 |
History
A 57-year-old black female presented for an eye examination with a chief complaint of near blur. She explained she had lost her job two years earlier and, with it, her vision and health insurance. She had no history of previous ocular or systemic disease, took no medication and reported no allergies.
Diagnostic Data
Her best corrected entering acuities with her -6.25/+2.25 spectacles 20/30, OU at distance and near. Her external examination was normal and no evidence of afferent pupil defect was noted. Her refraction uncovered -7.00D of myopia, correctable to 20/20.
Her biomicroscopic examination found normal anterior segment structures, open angles, mild cataracts and Goldmann applanation pressures measuring 15mm Hg in both of her eyes.
The pertinent dilated fundus finding in the right eye is documented in the photograph.
Discussion
The diagnosis in this issue is a horseshoe-shaped retinal tear with subretinal fluid (SRF) and small shallow focal retinal detachment.
Retinal detachments occur in approximately 6.1 in 100,000 people in the general phakic population.1-3 The rates between men and women are similar worldwide, with a slight preponderance for females who have myopia.2-4 There are three recognized forms of retinal detachment.1-9 These include: rhegmatogenous retinal detachment (RRD-resulting from a retinal break), exudative, or serous, retinal detachment (ERD-resulting from fluid accumulation under the sensory retina without a retinal break) and tractional retinal detachment (TRD-resulting from the pull of proliferative fibrovascular vitreal strands).1-10
Any type of retinal detachment may be initially asymptomatic. RRD may remain asymptomatic up to discovery.11,12 In symptomatic RRD, patients report photopsiae (flashes of light), floating spots, peripheral visual field loss (curtain phenomenon) and depending upon the involvement of the macula, central blurring of vision with or without metamorphopsia.1,13 There are anecdotal reports of exudative retinal detachments producing photopsiae (flashing purple lights) but the common symptoms experienced by these patients are vision loss and metamophopsia consistent with the degree of macular involvement, with or without a visual field deficit.5,14
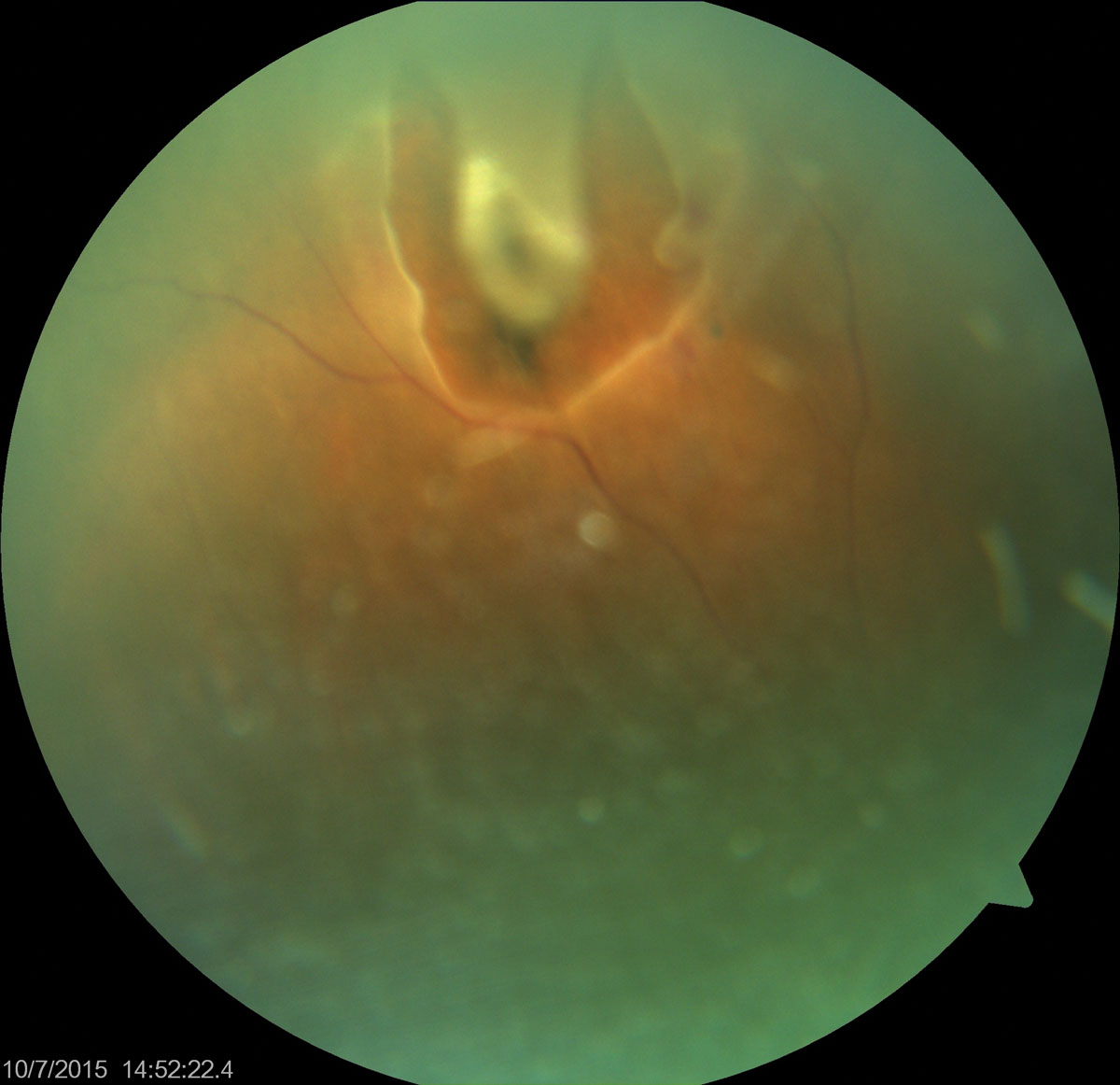 |
| Although this patient only presented for near vision blurring, imaging found something more pressing. Do you recognize this presentation? Click image to enlarge. |
TRD have the capacity to produce the same symptoms as rhegmatogenous and exudative retinal detachments. They may also remain asymptomatic until central vision is threatened.5,15 Pain is not a feature of any retinal detachment as the tissue has no pain receptors. In fact, the only sensory receptors in the retina are for light; hence the sensation of flashing lights experienced by patients from mechanical vitreoretinal tractional forces.12 Any pain encountered by a patient experiencing any form of retinal detachment is secondary to an associated cause such as headache, iritis, corneal abrasion, uveitis or raised intraocular pressure and not the detachment itself.1,5,16,17 Extensive unilateral retinal detachment will produce a relative afferent pupillary defect.1,16 Intraocular pressure may be notably reduced in eyes with acute retinal detachment.19-21
