 2014;9(4):444-8. |

