Seeing Systemic Disease within the EyeThis month in Review of Optometry, multiple ODs explain the connection between the eye and various systemic conditions, including hypertension, stroke, sleep disorders, COVID-19, diabetes, thyroid eye disease and more. Check out the other articles featured in the October issue:
|
Since the announcement of the first confirmed cases of COVID-19 in Wuhan, China at the end of 2019, this coronavirus disease continues to be a global health crisis. According to the World Health Organization, as of August 5, 2022, there have been almost 600 million confirmed cases of COVID-19, including 6.47 million deaths.1
COVID-19 is characterized by fever and severe respiratory illness or pneumonia, but it can also affect the ophthalmic, neurological, cardiovascular, gastroenterological and nephrological systems.2 COVID-19 is a multisystem disease and can present with a wide array of clinical manifestations, ranging from asymptomatic to symptomatic with mild, moderate or severe disease.
SARS-CoV-2 is mainly spread via the respiratory tract through respiratory droplet transmission when an infected host sneezes, talks or coughs.3 It enters a cell by binding its spike protein with the cell’s angiotensin-converting enzyme 2 (ACE2) receptor.3-5 Transmembrane protease serine 2 proteins (TMPRSS2) prime the spike protein on SARS-CoV-2 for contact with ACE2, which enables infection of cells. The ACE2 receptor is found throughout the body which allows the virus to infect multiple organs. In the human eye, ACE2 is found in the retina and choroid tissues.3 One study demonstrated ACE2 expression in conjunctival, limbic and corneal cells and TMPRSS2 expression in the conjunctiva.5 While the presence of ACE2 receptors and TMPRSS2 proteins on the ocular surface seems convincing, there remains discussion on whether the tear film of infected COVID-19 patients contains viral particles.3
 |
|
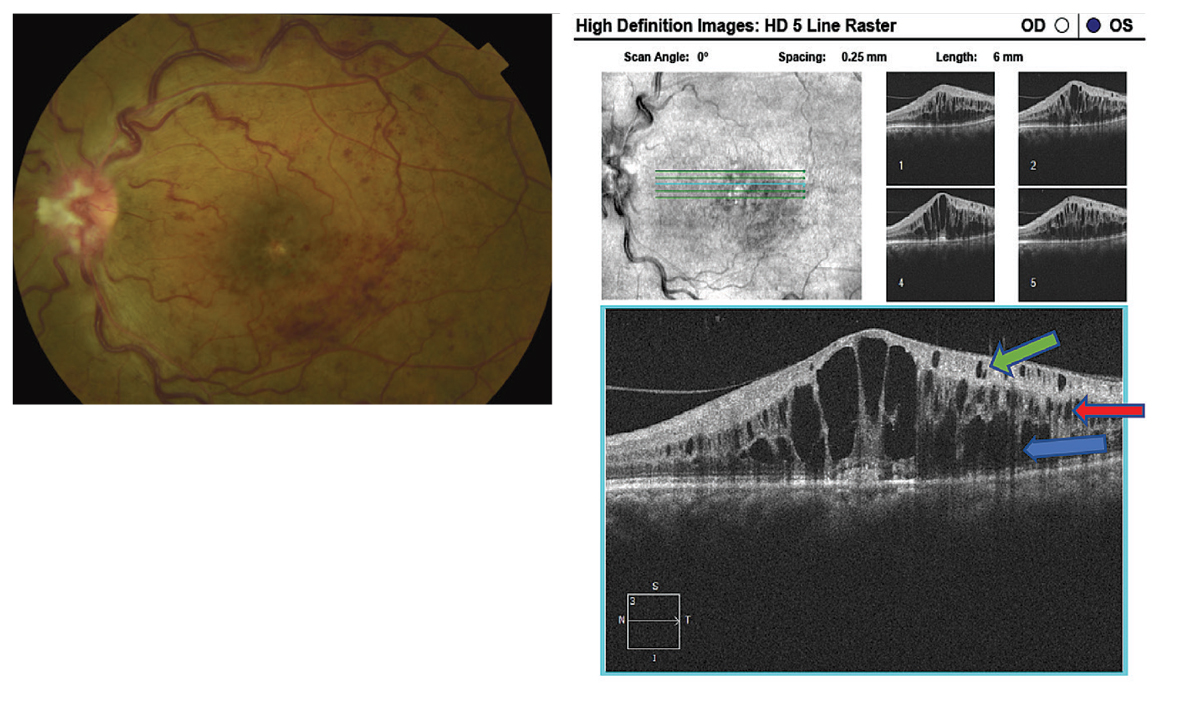
Fig. 1. Fundus photography demonstrating an ischemic retinal vein occlusion with, at right, SD-OCT illustrating the presence of cystoid macula edema with cysts located in the outer nuclear layer (blue arrow), inner nuclear (red arrow) and ganglion cell layer (green arrow). Click image to enlarge. |
Ocular Transmission and Complications
There are two different philosophies on COVID-19 transmission through the eye. The first considers the conjunctiva as a direct inoculation site of infected droplets. The second centers around the nasolacrimal duct as a way for viral movement to enter the gastrointestinal and respiratory tracts.3,6 One recent study showed that even though conjunctival cells expressed SARS-CoV-2, an infection did not result.7 A study published in July 2022 determined that SARS-CoV-2 was detectable with a conjunctival sac swab by polymerase chain reaction (PCR) testing in patients who had a PCR-positive nasopharyngeal swab, but the positivity rate was only about 12%.8
