Innovations in Eye CareCheck out the other feature articles in this month's issue:Bioengineering the Retinal Pigment Epithelium Iontophoresis: Wave of the Future Can We Lower IOP with Glasses? A Light in the DARC: Seeing Glaucoma Before it Strikes |
Ocular involvement in diabetes is a near certainty as time goes on. Already, more than four million Americans experience some form of visual impairment from diabetic retinopathy (DR), and with the rates of diabetes steadily rising, eye care practitioners can expect far more cases.1 As in so many chronic conditions, therapy is often hampered by patient compliance issues, diminished efficacy over time and adverse effects.
Right now, the therapies retina specialists turn to are rather crude: burning the peripheral retinal with laser photocoagulation to reduce the metabolic demands that give rise to new vessel growth, or injecting anti-VEGF roughly every six weeks to arrest angiogenesis. Neither of these therapies are patient-friendly nor particularly effective long-term. An interdisciplinary team of ophthalmologists and engineers have their sights set on reducing this treatment burden and are developing a new method of phototherapy using contact lenses that could improve outcomes and reduce the adverse effect profile of long-term management.
“The big problem with diabetes in the retina is that the hyperglycemic state influences the endothelial cells and pericytes and really starts to affect blood flow, and this results in ischemia,” says Mark A. Humayun, MD, PhD, professor of ophthalmology and biomedical engineering at the University of Southern California. His research team is partnering with engineers from California Institute of Technology led by Yu-Chong Tai, professor of electrical engineering and medical engineering. The groups have collaborated to design and test a glow-in-the-dark contact lens for patients to wear overnight.
“The thought came to us—could we modulate ischemia? In the retina, you have a wonderful opportunity to do so during the nighttime period when it’s very metabolically active and requires a lot of oxygen,” says Dr. Humayun.
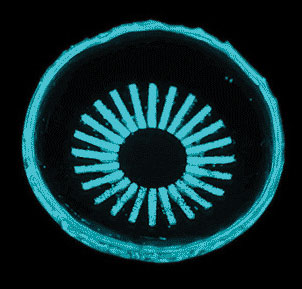 |
This glow-in-the-dark contact lens could help reduce DR-related damage from nighttime rod cell metabolism. |
Gas Guzzlers
The impaired vision in low light conditions that marks the beginning of visual loss in people with diabetes suggests that DR affects rod function first.2 This has subsequently been linked to increased oxygen consumption during dark adaptation, as well as retinal hypoxia and metabolic overload.3-6 This hypoxia upregulates VEGF and provokes the microvascular changes associated with the disease.
In the dark, rods consume more oxygen than any other cell in the body. When O2 is reduced, rod sensitivity decreases.7 If retinal disease constrains oxygen availability as a result of decreased retinal circulation, or the relative demand for oxygen increases due to metabolic stress, an imbalance will lead to hypoxia in both the inner and outer retina.7
“We wanted to modulate the activity of the retina and therefore not outstrip the oxygen supply of the retina in a diabetic patient, where it is already compromised,” Dr. Humayun says.
Research suggests that preventing dark adaptation by delivering 507nm light to the retina during the night might reduce the risk of DR progression.2 In 2017, PolyPhotonix Medical introduced the Noctura 400 Sleep Mask for treatment of late-stage DR patients and as a preventative measure for those at the early stages of the condition. The mask’s organic light emitting diodes (OLED) produce wavelengths that interact with the photoreceptors of the rods, but not those of the cones, at night, ensuring that it would not disturb the user’s sleep. The company suggested the OLED sleep masks would prevent the extra consumption of oxygen in dark adaptation while sleeping, which would then reduce inner retinal vascular stress.8
Published in The Lancet Diabetes & Endocrinology in early 2018, the CLEOPATRA study assessed the two-year outcomes of using the Noctura in non-central diabetic macular edema (DME). The results show that the light mask was not an effective option. While it significantly reduced DME and visible cysts in outer ETDRS zones at 12 months, the effect did not translate to significant change in retinal thickness and it was not sustained, suggesting that any positive morphological effects are transient and minimal.9 The study also found compliance waned over time and 75% reporting adverse effects, primarily related to disturbed sleep.10
While the study did note that the light mask was not an effective option to treat non-central DME, it was still worth evaluating other phototherapeutic techniques of rod suppression in DR and DME, a pursuit in which the interdisciplinary team has taken great lengths to engage.
