 |
History
A 64-year-old Caucasian male presented to the office requesting a second opinion regarding what he described as a constant right eye turn. He explained that he realized the eye was suffering from reduced vision for approximately two years but could not elaborate on how the vision was lost. His motivation for seeking the correction was that he had recently failed the vision test for driving a school bus.
His ocular history included bilateral upper eyelid ptosis and right exotropia since childhood. He denied a history of double vision. He added that his brother and father have the same upper right eyelid presentation. He reported no systemic illness, no medications and denied allergies of any kind.
Diagnostic Data
His best-corrected visual acuity measured 20/200 OD and 20/30 OS at distance and near through spectacles (-6.75 -2.75x050/+2.50, -6.00 -2.50x155/+2.50).
Refraction uncovered anisometropia OD>OS (-10.50 -3.50x60, -6.25 -2.50x50) not improving vision OD and improving vision OS to 20/25 at distance and near.
His extraocular motilities were sluggish in all gazes with decreased adduction ability OD. The pertinent external data is demonstrated in the photographs.
Confrontation fields were full once the lids were lifted. There was no afferent pupillary defect. Biomicroscopy of the anterior segment revealed normal tissues and anatomy with grade II nuclear sclerotic cataracts. Goldmann applanation tonometry measured 15mm Hg OU. The dilated fundus findings were normal peripherally and centrally with normal nerves (no evidence of optic atrophy) and maculae.
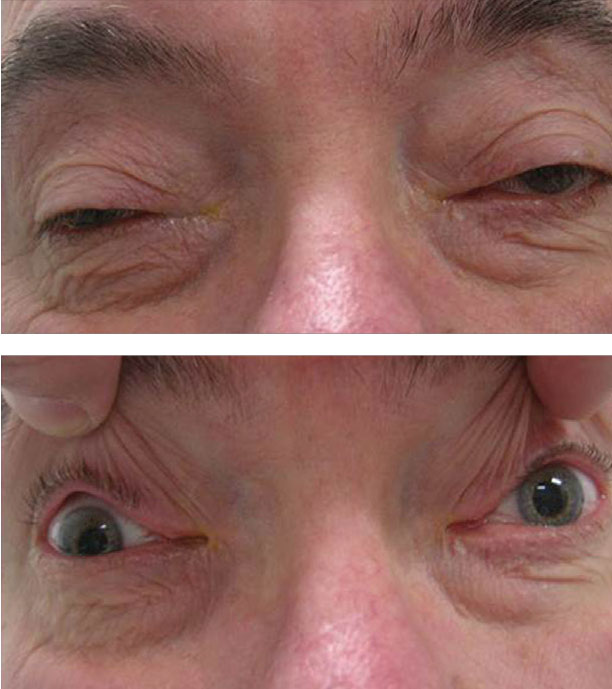 |
Bilateral upper lid ptosis was plainly evident on physical examination. What might it portend? Click image to enlarge. |
More Info Aids Diagnosis
Additional testing included the Randot Stereotest to quantify the level of suppression. Laser interferometry was performed to assess maximum macular acuity independent of the media or refractive error. Forced duction testing was employed OD to rule out restrictive myopathy in adduction, palpebral apertures were measured to document the ptosis OU and the patient was asked to bring in old photographs to confirm an evolving and longstanding issue as opposed to an acute event of only two years’ duration.
The patient was also systemically exercised and fatigued in different positions of gaze to grossly assess the possibility of myasthenia gravis. A neuro-ophthalmology referral was made to confirm the diagnosis and possibly provide additional genetic or lifestyle counseling.
The diagnosis in this case is bilateral mitochondrial myopathy associated with bilateral ptosis and chronic progressive external ophtalmoplegia (CPEO). The poor vision in the right eye is related to refractive and strabismic amblyopia as a result of asymmetric CPEO as well as amblyopia secondary to asymmetric ptosis produced by the evolving CPEO.