The prevalence of ophthalmic manifestations among COVID-19 patients ranges from 2% to 32%.4 Conjunctivitis has been the chief ophthalmic diagnosis identified in COVID-19 patients in the literature.2,4,6 Patients can present with numerous symptoms including ocular redness, soreness, foreign body sensation, light sensitivity, tearing, mucoid discharge, eyelid swelling and chemosis.2,4,6 These symptoms more commonly affect patients with severe systemic symptoms of COVID-19, although they can rarely present as an initial manifestation of the disease.
The onset of ocular symptoms typically occurs at the same time as systemic symptoms (47.5%), and 12.6% report symptoms lasting ≥14 days.9 Table 1 lists the most common orbital and anterior segment findings.2,4,6 Patients on ventilators or using respiratory masks in the ICU may also develop exposure keratopathy and ultimately severe corneal infections. Adequate ocular lubrication and closure of the eyelids may help prevent these ocular surface problems.2 As with other ocular viral infections, COVID-19 conjunctivitis is typically self-limiting and can be generally managed with symptomatic care. In the absence of significant ocular pain, decreased vision or light sensitivity, many patients can be managed with preservative-free artificial tears, cold compresses and lubricating ophthalmic ointment. In severe cases, a prophylactic antibiotic may be prescribed to minimize secondary opportunistic bacterial infections.
 |
| Click table to enlarge. |
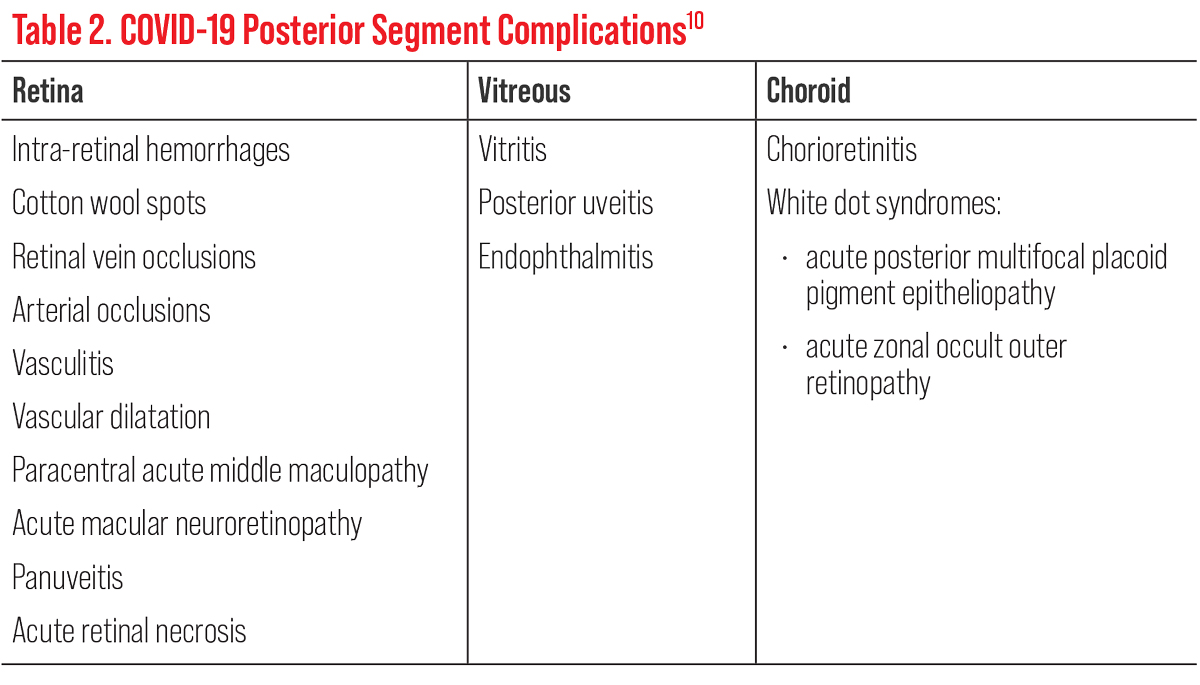
As seen in Table 2, the majority of COVID-19 posterior segment complications are retinal in nature but can manifest in the vitreous and/or choroid too.10 Posterior segment involvement has varied manifestations that are vascular, inflammatory and/or neuronal in response to viral COVID-19.11 These complications may occur within a week after the onset of COVID-19 symptoms up to more than six weeks after.12 Researchers believe that the retinal endothelium via the ACE2 receptors are affected from the virus. This causes compromise to the blood-retinal barrier, allowing a greater immune response.13 It can also cause severe endothelial disruption, complement activation and generalized inflammation.12 The choroid is highly vascularized, making it susceptible to systemic conditions, especially ones that affect the blood vessels.10
