Anti-vascular endothelial growth factor (anti-VEGF) agents have been used in ophthalmic care for nearly two decades since the first approval in 2004. Their development revolutionized the management of posterior segment diseases such as neovascular age-related macular degeneration (nAMD), diabetic retinopathy (DR), diabetic macular edema (DME), macular edema secondary to retinal vein occlusion (RVO) and choroidal neovascularization (CNV) from any source, among others (as depicted in Figures 1 to 3).
However, after a long period of dominance by the familiar triumvirate of Avastin, Lucentis and Eylea, the medication landscape in 2023 is far more varied. Optometrists would do well to keep up with recent anti-VEGF advances in order to have more accurate and fruitful discussions with patients prior to referral and during monitoring visits for those engaged in comanagement.
A Brief History
The first selective anti-VEGF molecule used for ophthalmic purposes was pegaptanib (Macugen), though its reign was short-lived; treated patients maintained but did not regain visual acuity. While that agent was on the market, bevacizumab (Avastin), originally designed to treat colon cancer, was soon found to help control neovascularization and vascular permeability in the aforementioned conditions when injected intravitreally into the eye. Numerous other medications have since been developed for ocular use with the goal to improve efficacy and durability of treatment.1
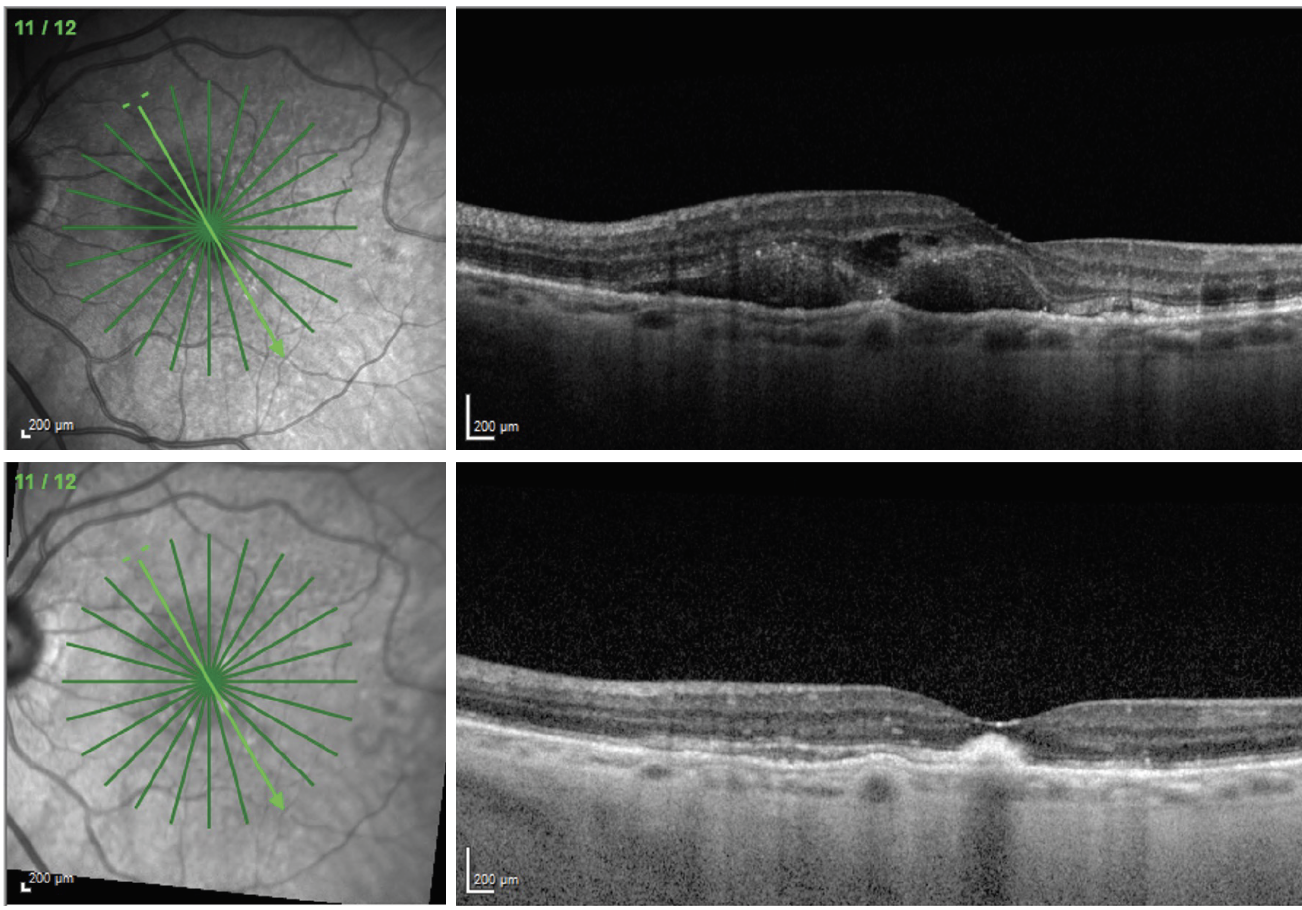 |
| Fig. 1. An 83-year-old female presented with nAMD and 20/400 visual acuity (top). She received an injection of Eylea and returned four weeks later with significant improvement in macular thickness (bottom). Her visual acuity was still 20/200 at this visit. The patient will continue to receive injections, and the visual outcome will depend on how much submacular scarring and photoreceptor atrophy is present once the macula stabilizes. Click image to enlarge. |
Drawbacks
