Among the 18,000 currently practicing ophthalmologists in the United States, only 50% actively perform cataract surgery.1,2 With approximately four million cataract surgeries per year in the United States, and with those numbers rising, this shortage of surgeons creates a strain on the system and calls for more involved care by comanaging optometrists.1,2
This is an opportunity to hone our cataract and refractive surgery discussion to better prepare our patients. More importantly, it is a call to become more of an extension of our surgical colleagues with pre- and postoperative care. With this being said, referring for cataract surgery isn’t always a simple task. Often, patients present with complicated and sometimes misleading clinical signs and symptoms. Every case is a chance to learn more about various ocular conditions that may require collaboration with colleagues to find the right diagnosis and treatment plan.
The purpose of this article is to elevate optometrists’ confidence in making important and sometimes difficult decisions when determining the root cause of the need for surgery and evaluating the relationship between the lens, retina and ocular surface. These three cases exemplify the collaborative process of diagnosis and care, as well as patients’ dependence on our expertise to guide their choice on whether or not to have surgery—and when.
 |
| Fig. 1. True exfoliation presents with wrinkled capsular membrane peeling (arrow) of the anterior lens capsule. |
A Closer Look: Lens
The lens is an elegant biconvex, elliptical, semi-solid avascular structure with an on-demand ability to perform accommodation based on the visual stimulus.3 The mechanical and physiological process of the lens is driven by the near pupillary reflex whereby the ciliary muscles tense to create a “bulky” or thickened lens to facilitate near work, such as reading. When viewing distance, the lens relaxes and the pupil is typically more dilated. Disruptions to the lens can cause many ocular conditions, including cataracts.4
Although cataracts are typically associated with the aging process, they may be present in neonates or may develop at any time in the life-span of an individual. Risk factors, other than increasing age, include: exposure to ultraviolet (UV) light, inadequate nutrition, smoking, high alcohol intake, diabetes, trauma and long-term use of antipsychotic medications or steroids. Cataracts will eventually lead to glare while night driving, as well as difficulty reading and focusing on distant or near objects for which corrective glasses may or may not help.4-6
When a patient presents with an unusual history and signs of cataracts, collaboration is often the key.
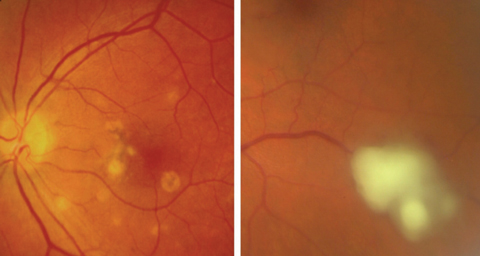 |
| Fig. 2. These images depict fungal involvement with endophthalmitis. The left image illustrates the coagulative fungal material laid down on the retina (perifoveal), while the right shows a robust vitritis (abscess). |
Turn Up the Heat
A 55-year-old Puerto Rican male came to the office with a rapid onset (over the last three months) of blurred vision greater in his left eye than his right eye while driving at night. I first asked about his work environment, and he acknowledged that his profession as a glassblower exposed him to temperatures exceeding 800˚F. His best-corrected visual acuity was 20/60 in his right eye and 20/80 in his left eye, but the potential acuity meter measured 20/25 in both eyes.
His anterior segment examination revealed 2+ to 3 milky nuclear sclerosis coupled with 2+ posterior subcapsular (PSC) changes in his right eye and 3+ milky nuclear sclerosis with 2+ to 3 PSC changes in his left eye. His intraocular pressures (IOP) were 24mm Hg in the right eye and 19mm Hg in the left eye with an unremarkable posterior segment evaluation with poor dilation. However, further investigation of the lens illustrated what appeared to be a peeled back anterior capsule and rosette features reminiscent of pseudoexfoliation and trauma in both eyes. Based on his clinical presentation, I knew it was not the latter conditions, but I educated the patient on the various differential diagnoses and the associated treatment options available.
Considering the patient had no prior history of trauma and was not of Scandinavian descent, I shared the case with my glaucoma specialist regarding the findings, which doubled as a cataract consultation. As I learned from my colleague—much to his surprise as well—the patient had glassblower’s cataract secondary to true exfoliation or lamellar delamination (Figure 1).7 With this diagnosis, the anterior lens capsule is thickened and the superficial portion of the lens capsule splits from the deeper layer and extends into the anterior chamber.8 The exact pathogenesis of this disorder is not clear but is theorized to be a result of intense infrared radiation from high heat exposure.9
Consequently, the patient was given the option to treat his cataracts by traditional phacoemulsification or femtosecond laser-assisted cataract surgery to extract and replace the lens with an intraocular lens (IOL). After much discussion, the surgeon and patient made the decision to perform the femtosecond procedure due to several factors. For one, the zonules can become brittle and break easily along with a weakened capsular structure, similar to pseudoexfoliation.10 Secondly, the phacoemulsive technique has the potential disadvantage of causing unforeseen complications (i.e., zonular rupture, posterior capsule tear) by applying more mechanical and heat stress to the intraocular space.10 While these issues might arise with femtosecond technology, the glaucoma specialist felt the risk would be reduced, allowing better recovery and post-op visual prognosis.
