 |
History
A 69-year-old Caucasian male presented with a chief complaint of blurry vision in the left eye for one month. Associated symptoms included redness, photophobia and a dull ocular ache. The patient denied trauma. His ocular history was remarkable for cataract surgery in both eyes a year earlier. His medical history included hypertension, hypercholesterolemia and depression, for which he was properly medicated with amlodipine, chlorthalidone, lisinopril, atorvastatin and fluoxetine. He also reported current tobacco use. His resting blood pressure reading was 156/79mm Hg. He denied allergies of any kind.
Diagnostic Data
His best-corrected entering visual acuities with -1.00D spheres were 20/20 OD and 20/80 OS with improvement upon use of the pinhole to 20/25 OS. Extraocular muscles and confrontation fields were full in both eyes. His right pupil was round and demonstrated a reactive reflex to light with no afferent pupillary defect. The left was mid-dilated, exhibiting a grade 3+ afferent pupillary defect. Color desaturation testing demonstrated a 30% red desaturation in his left eye, and brightness testing uncovered 30% diminished light sensitivity. Refraction uncovered -1.25D spheres in both eyes, improving acuity in the right to 20/60.
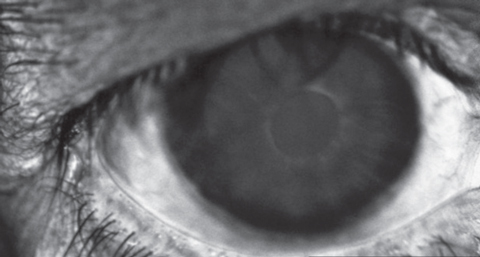
Biomicroscopy of the right eye was unremarkable. The left eye exhibited 1+ diffuse conjunctival injection, a clear cornea, a quiet anterior chamber and open 2x2 Van Herick angles. A critical anterior segment finding is illustrated in the photograph. Intraocular pressures by Goldmann tonometry measured 10mm Hg OD, 20mm Hg OS.
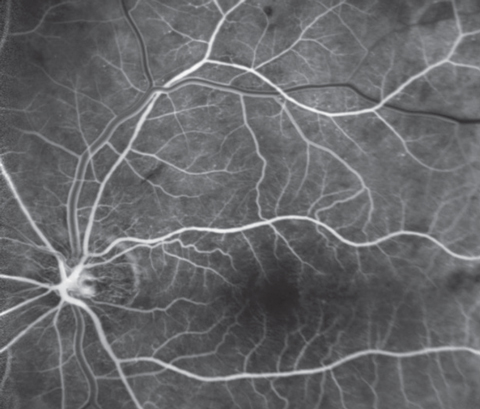
A dilated fundus examination found optic nerves that were flat with 0.4 round cup-to-disc ratios and healthy rim tissues in both eyes. An isolated splinter hemorrhage was visible superotemporal to the optic disc in the right eye. Maculae were flat and intact in both eyes. The periphery in the right eye was normal, but the left revealed mid-peripheral intraretinal hemorrhages, dilated veins and arteriolar narrowing in all quadrants.
Your Diagnosis
Based on the information provided, what’s the likely diagnosis?
 |
 |
 |
| Can any of these images help point to the diagnosis for this 69-year-old patient who underwent cataract removal a year earlier? |
Diagnosis
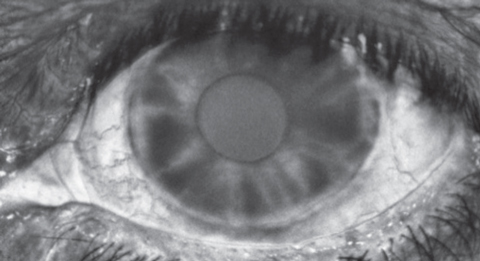
Additional testing included gonioscopy of the right eye, which found an open angle visible to the ciliary body (D40F) with no pigment, exfoliation, neovascularization or angle recession 360°and gonioscopy of the left eye, which found 360° of non-contiguous neovascularization with nasal angle closure that did not open upon compression technique. Additionally, we created fundus photos of both nerves and posterior poles, iris photos of the neovascularization in the left eye and peripheral fundus photos of the mid-peripheral hemorrhages in the left eye. A fluorescein angiogram was performed revealing reduced perfusion in the left eye (insufficient choroidal and retinal filling).
Ophthalmodynamometry was considered but not completed.