See the Light
According to a paper Dr. Tai presented at an engineering conference (MEMS 2018), a key factor likely contributing to disturbed sleep from phototherapy is the amount of time-varying stimuli on the retina that inevitably results from trans-eyelid illumination. Differential transmission of light through the eyelid, which varies in thickness, results in a spatially heterogeneous illumination field.11 Since the eye moves with respect to this field, photoreceptors experience a time-varying illumination, to which the visual system is highly attuned.
If it were possible to fix the illumination field with respect to the eye—not the eyelid—the perception of the light stimuli would rapidly decay through a neural adaptation process known as the Troxler effect.12 This would likely make phototherapy more tolerable to patients. A second major limitation of trans-eyelid phototherapy is the uncertainty of photon dosing to the retina. Eyelid transmissivity is known to vary across the human population.13 Also, the eye rolls backward during sleep, which affects retinal dosage from the illumination source.
“We believe there are clear benefits to providing phototherapy from a source under the eyelid and immobilized with respect to the eye—and this is where contact lenses play a key role,” Dr. Tai wrote in his MEMS 2018 paper.11 So, the team recently constructed a phototherapeutic contact lens that may help to manage DR by suppressing the dark current in the rod and reducing retinal metabolism
 |
A light-emitting contact lens could potentially help slow down pathogenesis in early proliferative diabetic retinopathy. |
Making Contact
Given that retinal hypoxia drives VEGF expression, the research team aimed to create a phototherapeutic contact lens that increased the minimum retinal oxygen tension by 100%. They used a silicone-based contact lens embedded with a ring of gaseous tritium light sources providing continuous illumination over the lifetime of the lens. Currently, the lens is made up of approved polymers that give it acceptable wettabilty, comfort and fouling resistance.
Using the 1D retinal model on theoretical estimation of retinal oxygenation during retinal artery occlusion, the team found that suppressing rod metabolism to 132mm Hg/s was necessary to double the minimum value of retinal oxygen tension. They then estimated that metabolic suppression required retinal irradiance of 30 photons/s/µm2.11
When designing the lighting system, the team took into account the limitations of a contact lens package size, reliability and safety. They ultimately decided to use radioluminescent gaseous tritium light sources (GTLS) to satisfy the design conditions. These light sources are composed of tritium gas encapsulated in a phosphor-coated glass shell, similar to those used in glow-in-the-dark markers seen on wristwatches.
The tritium emits a high-energy electron that strikes and excites the phosphor, leading to highly reliable light emission. The light sources’ minute profile (300µm D × 2000µm L) enables integration into thin contact lens designs. Dr. Tai’s team selected a green emitting phosphor to maximize stimulation of rod cells at 498nm peak absorbance. The sources have a 12-year half-life and do not emit any ionizing radiation outside the glass shell, making them remarkably safe and reliable. However, the team recommends the lens be replaced after one year of use.11
According to Colin Cook, PhD, a researcher who worked closely with the contact lens engineering team in Dr. Tai’s lab during his doctoral program, fouling of the lens material due to repeated use is the limiting factor for the product’s lifetime.