Intravitreal anti-VEGF agents provide highly effective and low-risk treatment for conditions that were previously very difficult or impossible to manage, but they are not without downsides. One difficulty that remains is known as treatment burden. Though advances in molecular design have led to longer-acting agents, these medications often require repeat injections over long periods of time. The original regimens required monthly or bimonthly visits.
For patients, this can mean lost productivity with more days off work, time away from family and friends, financial burdens, long travel distances, many hours spent each year in the office of a retina specialist and even an incrementally higher risk of endophthalmitis with each additional injection. In some cases, this burden is placed not only on the patient but also on those they rely on for support and transportation.
Another downside: While anti-VEGF agents target a key factor in the pathophysiology of a wide variety of vascular and angiogenic posterior segment conditions, some patients still have less than optimal response to treatment. For example, patients battling DME may suffer from persistent edema even after numerous treatments with the best available agents.2 In addition, anti-VEGF injections do not treat every aspect of these conditions, such as macular ischemia in DR or geographic atrophy (GA) in AMD, driving the need for different therapeutic options to be used in addition to or in place of anti-VEGF.
No matter how effective the agent, early detection and intervention is also key to achieving positive outcomes. Patients with nAMD, for example, can quickly develop submacular scarring and photoreceptor atrophy if intervention is not initiated promptly. Another consideration of anti-VEGF therapy is the potential of adverse events associated with any intravitreally delivered medication, which even includes severe complications like endophthalmitis.
While these pitfalls exist, use of anti-VEGF in the eye has saved the vision of countless patients, and innovation continues to improve the therapy over time. In the remainder of this article, we provide a review of the current tried-and-true anti-VEGF agents along with the newcomers in this space.