Discussion
The diagnosis in this case is ocular ischemic syndrome (OIS). The estimated incidence of OIS is 7.5 cases per million persons per year with a mean age of onset of 65 years, rarely occurring young than age 50.2 Most cases are unilateral with approximately 20% of cases presenting bilaterally.3 There is a male predilection for the disease, correlating with the higher incidence of cardiovascular disease in this gender.2 The most common cause of OIS is carotid artery occlusion, typically caused by atherosclerosis. Other known etiologies include giant cell arteritis, fibrovascular dysplasia, or other inflammatory conditions that result in carotid stenosis.4
Generally, patients with OIS have stenosis of 90% or more of the ipsilateral common or internal carotid arteries with poor collateral circulation between the external and internal carotids or the two internal carotids.5,6 The affected artery has been shown to be 100% blocked in approximately 50% of all cases.4-7 Presumably this obstruction reduces perfusion within the central retinal artery, ophthalmic artery and choroid.4,7Most patients experience reduced vision in the affected eye due to retinal ischemia.4,5,8 If care is delayed, progressive vision loss may occur from optic nerve damage secondary to variations of ischemic optic neuropathy and neovascular glaucoma. Vision ranges from 20/20 to hand motion. In one study, loss in excess of 20/400 was seen in 65% of patients.6,8 Loss of vision can be acute or may evolve over months.9 About 40% of patients complain of a dull ache in the orbital region.7 This ocular angina is caused by increased IOP secondary to neovascular glaucoma or hypoxia and may be relieved when the patient is supine. The pupil of an involved eye is typically fixed and mid-dilated having a sluggish response to light secondary to ischemia and atrophy of the sphincter muscle.5
Patients typically present with iris neovascularization specifically involving the irido-corneal angle.4,5,8 Anterior chamber flare and fibro-vascular synechiae are often associated. Intraocular pressure may be normal, elevated due to synechiae formation, or decreased due to poor ciliary body perfusion. In addition, patients may present with conjunctival and episcleral vessel dilation along with corneal edema.4
Narrowed retinal arteries, dilated retinal veins and microaneurysms are all posterior sequelae of OIS. Mid-peripheral intraretinal hemorrhages which are the result of poor “vascular push” are present approximately 80% of the time.4 Neovascularization of the optic disc (35% of eyes) or retina (8% of eyes), macular edema, and cotton wool spots can also be seen funduscopically.3,4 Clinically, a physician may observe a cherry-red spot in the macula in cases of macular ischemia and subsequent swelling of retinal ganglion cells.4
Iris neovascularization can obstruct the angle thus impairing aqueous humor outflow and leading to glaucomatous optic neuropathy. Poor perfusion to retrobulbar and anterior portions of the optic nerve may result in optic neuropathy and atrophy.4 In rare cases, orbital infarction syndrome can occur. This involves multiple intraorbital and intraocular structures leading to intraocular ischemia, ophthalmoplegia, orbital pain, hypotony and ptosis.10,11
Carotid artery imaging is mandated in order to accurately establish the diagnosis of OIS. If the carotid study appears normal, in spite of strong clinical suspicions, imaging of the retrobulbar vessels and the ophthalmic artery should be performed.2 Arteriography is the most sensitive test for carotid artery disease and needs to be employed in cases where carotid Doppler is equivocal.12
A macular stress test is a clinically useful test that can be performed during the examination. Recovery of visual function after exposure to bright light is usually delayed in cases where there is macular ischemia.2 This test is performed by directing the patient to look directly at an ophthalmoscope/transilluminator light for ten seconds. A macula exhibiting normal function will return to within one line of pretest visual acuity in less than 60 seconds.13
Fluorescein angiography will exhibit delayed choroidal filling, a delayed arteriovenous phase and large retinal vessel staining due to increased permeability.4,5 Hyperfluorescence of the optic disc can be seen due to leakage of disc capillaries secondary to increased permeability. Retinal capillary and choroidal nonperfusion may also be observed.4 Indocyanine green angiography studies can additionally assist in confirming abnormal choroid circulation manifesting as filling defects in the posterior pole and latent filling of the watershed zones.5
A combination of these anterior and posterior ischemic findings along with carotid imaging definitively leads to OIS diagnosis. Differential diagnoses include diabetic retinopathy and central retinal vein occlusion. In these disease states, intraretinal hemorrhages are more numerous and there is a presence of exudates in diabetic retinopathy. The retinal veins in a retinal vein occlusion will be tortuous.4 Unlike OIS, a central retinal vein occlusion will typically present with a swollen optic disc, hard exudates, and normal retinal arterial perfusion pressure.14 Fluorescein angiography studies can also help differentiate between OIS and these two conditions. Given the vision loss and ocular pain, giant cell arteritis must excluded with appropriate lab testing/ultrasound/biopsy. If there is enough clinical ambiguity and suspicion on the physician’s part, biopsy can be completed.4