The most described non–vision threatening retinal findings in patients with COVID-19 are microvascular in nature, such as retinal hemorrhages and cotton wool spots.10,12 By contrast, the most common vision threatening manifestation is retinal vein occlusion with associated macular edema (Figure 1).11,12 Paracentral acute middle maculopathy and acute macular neuroretinopathy are rare microvascular diseases that involve the deeper retinal vessels. Their symptoms include acute painless diminution of vision, faintly colorful paracentral scotoma and dyschromatopsia.11 OCT testing in COVID+ patients shows hyperreflective lesions in different retinal layers and can include the posterior vitreous hyaloid, inner plexiform layer and ganglion cell layer with disruption of the ellipsoid zone.11,14
Neurological complications were initially known to include headache (6.5% to 70.3%) and olfactory disturbances. Today, severe neurological complications can present in up to 36% of patients as ocular manifestations and/or stroke.15-17 A range of symptoms have been reported: anosmia, ageusia, headache, dizziness, myalgias, diplopia, focal neurologic deficits and encephalopathy.16,18 In the setting of COVID-19 infection, an abnormal immune response contributes to neurologic dysfunction. Studies have shown that COVID infection can cause increased levels of cytokines in the plasma, creating a “cytokine storm” that can cause immunologic upregulation, vasodilation, vascular permeability, endothelial dysfunction, coagulopathy and direct viral neurotropism.16
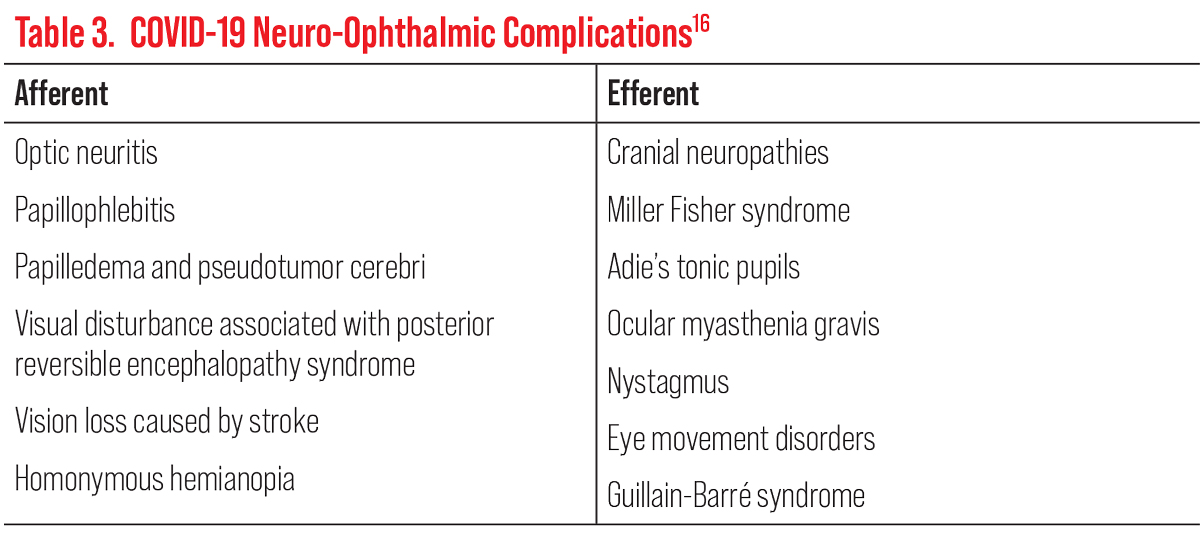
Current neuro-ophthalmic COVID anomalies can be classified into afferent and efferent visual pathway complications (Table 3).16 One of the most common noted afferent neurological complication is optic neuritis, often found in individuals with systemic inflammatory or autoimmune disorders.15 It is not uncommon for patients with optic neuritis and COVID to develop myelin oligodendrocyte glycoprotein antibody-associated optic neuritis or optic neuritis in the setting of acute disseminated encephalomyelitis.16 Efferent neurological complications commonly include pupillary and eye movement disorders with the sixth cranial nerve most affected.16 Systemic involvement can also include the loss of tendon reflexes and acute ataxia as seen in Miller Fisher syndrome.16