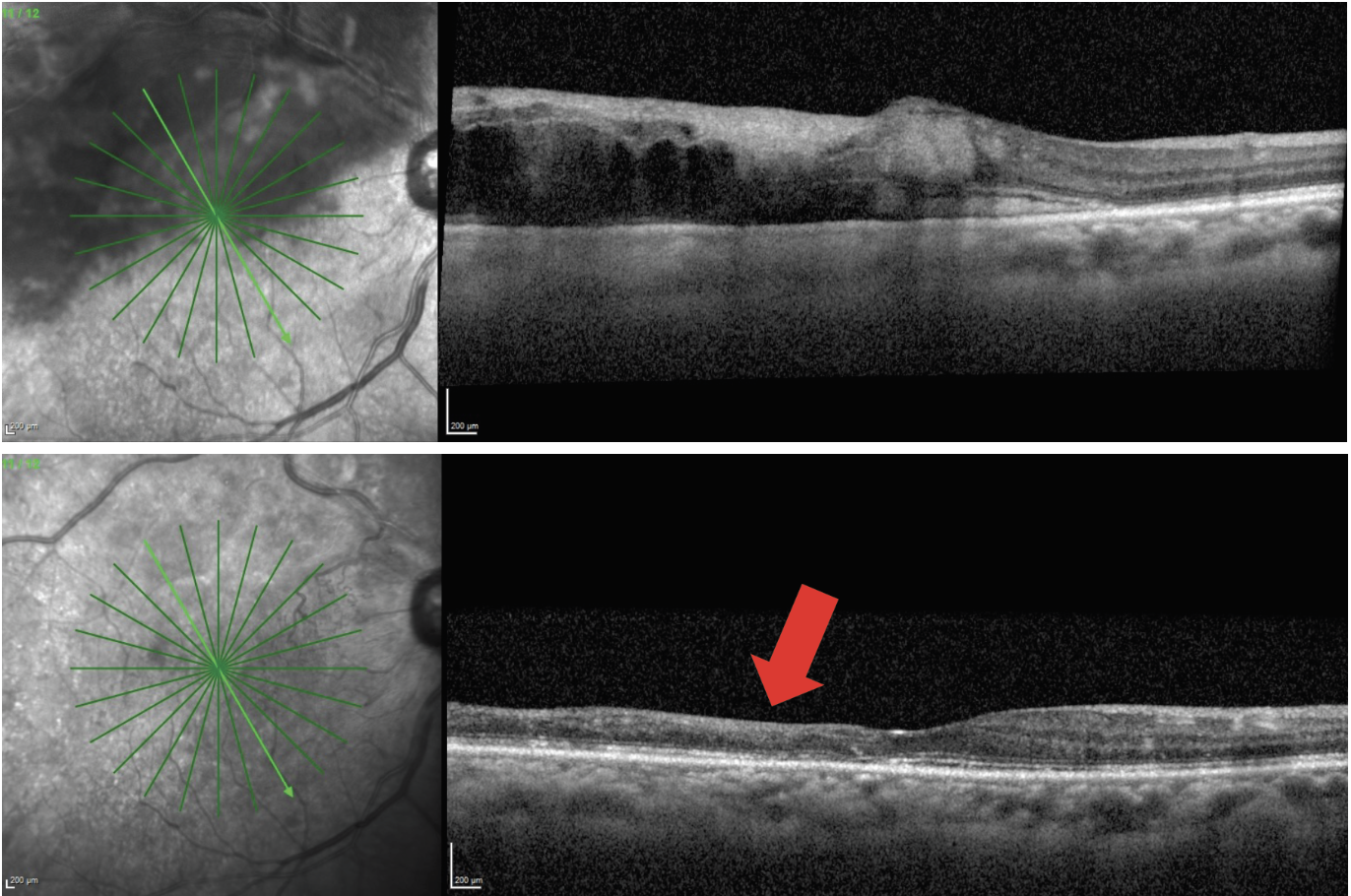 |
Fig. 2. A 68-year-old male patient presented with macular edema secondary to branch RVO and visual acuity of 20/80 (top). The patient had four monthly Avastin injections, which resulted in complete resolution of the macular edema (bottom). The patient’s visual acuity improved to 20/50 but was limited by the severity of macular ischemia that is evident from the significant inner retinal thinning (red arrow). Click image to enlarge. |
Anti-VEGF Classics
Though the landscape of anti-VEGF therapy is always evolving, the following three medications have historically been used most in the United States:
- Bevacizumab (Avastin), although off-label in eye care, is still commonly used intravitreally for the conditions described above. Its greatest advantage is that it is cost-effective.