Urgent referral to a multidisciplinary team of specialties includes retinal subspecialty, vascular surgery, internal medicine, cardiology, and neurology. Historically, in the presence of iris or posterior segment neovascularization, panretinal laser photocoagulation (PRP) has been performed to prevent neovascular glaucoma, intraocular hemorrhages, tractional retinal detachment and further ischemia leading to additional devastating vision loss. PRP is only effective in 36% of treated eyes given that choroidal ischemia can still drive the formation of additional neovascularization.4 In the early stages of neovascular glaucoma, topical steroids and cycloplegics can be used to control inflammation and help with comfort. Topical and oral aqueous suppressants can be used to lower intraocular pressure.5 Lowering intraocular pressure also helps to improve ocular perfusion and modify pain.15 Glaucoma filtering surgery, aqueous tube shunts, or ciliary body destruction may be needed post PRP if the issue is not relieved.16
Patients with active retinal and iris neovascularization have detectable vascular endothelial growth factor (VEGF), whereas patients with quiescent neovascularization from various conditions have low or undetectable concentrations. VEGF travels from the posterior to the anterior segment through both the vitreous and aqueous in response to ischemia. VEGF acts on the endothelial cells to induce new blood vessel growth.17 Although PRP has previously been the standard of care employed to decrease VEGF levels by decreasing ischemia in tissues, intravitreal injection of anti-VEGF agents is currently being used as the treatment of choice. In fact, repeated intravitreal injections of the anti-VEGF agent aflibercept have shown to be a viable option for pre-neovascular angle closure glaucoma.16 In patients with solitary iris neovascularization or early neovascular glaucoma, one injection of anti-VEGF bevacizumab can regress iris neovascularization and lower the risk of raised IOP. With this treatment, regression of iris neovascularization, in cases secondary to OIS, can occur in as little as one week post-injection.9,18 Patients presenting with advanced neovascular glaucoma usually require filtering or seton surgery in addition to the intravitreal bevacizumab to stabilize intraocular pressure.19
Systemically, a carotid endarterectomy should be considered in cases with severe occlusive artery disease, specifically those between 60%-99% stenosis, to restore carotid artery patency.4 Visual prognosis for OIS varies, as stated previously. Randomized clinical trials evaluating vision after carotid surgery have not been performed, but available data indicates approximately one-third have improved visual acuity, one-third are visually undeterred, and one-third have progressive vision loss despite surgery.20 After a year, approximately 60% of patients are hand motion or worse. About 25% of patients retain 20/50 or better vision.8 If performed in the early stages of neovascular glaucoma, retrobulbar blood flow can be restored. It is important to note that although there is a potential for restoration of vision, the patient should be educated that their prognosis is guarded as loss of vision may be irreversible.5 Surgery has been shown to decrease the two-year incidence of ipsilateral stroke by 65% in patients with 70%-99% stenosis.20 Patients with OIS have shown approximately a 40% 5-year mortality rate due to cardiovascular disease.8 Systemic arterial hypertension followed by diabetes are common comorbidities with about half of OIS patients having ischemic heart disease.8 Compliance with medications for conditions such as hypertension, atherosclerosis, diabetes mellitus, and coronary heart disease is important along with a healthy diet, cessation of smoking and physical activity.16 The importance of adherence can be promoted by the optometrist during follow-up care with these patients.
This patient was referred to a retinal specialist for further management. He was given an injection of 1.25mg Bevacizumab for the anterior segment neovascularization and will return for repeat injections as necessary. The patient was also sent for a timely carotid Doppler which revealed 80% to 99% stenosis of the left internal carotid artery. Vascular surgery specialists promptly performed a carotid endarterectomy on the left internal carotid artery within five days of his initial optometry examination.
The patient in this case was managed acutely with intraocular pressure lowering in the left eye accomplished via instillation of topical iopidine, a topical B-blocker and one drop of a topical prostaglandin. The subclinical iritis was managed with topical atropine 1% QD and topical prednisolone acetate QID. The patient was urgently referred to retinology for consideration of laser pan retinal photocoagulation (PRP) and/or vascular endothelial growth factor inhibitor (VEGFi) injection. A full vascular/medical work up was initiated through phone call and correspondence to the patient’s internal medicine physician. A complete blood count with differential and platelets (CBC c D/P), a fasting blood sugar (FBS), a lipid panel, a complete metabolic panel (CMP), a c-reactive protein test, a Westergren erythrocyte sedimentation test (ESR), an electrocardiogram (ECG) and a Doppler ultrasound of the carotid arteries (C-Duplex OU) was included.
Dr. Gurwood thanks Shaleen Ragha, OD, and Brittany Rigdon, OD, for contributing this case.
|
1. Yanoff M., Duke J. Ophthalmology: Expert Consult. Saunders: 2013; 4th ed: 442. |