Ischemic stroke, rather than hemorrhagic, accounts for about 2% to 4.6% patients with COVID-19 and has been one of the most notable and devastating neurological complications of COVID-19.16,19,20 Although stroke is more common in the younger male population, early outcome is worse in women with a higher in-hospital mortality, longer hospital stays and more long-term disability. Patients requiring hospitalization for COVID-19 have a three- to four-fold greater risk of stroke.21 Despite gender differences, many ischemic stroke patients with COVID-19 have shared vascular risk factors, such as hypertension, hyperlipidemia and diabetes. In women, obesity, atrial fibrillation and heart disease are more prevalent.
Although the cause of ischemic stroke associated with COVID-19 is unclear, studies have hypothesized that it is due to an immune-mediated etiology, particularly related to an inflammatory response and hypoxemia. This promotes the occurrence of embolic event and increased blood viscosity, which causes a hypercoagulable state and thromboembolism.20
Visual complications from COVID-19 related ischemic stroke can result in vision and/or field loss and visual snow syndrome.19 These issues are the results of acute temporal, parietal or occipital infarct due to vasodilation and vascular permeability or due to endothelial dysfunction and/or coagulopathy.16 For example, our 51-year-old diabetic female patient who acquired COVID-19 in January 2022 with hospitalization later developed secondary blood clots and two ischemic strokes, one under hospitalization and another two months later. Although the patient’s visual acuity was 20/20 OU, it was confirmed on a Humphrey 120-point screener visual field analysis that she suffered a left-sided homonymous hemianopia field defect (Figure 2). Therefore, the risk of ischemic stroke must be taken into consideration when a patient has been admitted with COVID-19. Patients may benefit from the early initiation of anti-inflammatory and anticoagulant therapy. However, further clinical trials are needed for verified evidence.20
 |
| Click table to enlarge. |
Vaccine Complications
In December 2020, the Food and Drug Administration approved the first COVID-19 vaccine for emergency use authorization. There are currently four approved or authorized COVID-19 vaccines in the United States: Pfizer-BioNTech, Moderna, Johnson & Johnson’s Janssen and, most recently, Novavax. As of September 1, 2022, a total of almost 12.45 billion vaccine doses have been administered worldwide.1
A 2022 study looked at the adverse ocular events reported to the Vaccine Adverse Event Reporting System following vaccination against COVID-19 between December 2020 and December 2021. The most-reported events were eye swelling, ocular hyperemia, conjunctivitis (33.33%), blurred vision (26.69%) and visual impairment (19.77%). These conditions accounted for more than 70% of all complications.22 All of the different vaccine types administered in the USA were associated with ocular adverse events.