- Ranibizumab (Lucentis, Genentech) is FDA-approved for nAMD, all forms of DR, macular edema secondary to RVO and myopic CNV.3
- Aflibercept (Eylea, Regeneron) is FDA-approved for nAMD, all forms of DR and macular edema secondary to RVO.4
New-generation Compounds
Development of novel drugs continues to provide greater efficacy and durability with goals of improving visual outcomes and decreasing burden of treatment. Two examples of more recently approved medications follow:
Brolucizumab-dbll (Beovu, Novartis). This agent was FDA approved in 2019 for the treatment of nAMD and later for DME. Since its approval, concerns surrounding its increased risk of intraocular inflammation have stunted its integration into clinical practice. Post-approval analysis suggests about 4% risk of intraocular inflammation with some being severe occlusive vasculitic events.5 A chief advantage of Beovu is durability, with about 50% of patients in both the nAMD and DME trials being able to maintain a q12 week dosing schedule.6,7
Faricimab-svoa (Vabysmo, Genentech). Vabysmo received FDA approval in 2022 for the treatment of nAMD and DME. The drug targets not only VEGF but also angiopoietin-2, another pro-angiogenic molecule, making it the first bispecific intravitreal anti-VEGF molecule. A major advantage of Vabysmo is its durability, with about 50% of patients in clinical trials able to maintain q16 week dosing for nAMD and DME.8,9
High-dose Aflibercept
Eylea was previously only approved at a dose of 2mg per injection; however, last month the FDA gave its stamp of approval to a higher aflibercept dose of 8mg for the treatment of wet AMD, DME and DR, allowing for extended intervals between treatments of up to 16 weeks. The recommended regimen for Eylea HD—the brand name of the high-dose form—is monthly injections for the first three months followed by treatments every eight to 16 weeks in wet AMD and DME and every eight to 12 weeks for DR.
The drug’s approval was sparked by the positive results of two clinical trials, Pulsar and Photon, which demonstrated non-inferiority and clinically equivalent vision gains at 48 weeks with eight-, 12- and 16-week dosing regimens after the three initial monthly doses. The Pulsar study, which evaluated the efficacy of Eylea HD on patients with nAMD, showed that the high-dose formulation provided longer dosing intervals and improved drying of fluid.10 The Photon study for DME also showed the benefit of longer treatment intervals.11
Adverse reactions, while only occurring in 3% or fewer of patients, included cataract, conjunctival hemorrhage, increased intraocular pressure, ocular discomfort, eye pain or irritation, blurry vision, floaters, vitreous detachment, corneal epithelium defect and retinal hemorrhage.
Step Therapy
With all these new treatment options, which are doctors most likely to choose? The current reality is that treatment decisions depend not only on doctor preferences, but logistical considerations and insurance coverage. Most insurances require some variety of “step therapy” where more affordable options like Avastin must be tried first, and then changed in a stepwise approach if response is suboptimal.
There is obvious concern that delaying treatment with more efficacious drugs could result in worse visual outcomes. DRCR Network Protocol AC evaluated the response of DME with initial Eylea treatment vs. initial Avastin followed by Eylea if response was suboptimal with Avastin. It found that the two arms had similar visual outcomes.12 While there are many struggles on an individual patient basis navigating the requirements of step therapy, these results were at least reassuring that, on average, patients will not be harmed by step therapy.
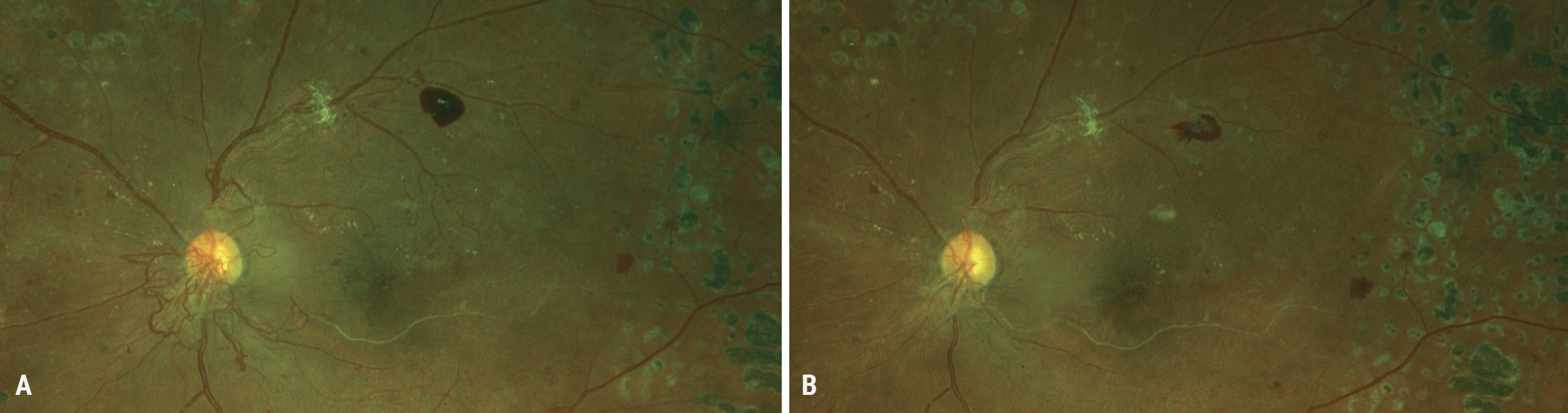 |
Fig. 3. (A) A 40-year-old Black female presented after noncompliance with significant neovascularization at the disc (NVD). She has previously been treated with panretinal photocoagulation. She received an Avastin injection and returned one month later with resolution of the NVD (B). The patient must be compliant with follow-ups, as the neovascularization can return and require the need for more injections. Click image to enlarge. |
