 |
Q:
I have a patient who recently started treatment for refractory multiple myeloma with Blenrep (belantamab, GlaxoSmithKline). Apparently, there are significant warnings regarding corneal (epithelial) toxicity. Any suggestions on what signs to look for? At what point would the medication have to be stopped? Are the corneal changes reversible?
Blenrep is a drug used in the treatment of relapsed and refractory multiple myeloma, says Manveen Bedi, OD, who practices in Toronto. The DREAMM-1 and DREAMM-2 trials noted several ocular toxicities that required dose adjustments, dose delays and discontinuation of therapy.1,2 Once the drug reaches the corneal epithelium through the tear film or vascularized limbal region, the cytotoxic component, monomethyl auristatin F (MMAF), is internalized by corneal epithelial cells in an off-target mechanism resulting in apoptosis.1-3 Subsequently, the cornea reflects signs of keratopathy and microcyst-like corneal epithelial changes.1-3 The most common ocular impacts were dry eye and blurred vision.1-3
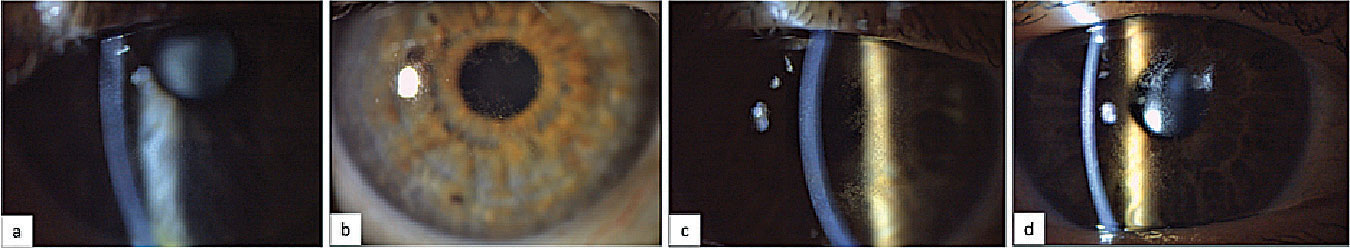 |
| Slit lamp images of patients with mild (a), moderate (b) and severe (c, d) microcystic epithelial changes.3 Click image to enlarge. |
Discussion
In the DREAMM-2 study, Dr. Bedi noted that in the presence of corneal changes the majority of patients became symptomatic of visual changes. Of those who were prescribed a dose of 2.5mg/kg, 72% experienced microcyst-like corneal epithelial changes, 25% had blurred vision and 15% suffered from dry eye.1,2 In the dosing cohort of 3.4mg/kg, 77% had microcyst-like corneal epithelial changes, 33% had blurred vision and 25% had dry eye.1,2 Symptom onset usually ranged from nine days to nine months after receiving Blenrep with a median of 36 days.2
Previously, the use of corticosteroids to mitigate MMAF-related adverse effects was considered, according to Dr. Bedi. However, the DREAMM-2 sub-study further investigated the role of steroids and increased the treatment duration from four to seven days, concluding that there was no significant effect on corneal change prevention.2
Also, long-term follow-up of patients from the DREAMM-1 study showed development of secondary glaucoma and cataracts; therefore, it was recommended to avoid steroids as a mitigation strategy to reduce ocular toxicity.2
Corneal changes are overall reversible with dose modification and cessation of therapy; however, in the presence of corneal adverse effects, recommendations from the DREAMM-2 study should be followed while working in conjunction with a hematologist/oncologist.1,2
Suggestions include the following:1
- Conduct a baseline eye examination with visual acuity and slit lamp assessment up to three weeks prior to initiation of Blenrep and monitor patient prior to every cycle of treatment (up to two weeks before) and upon worsening of symptoms.
- Comanage care with a hematologist/oncologist and provide information about corneal findings and visual acuity changes for dose management, which is based on the worst grade of the most affected eye.
- Use the Keratopathy and Visual Acuity Scale to determine dosing.1,2
- For patients with a history of dry eye, avoid contact lens wear and initiate use of preservative-free artificial tears four times a day with the first infusion of Blenrep until cessation of therapy, as this population is more likely to develop moderate/severe microcyst-like corneal epithelial changes.
1. Farooq AV, Degli Esposti S, Popat R, et al. Corneal epithelial findings in patients with multiple myeloma treated with antibody-drug conjugate belantamab mafodotin in the pivotal, randomized, DREAMM-2 study. Ophthalmol Ther. 2020;9(4):889-911. 2. Wahab A, Rafae A, Mushtaq K, et al. Ocular toxicity of belantamab mafodotin, an oncological perspective of management in relapsed and refractory multiple myeloma. Front Oncol. 2021;11:678634. 3. Aschauer J, Donner R, Lammer J, et al. Corneal toxicity associated with belantamab mafodotin is not restricted to the epithelium: neuropathy studied with confocal microscopy. Am J Ophthalmol. 2022;242:116-24. |