More severe anterior segment complications after COVID-19 vaccination include uveitis, scleritis, herpes zoster ophthalmicus, herpes stromal keratitis and corneal graft rejections.23 The underlying pathophysiology for vaccine-related uveitis is mostly unknown but has been hypothesized to be an autoimmune reaction triggered by the vaccines. Vaccination activates a cytokine cascade of pro-inflammatory type 1 interferon expression, which results in a defensive immune response. It can also trigger the production of autoantibodies responsible for an autoimmune response. Specifically for mRNA vaccines, mRNA can bind to Toll-like receptors that also activates a pro-inflammatory cytokine cascade.24
COVID-19 vaccinations may reactivate herpetic keratitis in patients with a previous herpetic keratitis or keratouveitis infection. Changes in immune status, including lymphocyte depletion, are thought to trigger the reactivation.25,26 Prophylactic antiviral treatment with oral valacyclovir should be considered, at least for high-risk patients with several previous episodes of herpetic uveitis.25,26
 |
| Click table to enlarge. |
Corneal graft rejection has been reported following administration of COVID-19 vaccines. Most cases had additional risk factors associated with rejection. Vaccination increases circulation of proinflammatory cytokines, CD4+ and CD8+ T-cell responses that may contribute to graft rejection. A patient’s risk of rejection varies depending on the transplant type (most commonly penetrating keratoplasty), the time elapsed since transplant and other associated risk factors.27,28 Prophylactic topical corticosteroids before and after vaccination may lessen the risk of rejection after vaccination but more research needs to be done. It is important to note that corneal graft rejection has also been reported following COVID-19 illness.27
Adverse neuro-ophthalmological complications related to vaccines against COVID-19 are shown in Table 4.29 At this time, optic neuritis is the most common. Most complications are mild and transient and have been clinically observed between four and 10 days post-vaccine.21,29 Seventeen days after receiving a second COVID-19 vaccination, a 65-year-old male suffered a right pupil-sparing third nerve palsy with external ophthalmalgia. MRI imaging was remarkable for edema and enhancement of the right oculomotor nerve. These clinical findings improved on day 30 and entirely resolved on day 71.30 Although, it has been hypothesized to be related to thrombosis and thrombocytopenia, cerebrovascular complications are rare.21 These immune-mediated events are respondent to high-dose steroids for treatment and have a favorable clinical outcome.30
Eyecare providers must be able to identify potential, although rare, post-COVID-19 vaccination adverse reactions. It should be kept in mind that ocular adverse events are not exclusive to COVID-19 vaccines and have been reported in many other vaccines, such as influenza, zoster, tetanus and pneumococcal vaccines.31 Overall, the benefits of COVID-19 vaccination outweigh the risks of ocular complications.