Biosimilars
In addition to the creation of novel medications, a class of drugs called biosimilars is being created to reduce costs and increase access to care. Anti-VEGF molecules are biological compounds created through complex and expensive manufacturing processes using living material. When their patents expire, biosimilars of the reference drug can be produced. Biosimilars have been described as the “generic versions” of biological compounds, but it is important to consider the difference between a traditional drug and a biological compound. Traditional drugs are smaller molecules whose known composition can be replicated exactly through chemical synthesis. Biological compounds, or biologics, are large-complex molecules. They cannot be precisely replicated due to their large size and complicated manufacturing processes. A biosimilar is a compound that is not identical to its reference drug but has been shown to be similar in structure, function, safety and efficacy.13
There are currently two FDA-approved biosimilars of Lucentis available for use in the United States: Byooviz (ranibizumab-nuna, Biogen/Samsung Bioepis) and Cimerli (ranibizumab-eqrn, Coherus BioSciences). Byooviz was the first to receive FDA approval in 2021 for the treatment of nAMD, macular edema following RVO and myopic CNV.14 The following year, in 2022, Cimerli became approved as an interchangeable biosimilar for the treatment of all prior indications of Lucentis (nAMD, DR, DME, myopic CNV and macular edema following RVO).15
Not all biosimilars are considered interchangeable. While interchangeable biosimilars must meet additional requirements by the FDA, this designation does not necessarily mean that they are safer or more effective.18
There is particular concern that the FDA approval of Avastin biosimilars could ultimately increase the cost of care. With such a biosimilar FDA-approved for ophthalmic use, pharmacies may no longer be able to compound off-label bevacizumab, thus removing the lowest-cost option from the market. At this year’s American Society of Retina Specialists meeting, Ravi Parikh, MD, of the NYU Grossman School of Medicine and Manhattan Retina and Eye Consultants in New York City, noted that “switching from Lucentis and Eylea to biosimilars would offset only 30% of the increase resulting from the higher cost of the bevacizumab biosimilar.”17
Nonetheless, in general, production of anti-VEGF biosimilars has the potential to decrease the cost of care and increase accessibility of more effective treatment options. However, it is unclear how well these medications will be accepted by physicians. Use of biosimilars throughout all fields of medicine in the US is fairly new, with the first FDA biosimilar being approved only in 2015.18 This is particularly novel in the retina space with the first FDA approval of an anti-VEGF biosimilar occurring in 2021.
Concerns stem from the complicated manufacturing process of these compounds, where seemingly benign changes could lead to increased adverse reactions, with particular concern for immunological responses.13 Expanded use on a broader scale will help to uncover if these concerns have merit or not. At present, it is encouraging to have these additional and more affordable options with numerous other biosimilars for Lucentis and Eylea in clinical trials.
Novel Delivery Methods
Given the burden and barriers in the care of chronic conditions such as nAMD and DME—caused by factors such as injection frequency, anxiety and fatigue—exploration of alternative dosing strategies to overcome these concerns has surged. One of these innovations includes permanent implantable devices such as the port delivery system (PDS; Susvimo implant, Genentech).19,20 This device received FDA approval for nAMD in 2021 and was in clinical trials for DME and DR.