The design has the GTLS arranged in an evenly spaced radial pattern starting at a radius of 1.5mm and ending at 3.5mm. “This annular arrangement of light sources provides an unobstructed view during photopic vision when the pupil is constricted, while directing the complete phototherapeutic dose through the dilated pupil under scotopic vision or sleep,” Dr. Tai explained in his paper.11
Testing the design with a human eye ensured that this annular arrangement provided a sufficient artificial pupil under photopic, or well lit, vision, while the GTLS could pass light through the dilated pupil under scotopic, or low light, vision. The wearer reported the disappearance of the light stimuli within several seconds since the contact lens was stationary on the eye under scotopic conditions; however, the patient could discern the light stimuli if the contact lens was manually repositioned on the eye.11 “Given the stability of the lens on the eye, the speed and completeness of the Troxler effect is remarkable,” Dr. Cook says.
Dr. Cook and the rest of Dr. Tai’s team transferred the prototypes over to Dr. Humayun’s group who then investigated the bioactivity of the lens through electroretinogram (ERG) flash response recordings in rabbits. The analyzed recordings revealed that both the amplitude and implicit time of the b-wave in the treated eye was found to be significantly shorter than in the untreated eye, a noted characteristic of suppressed dark adaptation.14 Regarding the difference in b-wave amplitude, the phototherapeutic contact lens caused an average suppression of rod cell dark adaptation to 32 ±2% of full dark adaptation.11 Adjusting for likely human response would put the expected rod cell suppression in diabetes patients at approximately 50%, exceeding the design specification but well within a tunable range.11
According to Dr. Humayun, they will need to collect human ERG data when possible to confirm or revise these estimates. He is also looking forward to verify and validate the lens on human test subjects as a baseline to understand what modifications and revisions need to be done in each future step. He encourages his colleagues to avoid over-engineering a prototype prior to human trials. “You have to do it safely, but there is a sense of urgency,” Dr. Humayun explains. “There are patients at the end of the day who could benefit from this.”
Ortho-K to the Rescue
Oxygen transmissivity of the lens has emerged as a key requirement to prevent corneal hypoxia during sleep. The FDA recommends a transmissivity of 125Fatt for overnight contact lenses.15 The 500µm thick lens has a transmissivity (Dk/t) of around 130Fatt, satisfying the FDA’s recommendation.
While many wear contact lenses in the daytime, the development of extended-wear lenses and orthokeratology (ortho-K) lenses has led to a better understanding of the requirements for overnight wear, including oxygen transmission.
Dr. Cook believes the key to ensuring long-term compliance with phototherapy for DR is coupling it with ortho-K myopia correction. Thus, the patient is motivated to wear the lens nightly for their daily vision correction, but in doing so helps prevents retinopathy from developing. Dr. Cook has so far been able to produce lenses with dual phototherapy and corneal refractive therapy functionality.
Since completing his doctoral program at Caltech, Dr. Cook has founded Retinox Medical to help continue his work translating phototherapy technology out of the lab and into the clinic. Currently, he has built functioning prototypes that rely on the same phototherapeutic principle but differ in their mechanism of light generation. There is a light-emitting diode (LED) variant that provides the ability to control light intensity and allow photon dosage to be fine-tuned for patients. Dr. Cook has also built a chemiluminescent version that could provide a disposable option similar to daily contact lenses.
Kids These DaysThe contact lens format in general may not be suitable for all DR patients, but is particularly well suited to adolescents and young adults. Type 1 diabetes specifically affects juveniles, who must manage the condition for the rest of their life. Given the near universal onset of retinopathy within 15 years of diabetes, they represent a particularly vulnerable group. Also, adolescents represent a unique subpopulation from a compliance perspective, as they are under the supervision of parents who are likely to enforce adherence to the preventative regimen. |
Night Moves
Dr. Humayun believes that the contact lens, if validated by human trials, could most likely be used with mild nonprolieferative DR patients to prevent progression. However, he says, “if severe nonproliferative, or even patients at the proliferative stages, can benefit somewhat from using the contact lens, the patient’s current injection intervals could be prolonged and they could reduce the number of injections in a year.”
