 |
A 58-year-old Hispanic male presented with blurry vision in his left eye for approximately two weeks. His medical history is significant for having chronic kidney disease which resulted in a kidney transplant one year earlier and is currently undergoing dialysis. His blood pressure is now under control. He has been maintained on oral prednisone 10mg as well as tacrolimus. He was also on ethambutol 400mg TID for two months because of a “spot” that was found on his lung.
Upon examination, his uncorrected distance visual acuities measured 20/40 in each eye but corrected to 20/20 with a mild astigmatic correction. Confrontation visual fields were full to careful counting in the right eye, but the left eye had a significant depression inferior nasal. His pupils were equally round and reactive to light; there was no afferent pupillary defect (APD). His extraocular motility was full and cover test was ortho at distance. His anterior segment was unremarkable. Tensions were 14mm Hg with Tonopen (Reichert).
On dilated fundus exam, the vitreous in both eyes was clear. The right eye looked completely normal and the left eye he had obvious fundus changes (Figure 1). An OCT was performed and is available for review.
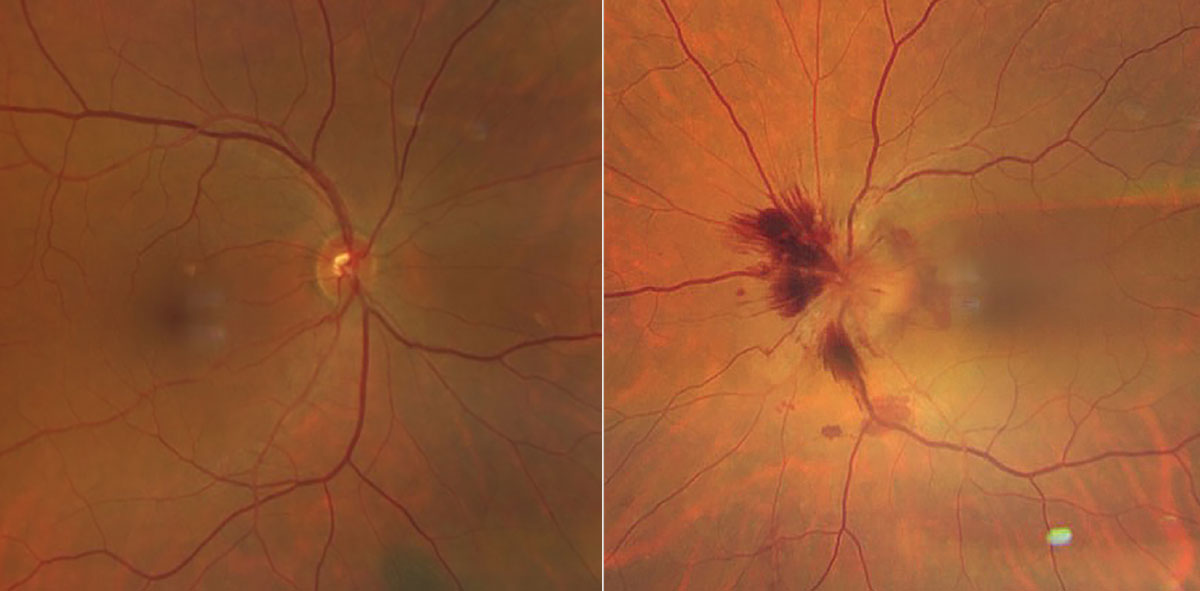 |
Fig. 1. Images of the right and left eyes. Note the appearance of the left eye with obvious fundus changes. Click image to enlarge. |
Take the Retina Quiz
1. What additional testing is necessary?
a. Visual field.
b. Sedimentation rate and C-reactive protein test.
c. CT/MRI.
d. All of the above.
2. What is the most likely diagnosis for this patient?
a. Malignant hypertension.
b. Toxic optic neuropathy.
c. Infiltrative optic neuropathy.
d. AION.
3. What is the likely cause?
a. Ethambutol.
b. Tacrolimus.
c. Focal ischemia to the optic nerve from vascular disease.
d. Large blood vessel inflammation.
4. How should this be managed?
a. Observation.
b. Stop the medications.
c. Increase prednisone dosage.
d. B or C.
For answers, see below.