Following surgical implantation of the device, patients had the port refilled every six months, which was more tolerable than frequent intravitreal injections. However, in October 2022, Genentech voluntarily recalled Susvimo due to a mechanical failure of the device during the refill procedure, and the company paused all new implantations, including those in ongoing global clinical trials.21 Currently, there are no updates on the device’s status. As for patients who received the PDS during clinical trials or post-FDA approval, as long as the device is intact, they can continue receiving the appropriate interval refills.
Other delivery alternatives under investigation include bioerodible implants, such as depot formulations of sunitinib malate GB-102 (Graybug Vision) and genetic therapies that introduce genetic material through subretinal or suprachoroidal injection of adeno-associated virus vectors.22 These genes alter the ocular tissue to produce endogenous anti-VEGF with the hope to seize or reduce the need for intravitreal injections. Several are in various stages of clinical trials for AMD, DR and even GA. Examples include RGX-314 (RegenxBio), 4D-150 (4D Molecular Therapeutics), EXG102-031 (Exegenesis Bio), GEM103 (Disc Medicine) and SK0106 (Skyline Therapeutics), the last which recently received its investigational new drug application for nAMD.23
Alternatives to Injections
In addition to all these possibilities of longer-acting agents, combination strategies, sustained-release and genetic therapies, there is potential for topical and oral agents in the treatment of these conditions. OTT166 (OcuTerra Therapeutics), an integrin inhibitor, is a proposed engineered molecule that has shown safety and efficiency via topical application for treatment of DR in early-phase trials. Currently, the DREAM (Diabetic Retinopathy Early Active Management) study is in Phase II trials for further investigation.24
APX3330 (Ocuphire Pharma) is an oral agent that—despite failing to meet its primary endpoint of reversal of moderately severe to severe DR (ETDRS severity levels 47 and 53) in its Phase II trial, ZETA-1—did demonstrate good systemic and ocular safety. In addition, it was announced recently that ZETA-1 showed a statistically significant reduction in the progression of DR to more advanced stages in those taking APX3330.25
Xiflam (InflammX) is also an oral agent that inhibits inflammasomes. It is currently under investigation for treatment of DR and DME.
Future Possibilities
These are definitely exciting times for research and development. Currently, there are nearly 90 investigational novel agents in the pipeline for various retinal diseases, primarily targeting wet and dry AMD, DR, DME and inherited retinal disorders. A comprehensive review of these can be found in the January/February 2023 issue of Retina Specialist (or click here).
Non-Anti-VEGF Interventions
As mentioned previously, while anti-VEGF medications have revolutionized care of the posterior segment, they do not treat every aspect of posterior segment disease. In the treatment of AMD, for example, anti-VEGF medications are used to manage the complication of CNV but they do not treat the degenerative process of AMD itself. Novel targets are needed to treat other aspects of disease such as complement inhibition for GA.
In February 2023, a new class of drug was released for the treatment of GA secondary to AMD. Pegcetacoplan (Syfovre, Appellis) inhibits complement factor C, stopping a vital step in the complement cascade, which promotes inflammation. Syfovre is an intravitreal injection with recommended dosing between 25 and 60 days. While Syfovre does not reverse GA or stop it entirely, it reduces GA growth between 17% and 22% at two years depending on the dosage.26 It is meant to be a long-term ongoing treatment and not a cure.
In clinical trials, Syfovre did increase the risk of CNV, with 12% occurrence in those treated monthly, 7% in those treated bimonthly and 3% in the sham group. Those who develop CNV will then require anti-VEGF injections.27
In mid-July, the American Society of Retina Specialists announced reports of retinal vasculitis in a handful of patients receiving injections of Syfovre, though this side effect was not reported in clinical trials.28 The ASRS statement noted that all occurrences took place between seven and 13 days after drug administration, and the etiology of the events remains unclear. At the time of that announcement, the outcomes of affected patients were still evolving.
While the situation continues to unfold, eyecare practitioners must stay vigilant for any updates that emerge and decide individually whether they plan to adjust their intervention protocols based on the new information.