A Closer Look: Retina
Some retinal conditions can mimic cataract and warrant careful examination to uncover the full clinical picture. Often, collaborating with a retina specialist is necessary before preparing a patient for cataract surgery. In addition, a cataract is a possible side effect for several treatment options, such as vitrectomy. For some patients, cataracts are the least of their problems, and cataract surgery is the last step in the referral and treatment process.
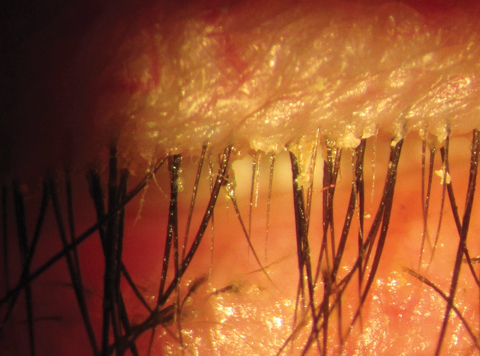  |
| Fig. 3. Multiple cylindrical dandruff and telangiectsia of lid margin and base of lashes may lead to a diagnosis of MGD/Demodex/posterior blepharitis. Click image to enlarge. |
Beware the Fungus
A 52-year-old Caucasian female called to say she lost her vision and was quite concerned. The patient stated the visual symptoms were ongoing for the past 10 days analogous to a fog in headlights with mild soreness and light sensitivity.
At examination, she explained that her diabetes management was poor, and she has received at least two Avastin (bevacizumab, Genentech) injections in each eye for macular edema approximately nine months prior to presentation. Further questioning revealed she was hospitalized two months prior for five days regarding an unknown “blood” infection that caused her to have recurrent lung symptoms beyond those caused by her cigarette smoking habit. Her visual acuity was 20/30 in the right eye without pinhole improvement and 20/100 in the left eye. She said there was a central blotch in her visual field, confirmed with Amsler grid.
Entrance testing illustrated some pain superiorly in both eyes on extraocular muscle evaluation and pupils were unremarkable. Anterior segment evaluation revealed 2+ nuclear sclerosis and arcus in both eyes. Perilimbal flush existed circumferentially in the left eye, coupled with significant infiltrative activity, multiple mutton fat keratic precipitates on the endothelium and 3+ anterior chamber cellular reaction without hypopyon. IOP was 11mm Hg in both eyes.
On dilated exam, the anterior vitreous showed a vigorous 2+ cellular reaction alongside a posterior fibrotic band hovering over the macula in the left eye. I became more concerned when I found several yellow, creamy, well-circumscribed lesions layered on top of the foveal space in the left eye, and retinal pigment perifoveal changes were noted in the right eye, likely from residual macular edema status post anti-VEGF injection (Figure 2). Also, the peripheral retinal examination revealed multiple dot-blot hemorrhages in the mid to far periphery in both eyes.
In addition to cataracts, my initial suspicion was an infectious panuveitis (endophthalmitis) with a potential fungal etiology based on the presentation. Endogenous endophthalmitis may mimic cataract, as well as conjunctivitis, non-infectious anterior uveitis, iritis, acute glaucoma, cellulitis and, retinoblastoma in children. Studies place misdiagnosis at initial presentation at 16% to 63% of cases.11,12
To ensure a timely diagnosis, I consulted with my retina specialist to discuss the case beyond just treating the surface issues with moxifloxacin 0.5% QID and cyclopentolate 1% BID in the left eye. Since we have a great working relationship, I suggested it might be necessary to perform an anterior chamber tap and order bloodwork to help determine the true nature of her condition. He agreed with my assessment and wanted to the see the patient the following day before finalizing the treatment plan.
After testing, the definitive diagnosis was fungal endogenous endophthalmitis secondary to Candida albicans. The condition is uncommon, but can be a potentially devastating intraocular infection in which pathogens reach the eye via the blood stream. Risk factors for endogenous endophthalmitis include: chronic diseases (i.e., diabetes mellitus (DM), renal failure, malignancies and acquired immunodeficiency syndrome); immunosupressive treatment; recent invasive surgery; intravenous drug abuse; indwelling catheter; endocarditis; hepatobiliary tract infections; organ transplantation; pregnancy or delivery; genitourinary surgeries; and dental procedures.13 One study found a preexisting predisposing condition in 90.9% of patients, with the most common systemic condition being DM at 50%.14
The retina specialist managed the condition empirically with voriconazole 200mg (one tablet BID) for the first two days while waiting on test results and then an intravitreal injection of voriconazole (100µg/0.1ml). She responded well initially, but eventually needed a vitrectomy.