 |
|
Fig. 2. Humphrey visual field analysis with 120-point screen demonstrating a left-sided homonymous hemianopia visual field defect secondary to ischemic stroke from COVID-19. Click image to enlarge. |
Face Mask Complications
During the COVID-19 pandemic, the prevalent use of face masks was recommended to protect against the spread of SARS-CoV-2. Mask-associated dry eye was first mentioned in literature in June 2020.32 Since then, several articles have investigated mask-associated dry eye in the general population as well as in the healthcare workplace. A survey found that mask-associated dry eye risk factors may include longer mask wearing time, improper wearing of face masks, dry environment, older age, female sex, higher education and less outdoor time.33 Mask wearing has also be associated with meibomian gland dysfunction. One study noted that increased mask wear appears to correlate to an increased incidence of chalazion.34 Another determined that wearing masks decreased tear break-up time and increased ocular surface temperature and blood flow. A tightly fitting mask or taping the top of the mask may reduce the risk of dry eye symptoms associated with mask wear.12
Endophthalmitis, a purulent inflammation of the vitreous and aqueous material, has been known to manifest from exogenous or endogenous origin. Initially, it was hypothesized that wearing a face mask while receiving an intravitreal injection (IVI) may be associated with a higher risk of endophthalmitis due to the possibility of the increased chance of contamination by Streptococcus via oropharyngeal droplet transmission.35 A multicenter large-scale retrospective study found that universal masking during intravitreal injections did not alter the risk of post-injection endophthalmitis or post-IVI endophthalmitis; 0.020% vs. 0.035% in pre-COVID and COVID periods.35,36 Current standards of facial draping and 0.25% povidone-iodine ocular surface irrigation are successful prophylactic measures to reduce case incidences.35
 |
| Click table to enlarge. |
The Future of COVID
As we continue to understand COVID, what future outcomes are on the horizon? Present long-term complications include cardiovascular, neurological, psychological, hematological, pulmonary and dermatological problems.37 Current studies are delving into the most common “long-COVID” complaints, pulmonary and neurologic symptom-complex.38 Rapidly changing variants also complicate long-term studies of viral complications. Larger, published studies are needed to classify long-term symptom presentations and patient follow-up trends.37
The new mRNA vaccines must still overcome several challenges to meet worldwide market needs. The inability to control the virus has led to development of new variants that have shown a reduction in the effectiveness of therapeutics and vaccinations.37,39 Upcoming research and development for future variants rely on immunological vaccine design, total number of doses needed, dosing intervals and approaches to achieve safe, durable vaccine immunity in both children and adults.39 Further development is needed to find an adjustable formulation platform resulting in less or no adverse effects, access to low-income countries and tackling previous racial discrepancies.37,40
The success of mRNA vaccines in COVID-19 highlighted the potential for future disease management in infectious diseases, cancer and protein replacement therapies.40 Traditional administration routes of mRNA therapies are limited to intramuscular and intravenous in delivery, but forthcoming researchers are evaluating intranasal and oral delivery options.40 Patients with COVID-19, especially those with severe cases with multi-organ failure, have been shown to have markedly elevated inflammatory markers and proinflammatory cytokines. Multiple trials are underway to evaluate blocking these proinflammatory pathways to ease disease progression.
As we continue to learn more about COVID-19 variants and “long COVID,” it is likely that there are other ophthalmic manifestations that are still unknown. Clinicians must be aware of related complications of ocular disease with or without a formal COVID diagnosis and/or receipt of vaccinations. Future research will allow for better understanding of underlying mechanisms of disease variants, accurate treatments and the ability to prevent unwanted consequences of the infection or the vaccines.22.41
The CDC-supported COVID Data Tracker (covid.cdc.gov/covid-data-tracker/#datatracker-home) provides daily epidemiologic updates for the US regarding COVID-19. Also, one can further delve into local communal transmission data with trending variants and travel guidance recommendations.
Nonetheless, as more of the population is vaccinated, we are hopeful that systemic suffering and ocular complications will soon cease.
Dr. Smith is a staff optometrist at the Hampton VA Medical Center, where she is the externship director and adjunct faculty for The Ohio State University. She is an active Fellow of the American Academy of Optometry where she lectures and currently sits on the fellowship committee advisory board.
Dr. Pennington is a staff optometrist at the Hampton VA Medical Center, where she is adjunct faculty for The Ohio State University College of Optometry. She is the president of the Tidewater Optometric Society and a member of the board of trustees for the Virginia Optometric Association. Neither has any financial disclosures.