Chronic progressive external ophtalmoplegia (CPEO) is a disease of altered DNA.1-3 Mitochondria are ubiquitous organelles that play roles in vital functions; most importantly, they facilitate oxidative phosphorylation and energy metabolism.4 Mitochondrial dysfunction can affect numerous tissues—muscle and nerve included.4 Mitochondrial myopathy is a common clinical phenotype, which is characterized by early fatigue and/or fixed muscle weakness.1-5 When the condition slowly affects the extraocular muscles, producing progressively limited function, it is known as chronic external ophthalmoplegia.2
DNA Details
Approximately 1500 nuclear genes can affect mitochondrial structure and function.4 The identification of causative molecular defects in the nuclear or mitochondrial genome is what permits the diagnosis of mitochondrial myopathy.4
Mitochondrial diseases are considered to be a group of heterogeneous disorders that occur secondary to impaired cellular respiratory chain function.8 The result of this is defective ATP production.8 Although the disruption of oxidative phosphorylation is central to the mitochondrial disease pathophysiology, other factors contribute.8
The cellular localization of tumor necrosis factor alpha (TNF-α) and its receptors TNFR1 and TNFR2 in muscle biopsies from patients in one study with mitochondrial respiratory chain dysfunction suggests that TNF-α is expressed in muscle fibers with abnormal focal accumulations of mitochondria, producing the classic biopsy finding of ragged red fibers.8 Further, TNF receptors seem to be regulated in the muscles of patients where the expression levels of TNFR1 messenger RNA (mRNA) are decreased and the expression levels of TNFR2 mRNA are increased compared with controls.8 These findings suggest for the first time that TNF-α could exert a direct effect on mitochondria via its receptors.8
Prognosis
The clinical course of mitochondrial myopathy is slow and progressive.1 It is rare to find rapidly progressive disease in either the eye or system.1-7 Systemically reported symptoms of mitochondrial myopathies are permanent tiredness, easy fatigability, muscle aching at rest or already after moderate exercise, muscle cramps, muscle stiffness and muscle weakness.1 The clinical diagnosis is based on the history, clinical neurologic examination, blood chemical investigations, lactate stress test, electromyography, magnetic resonance imaging, magnetic resonance spectroscopy, muscle biopsy, biochemical investigations of the skeletal muscles and genetic investigations.1
At the moment, only symptomatic therapy is available and includes physiotherapy and orthopedic supportive devices, diet, symptomatic drug therapy (e.g., analgesics, cramp-releasing drugs, antioxidants, lactate-lowering drugs, alternative energy sources and co-factors), avoidance of mitochondrion-toxic drugs, surgery (e.g., correction of ptosis or orthopedic problems) and, in case of respiratory failure, invasive or non-invasive mechanical positive pressure ventilation.1 While it is rare, cardiac complications have been reported in the literature in association with CPEO. Therefore, a cardiac evaluation should be arranged.9
The patient’s full time spectacle prescription was updated to provide the best vision at distance and near, ordered in polycarbonate material to provide protection for the good eye. The patient was educated that their poor vision OD and missing binocularity was the result of a combination of the refractive amblyopia and strabismic amblyopia produced by the CPEO, along with the accompanying natural suppressive adaptation that developed. Also discussed was the possibility that an adult strabismus surgeon might be able to increase the cosmetic appearance but that functional improvement was not possible. The patient was also educated that ptosis surgery was another option to improve both cosmetic appearance and vision by lifting the eyelids so that they no longer obstructed viewing. The disadvantage of the procedure is that lifting the eyelids would expose the now large-angle constant exotropia OD. The patient did follow through with the neuro-ophthalmology consultation but has not yet decided on the other procedures.
| 1. Finsterer J. Mitochondrial myopathies. Fortschr Neurol Psychiatr. 2009;77(11):631-8. 2. Finsterer J, Ahting U. Mitochondrial depletion syndromes in children and adults. Can J Neurol Sci. 2013;40(5):635-44. 3. Russell AP, Foletta VC, Snow RJ, Wadley GD. Skeletal muscle mitochondria: a major player in exercise, health and disease. Biochim Biophys Acta. 2014;1840(4):1276-84. 4. Milone M, Wong LJ. Diagnosis of mitochondrial myopathies. Mol Genet Metab. 2013;110(1-2):35-41. 5. Lenaers G, Hamel C, Delettre C. Dominant optic atrophy. Orphanet J Rare Dis. 2012;7(7):46. 6. Wiwatwongwana A, Lyons CJ. Eye movement control and its disorders. Handb Clin Neurol. 2013;113(1):1505-13. 7. Caballero PE, Candela MS, Alvarez CI, Tejerina AA. Chronic progressive external ophthalmoplegia: a report of 6 cases and a review of the literature.Neurologist. 2007;13(1):33-6. 8. Vattemi G, Marini M, Ferreri NR, et al. Overexpression of TNF-α in mitochondrial diseases caused by mutations in mtDNA: evidence for signaling through its receptors on mitochondria. Free Radic Biol Med. 2013;63(10):108-14. 9. Pfeffer G, Mezei MM. Cardiac screening investigations in adult-onset progressive external ophthalmoplegia patients. Muscle Nerve. 2012;46(4):593-6. |