During treatment, her cataract advanced rapidly to 3+ to 4 nuclear sclerosis and 2+ PSC in the left eye—not a surprising finding, considering research suggests vitrectomy increases oxygen tension and exposure within the eye, potentially leading to progressive nuclear sclerotic cataract formation.15,16 Consequently, the retina specialist let me know that he intended to perform monofocal cataract surgery in the left eye only. He warned against a multifocal IOL because the light-splitting optics of this design would be inadequate given the level of retinal impairment in this case (as is common from diabetes, AMD, trauma, etc.). The outcome gave her best-corrected acuity of 20/40 in the left eye with residual central chorioretinal scar tissue.
The patient stopped by after the procedure to express her gratitude for our prompt response to her emergency and congenial collaboration with the retina practice.
A Closer Look: Cornea
Many ocular surface conditions, such as keratoconus, dry eye disease (DED) and meibomian gland dysfunction (MGD), affect vision and lead to patient complaints of reduced acuity. For example, an estimated 0.3 to 2,300 per 100,000 people have keratoconus, more than one-third of patients who present in eye clinics have DED, and anywhere from 3.5% to more than 70% of patients have MGD.17-20
While dry eye is a known side effect after cataract surgery, dry eye as part of the preoperative period is less well known. As of 2012, the American Academy of Ophthalmology has considered uncontrolled external disease, including dry eye, a contraindication for surgery.21-25 The most striking and detailed landmark information came from the Prospective Health Assessment of Cataract Patients Ocular Surface (PHACO) study, which found approximately 80% of the enrolled population had a moderate to severe dry eye classification, although only 22.1% had been previously treated for the disease state.22
Addressing these ocular surface issues is always a priority, but it is particularly important for potential cataract patients. The next case exemplifies the complex nature of the ocular surface and the importance of addressing ocular surface disease (OSD) before referring for cataract surgery.
Restoring Balance
A 62-year-old Asian female presented more than a year ago stating she has been experiencing “mild” dryness and significant fluctuation in her vision. Her medical history was remarkable only for well-controlled hypothyroidism with Synthroid (levothyroxine, AbbVie). She mentioned her last eye doctor told her she had early signs of cataracts, but could not remember much else besides her glasses prescription (-2.25D in the right eye, -2.75D in the left eye). The technician performed testing, including osmolarity (with TearLab’s osmolarity system) with readings of 325mOSm/L in the right eye and 332mOSm/L in the left eye and InflammaDry (Quidel) with a mild pink line in the right eye and subtle pink line in the left eye—suggesting moderate dry eye and mild matrix metalloproteinase 9 (MMP-9) activity in both eyes.26,27
Startlingly, her visual acuity was 20/50 OD and 20/80 OS. Although in-office refraction did not yield much improvement, auto and manifest refraction found a substantial myopic shift (-4.00 -0.75x110 OD, -4.25 DS OS).
Given her complaints and my initial findings, I wasn’t shocked to find 2+ superficial keratopathy (SPK) in all quadrants and centrally combined with obvious meibomian gland toothpaste-like expression including 1+ cylindrical dandruff plus map/finger/dot regions superiorly in both eyes (Figure 3). The lens characteristics in both eyes showed 2+ to 3 nuclear sclerosis and cortical spoking extending closely into the visual axis.
A patient with both OSD and cataracts begs the age-old visual impact question, which came first? Regardless of the answer, the first step is aggressively treating the OSD prior to a cataract consultation. Although the patient was prepared for surgery, I educated her on the importance of first stabilizing the ocular surface to ensure a great outcome. Because the cataract surgery referral was inevitable, I also provided information on the surgical options to help prepare her for surgery in advance.
She was initially disappointed cataract surgery was postponed, but began treatment with Restasis (cyclosporine 0.05%, Allergan) BID, warm compresses and Cliradex wipes (Bio-Tissue) QD and scheduled a four-week follow up with refraction. Xiidra (lifitegrast 5%, Shire)—not commercially available at the time—would have been an excellent choice in this situation, considering she had both signs and symptoms of DED. Additionally, Prokera (Bio-Tissue) with or without debridement would have been an option if the anterior basement membrane yielded more significant impact on the visual axis. Of note, dehydrated amniotic membranes are FDA approved for wound coverage, whereas wet amniotic membranes are indicated for wound healing.28 Before leaving the office, she was educated that these conditions are chronic diseases that need daily maintenance even after surgery.