1. World Health Organization. WHO coronavirus (COVID-19) dashboard. Last updated August 31, 2022. covid19.who.int. Accessed September 1, 2022. 2. Leung EH, Fan J, Flynn Jr. HW, Albini TA. Ocular and systemic complications of COVID-19: impact on patients and healthcare. Clin Ophthalmol. 2022;16:1-13. 3. Davis G, Li K, Thankam FG, et al Ocular transmissibility of COVID-19: possibilities and perspectives. Mol Cell Biochem. 2022;477(3): 849-64. 4. Hu K, Patel J, Swiston C, et al. Ophthalmic manifestations of coronavirus (COVID-19). In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Updated May 24, 2022. ncbi.nlm.nih.gov/books/NBK556093/. Accessed September 1, 2022. 5. Zhou L, Xu Z, Castiglione GM, et al. ACE2 and TMPRSS2 are expressed on the humanocular surface, suggesting susceptibility to SARS-CoV-2infection. Ocul Surf. 2020;18:537-44. 6. Baj J, Forma A, Teresińska B, et al. How does SARS-CoV-2 affect our eyes-what have we learnt so far about the ophthalmic manifestations of COVID-19? J Clin Med. 2022;11(12):3379. 7. Jackson RM, Hatton CF, Spegarova JS, et al. Conjunctival epithelial cells resist productive SARS-CoV-2 infection. Stem Cell Reports. 2022;17(7):1699-713. 8. Furdova A, Vesely P, Trnka M, et al. Conjunctival swab findings in 484 COVID-19 patients in four hospital centers in Slovakia. Vision (Basel). 2022;6(3):46. 9. McHarg M, Wang Y, Yakin M, et al. Ocular symptoms In COVID-19 infection: a survey study. Res Sq. June 1, 2022. [Preprint]. 10. Zhang Y, Stewart JM. Retinal and choroidal manifestations of COVID-19. Curr Opin Ophthalmol. 2021;32(6):536-40. 11. Sen M, Honavar SG, Sharma N, Sachdev MS. COVID-19 and eye: a review of ophthalmic manifestations of COVID-19. Indian J Ophthalmol. 2021;69(3):488-509. 12. Sen S, Kannan NB, Kumar J, et al. Retinal manifestations in patients with SARS-CoV-2 infection and pathogenetic implications: a systematic review. Int Ophthalmol. 2022;42(1):323-36. 13. Sutandi N, Lee F. Vitreoretinal abnormalities in corona virus disease 2019 patients: what we know so far. Taiwan J Ophthalmol. 2021;11(3):232-43. 14. Abrishami M, Daneshvar R, Emamverdian Z, et al. Spectral-domain optical coherence tomography assessment of retinal and choroidal changes in patients with coronavirus disease 2019: a case-control study. J Ophthalmic Inflamm Infect. 2022;12(1):18. 15. Landis BC, Brooks AE, Digre KB, Seay MD. Coronavirus disease 2019, eye pain, headache and beyond. J Neuroophthalmol. 2022;42(1):18-25. 16. Tisdale AK, Dinkin M, Chwalisz BK. Afferent and efferent neuro-ophthalmic complications of coronavirus disease 19. J Neuroophthalmol. 2021;41(2):154-65. 17. Douedi S, Naser H, Mazahir U, Hamad AI, Sedarous M. Third cranial nerve palsy due to COVID-19 infection. Cureus. 2021;13(4):e14280. 18. Marsiglia M, Chwalisz BK, Maher M. neuroradiologic imaging of neurologic and neuro-ophthalmic complications of coronavirus-19 infection. J Neuroophthalmol. 2021;41(4):452-60. 19. Gold DM, Galetta SL. Neuro-ophthalmologic complications of coronavirus disease 2019 (COVID-19). Neurosci Lett. 2021;742:135531. 20. Luo W, Liu X, Bao K, Huang C. Ischemic stroke associated with COVID-19: a systematic review and meta-analysis. J Neurol. 2022;269(4):1731-40. 21. De Michele M, Kahan J, Berto I, et al. Cerebrovascular complications of COVID-19 and COVID-19 vaccination. Circ Res. 2022;130(8):1187-1203. 