Last month, a second drug for GA became FDA-approved: an intravitreally dosed complement factor V inhibitor, avacincaptad pegol (Izervay), from Iveric Bio. In its Phase III trials, slowing of disease progression was observed after as few as six injections; after 12 injections, patients experienced up to a 35% reduction in GA progression rate compared with sham. The most common adverse reactions reported by patients during trials included conjunctival hemorrhage (13%), increased intraocular pressure (9%) and blurry vision (8%).29
Other intravitreal injectable treatment options include intravitreal steroids. While this approach can have the well-known side effects of cataract formation and increased intraocular pressure, it continues to have a role in treating posterior segment disease. Kenalog can be used off-label intravitreally or via sub-Tenon delivery. In addition, sustained-release intravitreal steroid injections include Ozurdex, Yutiq and Iluvien. While each has a unique list of approvals, this drug category can be helpful in treating conditions such as DME, macular edema from RVO, cystoid macular edema and non-infectious posterior uveitis. Though anti-VEGF is considered the first-line treatment for DME, DRCR Network Protocol U showed that persistent DME despite multiple Lucentis injections may have improvement of macular thickness when supplemented with Ozurdex.30
Takeaways
Intravitreal anti-VEGF injections have revolutionized posterior segment care, providing vision-saving treatments for a variety of conditions. Longer-acting and more efficacious medications as well as biosimilars continue to be developed to improve cost and patient access. Sustained delivery systems and alternate delivery methods are also a likely possibility in the near future. Drugs with new mechanisms of action will continue to advance as well, providing new delivery methods and treating aspects of disease that do not respond to anti-VEGF.
The trade-off between the simplicity of yesterday and the complexity of today is our much more robust capability to fight posterior segment disease at present. Both the complexity and the capability will no doubt continue to increase over time.
1. Kim LA, D’Amore PA. A brief history of anti-VEGF for the treatment of ocular angiogenesis. Am J Pathol. 2012;181(2):376-9. 2. Kuroiwa DAK, Malerbi FK, Regatieri CVS. New insights in resistant diabetic macular edema. Ophthalmologica. 2021;244(6):485-94. 3. Lucentis. [package insert]. San Francisco, CA: Genetech, Inc. 2014. 4. Eylea. [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc. 2023. 5. Monés J, Srivastava SK, Jaffe GJ, et al. Risk of inflammation, retinal vasculitis, and retinal occlusion–related events with brolucizumab: post hoc review of HAWK and HARRIER. Ophthalmology. 2021;128:1050-9. 6. Dugel PU, Koh A, Ogura Y, et al. HAWK and HARRIER: Phase 3, multicenter, randomized, double-masked trials of brolucizumab for neovascular age-related macular degeneration. Ophthalmology. 2020;127(1):72-84. 7. Brown DM, Emanuelli A, Bandello F, et al. KESTREL & KITE: 52-week results from two phase III pivotal trials of brolucizumab for diabetic macular edema. Am J Ophthalmol. 2022;238:157-72. 8. Heier JS, Khanani AM, Quezada Ruiz C, et al. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet (London, England). 2022;399(10326):729-40. 9. Wykoff CC, Abreu F, Adamis AP, et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): two randomised, double-masked, phase 3 trials. Lancet (London, England). 2022;399(10326):741-55. 10. Lanzetta P. Intravitreal aflibercept injection 8 mg for nAMD: 48-week results from the phase 3 PULSAR trial. Presented at American Academy of Ophthalmology 2022 annual meeting, September 30 to October 3, 2022. investor.regeneron.com/static-files/e3307e7d-d495-438c-b8bb-c62cdacdb37. Accessed July 9, 2023. 