Diagnosis
It was surprising to see that our patient had significant disc swelling in the left eye with surrounding flame hemorrhages and subretinal hemorrhage. What’s more, he still has excellent visual function being correctable to 20/20. A 24-2 full threshold visual field was performed and showed an enlarged blind spot in addition to an inferior nasal defect in the left eye, which is not surprising considering the amount of disc swelling that was present. The right eye was completely normal.
So, what is going on with our patient? Does this represent ethambutol toxicity? Indeed, he was on ethambutol for a “spot” on his lung which turned out to be from mycobacterium avium complex. Ethambutol is a bacteriostatic antimicrobial agent that is used as a first-line agent against tuberculosis. It acts as a chelating agent that disrupts one of several metal-containing enzyme systems in the nucleic acid structure of mycobacteria. Unfortunately, ethambutol has the potential to cause a toxic optic neuropathy, even at doses that are considered appropriate. In the optic nerve, the agent is thought to chelate copper. The reduced copper levels impair mitochondrial activity, thus reducing axon transport leading to a toxic optic neuropathy.1
Patients with ethambutol toxicity may have reduced acuity, visual field defects (cecocentral or bitemporal defects) as well as reduced color vision, even in the presence of a normal appearing optic nerves. Over time, the optic nerves develop pallor.
We decided this was not from ethambutol toxicity, but rather from an anterior ischemic optic neuropathy (AION). Ethambutol toxicity is bilateral, and patients generally don’t get disc swelling like our patient had. With AION, patients present with an acute painless loss of vision in one or both eyes. Disc swelling and/or pallor is present, which is often segmental. Patients will also have visual field loss—often an altitudinal defect—and an APD. Note that our patient did not have an APD, which may have been because he still had good visual function, or it’s possible the APD was subtle and could have been missed when doing pupillary testing.
Most patients with AION have underlying vascular disease, such as hypertension, diabetes or hypercholesterolemia, which puts them at a greater risk for stroke, heart attack and even death from their vascular disease. Our patient did have chronic hypertension from his longstanding kidney disease but he is now controlled. Sleep apnea is also a significant risk factor for developing AION. He denied a history of snoring or sleep apnea.
Some medications have been linked to the development of AION. In particular, erectile dysfunction medications have been known to increase the risk of AION, especially in those with “disc at risk” configurations. Those are optic nerves that are smaller or crowded with minimal or absent cups. Looking at the fellow eye can often provide a clue in determining if the affected eye has a “disc at risk.” The nerve in the fellow eye of our patient did not seem to fit this configuration with a cup-to-disc ratio of around 0.4.