Dr. Humayun sees the contact lens working in both scenarios, but it is too early to tell because they have yet to demonstrate how much the lens can actually modulate metabolic function in a severely compromised eye. “If we can modulate only a little bit, then obviously it will work on the milder form of the disease, but if we can modulate a fair amount, then can we work with the moderate and severe forms; in the latter two, it would be in combination with existing therapies,” Dr. Humayun notes.
Phototherapy represents a promising noninvasive preventative measure for diabetic retinopathy, and likely other hypoxic eye diseases such as macular edema and age-related macular degeneration. Incorporating a glow-in-the-dark light source for phototherapy inside an overnight lens could provide constant illumination to the retina while maintaining sleep quality. The lens may also combat patient compliance issues, as it minimally impacts the patient’s habits and quality of life.
| 1. Klein R, Klein BEK, Moss SE, et al. The Wisconsin epidemiologic study of diabetic retinopathy. Arch Ophthalmol. 1984;102(4):520-32. 2. Sivaprasad S, Arden G. Spare the rods and spoil the retina: revisited. Eye (Lond). 2016;30(2):189–192. 3. Arden GB, Wolf JE, Tsang Y. Does dark adaptation exacerbate diabetic retinopathy? Evidence and a linking hypothesis. Vision Res 1998; 38: 1723–1729. 4. Drasdo N,Chiti Z, Owens DR, North RV. Effect of darkness on inner retinal hypoxia in diabetes. Lancet. 2002;359: 2251–2253. 5. Harris A, Arend O, Danis RP, et al. Hyperoxia improves contrast sensitivity in early diabetic retinopathy. Br J Ophthalmol. 1996;80:209–13. 6. Kurtenbach A, Mayser HM, Jagle H, Fritsche A, Zrenner E. Hyperoxia, hyperglycemia, and photoreceptor sensitivity in normal and diabetic subjects. Vis Neurosci 2006;23:651–661. 7. Arden GB, Ramsey DJ. Diabetic retinopathy and a novel treatment based on the biophysics of rod photoreceptors and dark adaptation. In: Kolb H, Fernandez E, Nelson R, ed. Webvision: The Organization of the Retina and Visual System. Salt Lake City, UT: University of Utah Health Sciences Center; 2015. 8. PolyPhotonix. Noctura 400 media pack:a new approach to the management of diabetic retinopathy. noctura.com/wp-content/uploads/2017/12/Noctura-400-Media-Pack-20171214-01-1.pdf. December 12, 2017. Accessed January 9, 2019. 9. Sivaprasad S, Vasconcelos JC, Prevost AT, et al. Clinical efficacy and safety of a light mask for prevention of dark adaptation in treating and preventing progression of early diabetic macular oedema at 24 months (CLEOPATRA): a multicentre, phase 3, randomised controlled trial.Lancet Diabetes Endocrinol. 2018;6:382–91. 10. Sahni JN, Czanner G, Gutu T. Safety and acceptability of an organic light-emitting diode sleep mask as a potential therapy for retinal disease. Eye. 2017;31(1):97-106. 11. Cook CA,Martinez-Camarillo JC, Yang Q, et al. Phototherapeutic contact lens for diabetic retinopathy. Paper presented at: 2018 IEEE Micro Electro Mechanical Systems;January 21-25, 2018; Belfast, UK. 12. Troxler IPV, Uber das Verschwinden gegeb- ener Gegenstande innerhalb unseres Gesichtskreises. Ophthalmologische Bibliothek. 1804;2(2). 13. Bierman A, Figueiro MG, Rea MS, Measuring and predicting eyelid spectral transmittance. J. Biomed. Opt. 2011;16(6):67011. 14. Cameron A, Mahroo O, Lamb T, Dark adaptation of human rod bipolar cells measured from the b-wave of the scotopic electroretinogram. J. Physiol. 2006;575(2):507–26. 15. Harvit DM, Bonanno JA. Re-evaluation of the oxygen diffusion model for predicting minimum contact lens Dk/t values needed to avoid corneal anoxia. Optom Vis Sci. 1999;76(10):712–9. |

