 |
A 67-year-old male presented for a glaucoma evaluation due to elevated intraocular pressure (IOP). His history was significant for right eye cataract surgery and astigmatic LASIK correction approximately eight years earlier and laser capsulotomy in his right eye seven years earlier. He reported no complications with any of the surgical procedures. He was diagnosed with primary open-angle glaucoma and, after a discussion of risks and benefits of treatment, was prescribed travoprost 0.004% QHS bilaterally.
Examination
The patient returned four months later and reported that travoprost was well tolerated and that he was adherent. He also reported blurred vision, in his right eye more than the left, beginning soon after the initiation of travoprost. At this time, his corrected visual acuity was 20/50-2 OD. No biomicroscopic changes were noted and no evidence of anterior chamber or vitreal inflammation were present. IOP was reduced to 15mm Hg OU.
Ophthalmoscopically, mild macular thickening appreciated in his right eye with no other apparent changes. Subsequent optical coherence tomography (OCT) analysis revealed significant cystoid macular edema (CME) in the right eye.
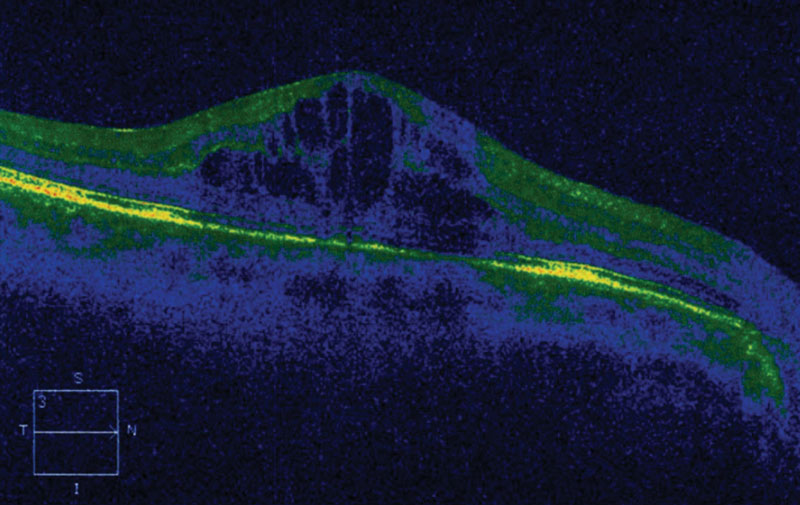 |
| This OCT shows medication-induced CME in a glaucoma patient. |
Diagnosis
Based upon the history, timing and findings, he was diagnosed with CME in the right eye, presumably from travoprost use. Travoprost was discontinued and replaced with Cosopt (dorzolamide 2%/timolol 0.5% fixed combination, Akorn) BID OU. To address the CME, he was prescribed Pred Forte, (prednisolone acetate 0.1% Allergan) QID and Bromday (bromfenac 0.09%, Bausch + Lomb) BID OD. Over the next several months, the patient’s IOP remained stable at 16mm Hg OU with resolution of CME and subsequent improvement in visual acuity. The Bromday and Pred Forte were discontinued.
CME is not a true diagnosis but rather a finding that occurs from numerous causes. These causative factors can include ocular eye drop preservatives, topical prostaglandin analogs and topical beta-blockers.6-8 After cataract surgery, however, the second most common cause of CME is diabetes.4
Monitoring
The predominant symptom caused by CME is visual distortion (metamorphopsia) and reduced acuity. Visual acuity may be minimally or significantly reduced, depending upon the underlying cause of CME. Most patients who have CME detected only by imaging have no visual disturbances.5
The ophthalmoscopic appearance of perifoveal retinal thickening is difficult to observe. The true petaloid appearance of CME is best appreciated with fluorescein angiography (FA).4-6 OCT testing is preferred when possible, as it permits noninvasive observation of the cystic, fluid-filled spaces.7 Intracellular fluid and Müller cell swelling produce the condition’s distinctive hexagonal appearance.8
Leaking perifoveal capillaries, subject to the pathophysiology of the underlying cause, go on to create the formation of intraretinal polycystic fluid-filled spaces that disrupt light from reaching the photoreceptors and slow efficient dialogue to the visual pathway.1,8,9 Exudative or transudative fluid collects in the loosely arranged outer plexiform layer-of-Henle.
The fibers in Henle’s layer are horizontally arranged, allowing maximum light transmission. This is what creates the fovea’s parabolic shape with the thinnest region being the foveola. This anatomy, along with the sequential filling of cysts is what fosters the petaloid appearance seen during FA.5,6
Various factors and mechanisms are involved in the pathogenesis of CME, including the release of inflammatory mediators such as prostaglandins.4-6 Light toxicity from the operating microscope and mechanical irritation of the internal ocular tissues are also provocative.4-6 Inflammatory mediators disrupt the blood-aqueous barrier (and blood-retinal barrier), leading to increased vascular permeability.4-6 Any disease process that can breakdown these barriers can induce CME.10,11
Treatment Options
Topical non-steroidal medications such as Acular (ketorolac tromethamine, Allergan), Nevanac (nepafenac 0.1%, Novartis) and Bromday all show success.12,13 Topical corticosteroid drops such as Pred Forte, Lotemax (loteprednol etabonate, Bausch + Lomb) and Durezol (difluprednate, Novartis) can be added for more severe cases.12,13 Common dosing ranges from QID to Q2H. Often, a loading dose of Q2H is initiated and then rapidly dropped to QID after several days. Duration may be several days to months, depending on the CME’s severity. Oral carbonic anhydrase inhibitors (CAIs) such as acetazolamide and methazolamide can be helpful in recalcitrant cases.14
Significant research supports adjunctive use of topical dorzolamide to manage CME associated with retinitis pigmentosa (RP). Long-term treatment of RP-related CME can improve structure and function with reduced edema and improved acuity.15-17 CAIs may even reduce outer nuclear layer fluid more effectively than inner nuclear layer fluid due to better access to retinal pigment epithelium basolateral membrane than neurosensory retina.18 Cosopt also reduces retinal thickness and improves function in CME arising from numerous other causes.29-33
Additionally, Cosopt may also have benefits as adjunctive topical therapy for macular edema. Researchers report patients dosed with topical dorzolamide-timolol twice daily and maintained on the same anti-vascular endothelial growth factor drug and same interval between injections saw a significant decrease in macular thickness. They concluded that topical dorzolamide-timolol may have a beneficial anatomical and functional effect in eyes with macular edema secondary to retinal vein occlusion resistant to treatment.34
Another study evaluated the effect of topical dorzolamide hydrochloride-timolol maleate on anatomic and functional outcomes in eyes with neovascular age-related macular degeneration (AMD) that had an incomplete response to anti-VEGF therapy. They found that adjunctive topical therapy with Cosopt may reduce central subfield thickness and subretinal fluid in eyes with persistent exudation despite consistent, fixed-interval intravitreous anti-VEGF treatment for neovascular AMD.35
While topical therapy for macular edema is inconsistently used, possibly due to the perception that this route of administration has limited success, it appears that there is benefit. Topical dorzolamide and Cosopt especially may have a surprising benefit in managing macular edema arising from many conditions. Combined safety and availability of these agents make for a possibly valuable adjunctive therapy that should be considered.
| 1. Fardeau C, Champion E, Massamba N, LeHoang P. Uveitic macular edema. J Fr Ophtalmol. 2015;38(1):74-81. 2. Sigler EJ. Microcysts in the inner nuclear layer, a nonspecific SD-OCT sign of cystoid macular edema. Invest Ophthalmol Vis Sci. 2014;55(5):3282-4. |