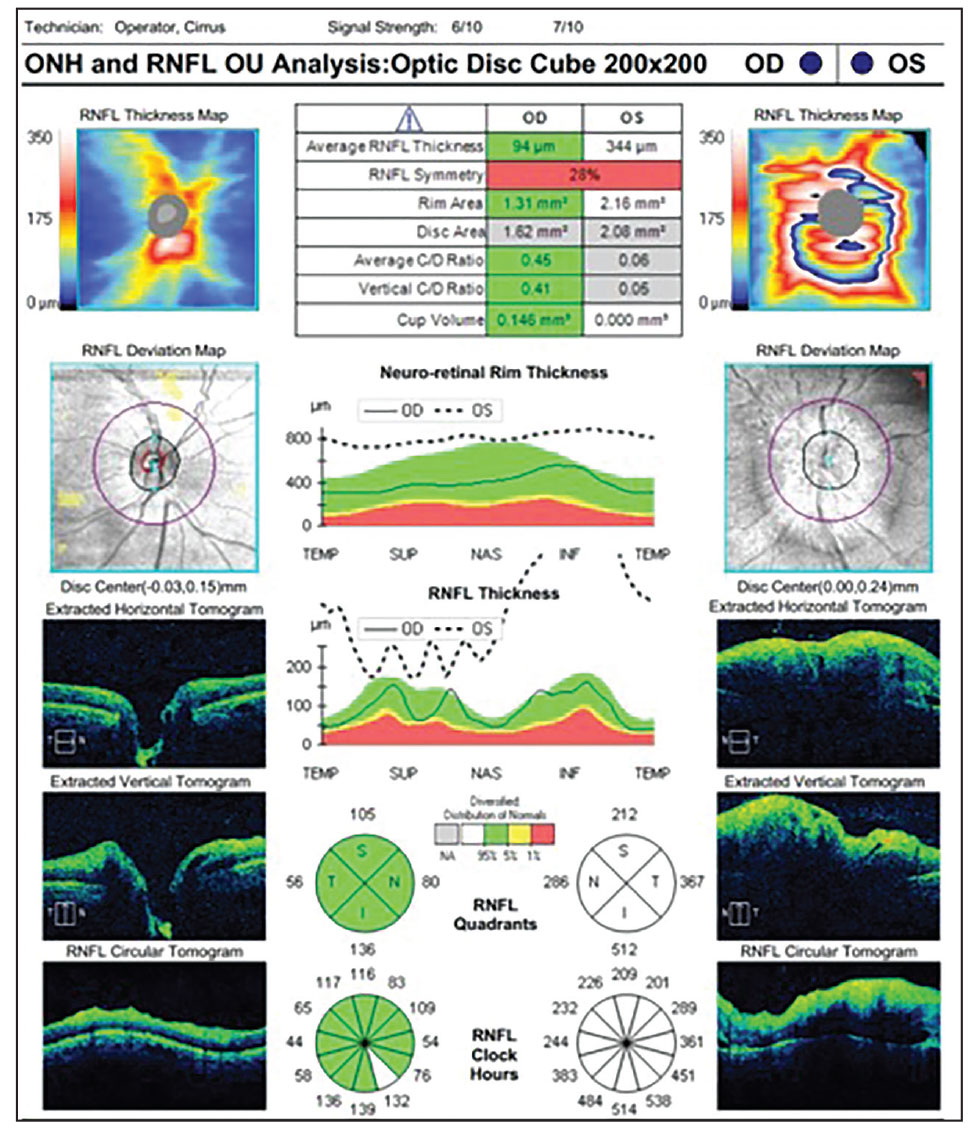 |
| Fig. 2. OCT of the right and left eyes. How do you explain the RNFL findings in the left eye? Click image to enlarge. |
Our patient was not on any erectile dysfunction medications, but he was on tacrolimus, which is primarily used as an immunosuppressant medication after organ transplants to prevent rejection. Although it is well-tolerated, AION has been reported as a rare but serious complication from this drug.2-4 It is not understood why patients develop this, but several theories have been postulated including vasoconstriction and ischemia to the optic nerve that occurs from the tacrolimus or possibly from a direct neurotoxic effect of the drug.
In our patient, it is difficult to know if the AION was caused by tacrolimus use or if it was just a chance occurrence in an at-risk vasculopathic individual. Nonetheless, an extensive workup including complete blood count, comprehensive metabolic panel, toxoplasmosis and Toxocara titers were performed, as well as a rapid plasma reagin for syphilis screening, fluorescent treponemal antibody-absorption, bartonella, anti-mitochondrial antibody, and an erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) was obtained. Everything was negative with the exception of an elevated ESR and CRP. A CT scan was also obtained and came back normal, but we were not able to use contrast because of his kidney transplant, nor were we able to do an MRI because he had a defibrillator.
The transplant team was notified of these findings and it was recommended that he stop the ethambutol. We increased his dosage of prednisone to 30mg per day with a tapering dose and he was followed closely. At his three-month follow-up, the disc swelling was significantly reduced and his vision was still 20/20, although he still had an inferior nasal visual field defect. We decreased prednisone back to 10mg and he continues to be followed
Retina Quiz Answers
1: d; 2: d; 3: b, 4; d
Dr. Dunbar is the director of optometric services and optometry residency supervisor at the Bascom Palmer Eye Institute at the University of Miami. He is a founding member of the Optometric Glaucoma Society and the Optometric Retina Society. Dr. Dunbar is a consultant for Carl Zeiss Meditec, Allergan, Regeneron and Genentech.
| 1. Melamud A, Kosmorsky GS, Lee MS. Ocular ethambutol toxicity. Mayo Clin Proc. 2003;(11):1409-11. 2. Brazis PW, Spivey JR, Bolling JP, Steers JL. A case of bilateral optic neuropathy in a patient on tacrolimus (FK506) therapy after liver transplantation. Am J Ophthalmol. 2000;129(4):536-8. 3. Kessler L, Lucescu C, Pinget M, et al. Tacrolimus-associated optic neuropathy after pancreatic islet transplantation using a sirolimus/tacrolimus immunosuppressive regimen. Transplantation 2006;81(4):636-7. 4. Yun J, Park KA, Oh SY. Bilateral ischemic optic neuropathy in a patient using tacrolimus (FK 506) after liver transplantation. Transplantation. 2010;89:1541-2. |