11. Brown DM. Intravitreal aflibercept injection 8 mg for DME: 48-week results from the phase 2/3 PHOTON trial. Presented at American Academy of Ophthalmology 2022 annual meeting, September 30 to October 3, 2022. investor.regeneron.com/static-files/da20405e-b843-402e-855b-d824a15dec60. Accessed July 9, 2023. 12. Jhaveri CD, Glassman AR, Ferris FL, et al. Aflibercept monotherapy or bevacizumab first for diabetic macular edema. N Engl J Med. 2022;387(8):692-703. 13. Biosimilars. US FDA. Updated March 1, 2023. www.fda.gov/drugs/therapeutic-biologics-applications-bla/biosimilars. Accessed July 9, 2023. 14. FDA Approves First Biosimilar to Treat Macular Degeneration Disease and Other Eye Conditions. US FDA. Published September 17, 2021. www.fda.gov/news-events/press-announcements/fda-approves-first-biosimilar-treat-macular-degeneration-disease-and-other-eye-conditions. Accessed July 1, 2023. 15. CIMERLI™ (ranibizumab-eqrn) U.S. Prescribing Information, August 2022. www.accessdata.fda.gov/drugsatfda_docs/label/2022/761165s000lbl.pdf. 16. Interchangeable Biological Products. US FDA. Updated March 1, 2023. www.fda.gov/biosimilars. Accessed July 9, 2023. 17. Bankhead C. Sticker shock: ophthalmic bevacizumab biosimilar could drive up costs. MedPage Today. Published August 1, 2023. www.medpagetoday.com/meetingcoverage/asrs/105715#:~:text=The%20analysis%20showed%20that%20an,would%20increase%20by%20about%2015%25. Accessed August 17, 2023. 18. Biosimilars approved in the US. Generics And Biosimilars Initiative. Updated May 19, 2023. www.gabionline.net/biosimilars/general/biosimilars-approved-in-the-us. Accessed June 30, 2023. 19. Ranade S V, Wieland MR, Tam T, Rea JC, et al. The Port Delivery System with ranibizumab: a new paradigm for long-acting retinal drug delivery. Drug Deliv. 2022;29(1):1326-34. 20. Holekamp NM, Campochiaro PA, Chang MA, et al. Archway randomized phase 3 trial of the port delivery system with ranibizumab for neovascular age-related macular degeneration. Ophthalmology. 2022;129(3):295-307. 21. Hutton D, Charters L. Genentech recalling ranibizumab injection ocular implant in US. Ophthalmology Times. Published October 17, 2022. www.ophthalmologytimes.com/view/genentech-recalling-ranibizumab-injection-ocular-implant-in-us. Accessed July 9, 2023. 22. Patel P, Sheth V. New and innovative treatments for neovascular age-related macular degeneration (nAMD). J Clin Med. 2021;10(11). 23. Crago SM. FDA Gives Clearance of IND for SK0106, Skyline Therapeutics AAV gene therapy candidate for nAMD. Modern Retina. Published July 6, 2023. www.modernretina.com/view/fda-gives-clearance-of-ind-for-sk0106-skyline-therapeutics-aav-gene-therapy-candidate-for-namd. Accessed July 9, 2023. 24. OTT166 in Diabetic Retinopathy (DR). Clinicaltrials.gov. Updated July 17, 2023. classic.clinicaltrials.gov/ct2/show/NCT05409235. Accessed July 9, 2023. 25. Hutton D. Efficacy and safety results of the ZETA-1 Phase 2 trial in diabetic retinopathy at the 83rd Scientific Sessions of the ADA. Modern Retina. Published July 3, 2023. www.modernretina.com/view/efficacy-and-safety-results-of-the-zeta-1-phase-2-trial-in-diabetic-retinopathy-at-the-83rd-scientific-sessions-of-the-ada. Accessed July 9, 2023. 26. Syfovre Efficacy. Syfovre. syfovreecp.com/oaks-and-derby-efficacy. Accessed July 9, 2023. 27. Syfovre Safety. Syfovre. syfovreecp.com/safety. Accessed July 9, 2023. 28. Apellis shares sink on reports of rare side effects with new vision loss drug. Fidler B. Biopharma Dive. Published July 17, 2023. www.biopharmadive.com/news/apellis-syfovre-inflammation-side-effects-geographic-atrophy/687989. Accessed August 22, 2023. 29. Innovations for Tomorrow’s Retinal Disease Patients: Therapeutic Pipeline. Iveric Bio. 2023. ivericbio.com/our-science/#pipeline. Accessed July 9, 2023. 30. Maturi RK, Glassman AR, Liu D, et al. Effect of adding dexamethasone to continued ranibizumab treatment in patients with persistent diabetic macular edema: a DRCR network phase 2 randomized clinical trial. JAMA Ophthalmol. 2018;136(1):29-38. |