At her next visit, corrected vision was 20/40 in both eyes without pinhole improvement, and her refraction was -3.25 -0.75x100 OD and -3.50DS OS. Anterior segment analysis illustrated 1+ SPK in all quadrants, trace cylindrical dandruff, cloudy meibomian gland expression and consistent epithelial basement membrane regions superiorly in both eyes. Further evaluation showed some meibomian gland dropout and glandular structure shortening. TearLab and InflammaDry tests decreased to 315mOSm/L and subtle pink lines in both eyes.
With ocular surface improvement, I initiated the cataract surgery discussion, during which she expressed a desire to be free of spectacles, which would require a multifocal lens implants. I educated her on the IOL options and overall less dependence on reading glasses, but emphasized that the ultimate decision is based on the refractive surgeon’s recommendation. As comanaging ODs, we do not want to overpromise a particular refractive option, considering it may not be available based on their calculations. When making the referral to the refractive surgeon, I shared her recent DED treatment regimen and her interest in multifocal IOL options. Upon consultation, they decided she was eligible for a multifocal upgrade and went forward with the procedure. The patient was quite pleased postoperatively with her final result of 20/25 in both eyes at distance and near without glasses.
As an extra practice-building bonus moment, she referred her siblings and children to see me for their comprehensive eye examinations.
More Than Cataracts
These cases highlight the often-complex process of diagnosis and proper referral, stressing the opportunity for collaborative care with various subspecialties. Cataract patients are rarely just cataract patients, and knowing the best avenue for treatment will ensure timely care and congenial inter-professional relations. As optometry progresses, we must prepare to shoulder more of the workload with our surgical colleagues. Whether the problem lies within the lens, retina or ocular surface, we can comanage these patients to ensure exceptional care. n
Dr. Cooper practices at Solinsky EyeCare in West Hartford, Conn., concentrating on ocular disease, perioperative cataract surgery management and DED.
1. Cataract Surgeons for Improved Eyecare. Analysis of the economic impacts of dropless cataract therapy on Medicare, Medicaid, state governments, and patient costs. October 2015. https://stateofreform.com/wp-content/uploads/2015/11/CSIE_Dropless_Economic_Study.pdf. Accessed Sept. 19, 2017. |
Take Charge of Cataract ReferralsBy , and By 2050, the number of people in the United States with cataracts will double from 24.4 million to 50 million.1 Optometrists must not only identify when these patients are ready for surgery, but also provide IOL education. Patients want a surgery that has evolved to keep up with technological advances, so be ready to discuss these latest options: Laser vs. standard. Although the visual outcomes and safety are not significantly different in femtosecond laser-assisted cataract surgery vs. standard phacoemulsification, a “bladeless” option may reduce patient anxiety.2,3 Astigmatism. Due to astigmatism’s effect on distance, intermediate and near vision, a monofocal IOL alone is inadequate to meet patients’ visual needs. For patients with up to 0.75D to 1.00D of regular corneal astigmatism, laser arcuate incisions may help to minimize their dependence on distance glasses. For patients with more than 1.00D of regular corneal astigmatism, a toric IOL may be best. Specialty IOLs. Patients must have realistic expectations when considering a multifocal, extended depth of focus or accommodating IOL. While the goal is to significantly reduce dependence on glasses, a low add power during strenuous near tasks may still be necessary. Multifocal and extended depth of focus IOLs may also cause postoperative glare and halos—a possible outcome of which ODs must make patients aware.4 A careful review of corneal and retinal pathologies is necessary to determine if patients are candidates for specialty IOLs. Standard multifocal IOLs are valuable for patients who require good distance and near vision. Add power differs based on the patient’s near point location. This option may be beneficial for patients who prefer a closer near point rather than intermediate. Accommodating IOLs can help improve near vision by allowing some accommodation at near while minimizing glare. For the best possible outcome with an accommodating IOL, the non-dominant eye may be set with a small amount of residual myopia to allow the widest range of near vision.5 Compared with a standard multifocal IOL with a specific near point, extended depth of focus IOLs—the newest entry into the marketplace—can provide more intermediate and near vision and are better suited for patients who do ample computer and deskwork. Dr. Evans practices at Loden Vision Center in Nashville, TN, concentrating on perioperative surgical management, ocular disease and DED. Dr. Coats practices at McDonald Eye Associates in Rogers, AR, focusing on perioperative surgical management, ocular disease and DED. 1. National Eye Institute. Cataracts. www.nei.nih.gov/eyedata/cataract. Accessed September 3, 2017. |