22. Nyankerh CNA, Boateng AK, Appah M. Ocular complications after COVID-19 vaccination, vaccine adverse event reporting system. Vaccines (Basel). 2022;10(6):941. 23. Wang MTM, Niederer RL, McGhee CNJ, Danesh-Meyer HV. COVID-19 vaccination and the eye. Am J Ophthalmol. 2022;240:79-98. 24. Chew MC, Wiryasaputra S, Wu M, et al. Incidence of COVID-19 vaccination-related uveitis and effects of booster dose in a tertiary uveitis referral center. Front Med (Lausanne). 2022;9:925683. 25. Bolletta E, Iannetta D, Mastrofilippo V, et al. Uveitis and other ocular complications following COVID-19 vaccination. J Clin Med. 2021;10(24):5960. 26. Ortiz-Egea JM, Sánchez CG, López-Jiménez A, Navarro OD. Herpetic anterior uveitis following Pfizer-BioNTech coronavirus disease 2019 vaccine: two case reports. J Med Case Rep. 2022;16(1):127. 27. Dugan SP, Mian SI. Impact of vaccination on keratoplasty. Curr Opin Ophthalmol. 2022;33(4):296-305. 28. Sen M, Honavar SG. After the Storm: Ophthalmic manifestations of COVID-19 vaccines. Indian J Ophthalmol. 2021;69(12):3398-420. 29. Lotan I, Lydston M, Levy M. Neuro-ophthalmological complications of the COVID-19 vaccines: a systematic review. J Neuroophthalmol. 2022;42(2):154-62. 30. Kubota T, Hasegawa T, Ikeda K and Aoki M. Case Report: Isolated, unilateral oculomotor palsy with anti-GQ1b antibody following COVID-19 vaccination. F1000Res. 2022;10:1142. 31. Cheng JY, Margo CE. Ocular adverse events following vaccination: overview and update. Surv Ophthalmol. 2022;67(2):293-306. 32. White DE. Blog: MADE: a new coronavirus-associated eye disease. June 22, 2020. www.healio.com/news/ophthalmology/20200622/blog-a-new-coronavirusassociated-eye-disease. Accessed September 1, 2022. 33. Fan Q, Liang M, Kong W, et al. Wearing face masks and possibility for dry eye during the COVID-19 pandemic. Sci Rep. 2022;12(1):6214. 34. Charters, Linda. Taping surgical masks to the skin decreases risk of ocular surface damage during the COVID-19 pandemic. Ophthalmology Times. May 2, 2022. www.ophthalmologytimes.com/view/taping-surgical-masks-to-the-skin-decreases-risk-of-ocular-surface-damage-during-the-covid-19-pandemic. Accessed September 1, 2022. 35. Tanaka K, Shimada H, Mori R, et al. Safety measures for maintaining low endophthalmitis rate after intravitreal anti-vascular endothelial growth factor injection before and during the COVID-19 pandemic. J Clin Med. 2022;11(3):876. 36. Mirghorbani M, Bazvand F, Riazi-Esfahani H, et al. Post-intravitreal injection endophthalmitis pattern during the COVID-19 pandemic with implementation of patients’ masking. Graefes Arch Clin Exp Ophthalmol. June 9, 2022. [Epub ahead of print]. 37. Desai AD, Lavelle M, Boursiquot BC, Wan EY. Long-term complications of COVID-19. Am J Physiol Cell Physiol. 2022;322(1):C1-11. 38. Robinson PC, Liew DFL, Tanner HL, et al. COVID-19 therapeutics: challenges and directions for the future. Proc Natl Acad Sci USA. 2022;119(15):e2119893119. 39. Altmann DM, Boyton RJ. COVID-19 vaccination: The road ahead. Science. 2022;375(6585):1127-32. 40 Szabó GT, Mahiny AJ, Vlatkovic I. COVID-19 mRNA vaccines: platforms and current developments. Mol Ther. 2022;30(5):1850-68. 41. Baj J, Forma A, Teresińska B, et al. How does SARS-CoV-2 affect our eyes-what have we learnt so far about the ophthalmic manifestations of COVID-19? J Clin Med. 2022;11(12):3379. |