Clinical observation of fresh RRD usually reveals a clumping of pigment cells within the vitreous (Shaffer’s sign/tobacco dust) adjacent to the retinal break.1,22 An area of white or grayish elevated retina may be seen adjacent to the instigating retinal break secondary to influx of subretinal fluid.4 If a significant area of the retina is involved it may appear bullous and undulating. A rhegmatogenous detachment is produced by a retinal break that allows liquefied vitreous to separate the sensory retina from the retinal pigment epithelium (RPE) through poorly understood posterior segment fluid mechanics.21 Osmotic and oncotic pressures help keep the retina in place.22 As such, RRD do not change positions when body posture is altered.23 RRD do shift and return to their original orientation with quick eye movements.1 Associated findings of RRD may include posterior vitreous detachment and preretinal or vitreal hemorrhage.24-26 Retinal pigment epithelial hyperplasia may be noted in cases of long-standing retinal detachment of any kind (pigment demarcation line). Increased RPE density is a feature of attempted self repair.1,5,27
ERD appear as focal, serous elevations of the retina in the absence of retinal breaks.5,28-34 Because the fluid is contained underneath an intact neurosensory boundary, the bullous separation possesses the characteristic of following gravity, shifting position with changes in posture and eye movement.1,5 Ophthalmoscopic observation reveals a smooth, translucent, dome-shaped protrusion of the retina along with variable other signs secondary to the causative etiology (blood, exudate, or serosanguinous fluid.5 Causes of ERD include Coat’s disease, age-related macular degeneration, idiopathic central serous chorioretinopathy (ICSC), fluid exudation from chroidal tumors and Vogt-Koyanagi Harada syndrome, among others.5,28-34
TRD is always associated with fibrovascular vitreal strands and membranes.1,35-37 The clinical appearance of these detachments is varied with tangential fibrovascular bands anchoring into the vitreous body and extending to the dis-inserted retina.1,35-37 The tractional membranes may encircle intact retina, resulting in a “pseudo-hole” appearance. TRDs are dense and immobile, as compared with ERD and RRD. Any pathology that can induce posterior segment ischemia and retinal neovascularization can proceed to TRD. The common underlying causes include diabetes, vein occlusion, ocular ischemic syndrome, retinopathy of prematurity and sickle cell disease.35-39
All retinal detachments involve a dissection of the sensory retina from its underlying RPE layer by SRF.1-40 In rhegmatogenous detachments this fluid is thought to be composed of liquefied vitreous, which gains access to the subretinal space via a retinal break.1,22 In exudative detachments, the fluid is derived from the choroid, passing through a breach in Bruch’s membrane.5,28-34 The origin of the SRF in tractional retinal detachments is similar with slightly varied mechanisms. Generally, altered balance between the passive and active movement of SRF induces RD progression.1 While all retinal detachments have the potential to produce visual scotomata (depending upon their size and location), it is the involvement of the macular region, where apoptotic mechanisms deteriorate macular photoreceptors, that will determine the extent of acuity loss.40
Retinal breaks are the predisposing factor in patients with rhegmatogenous retinal detachment.1,22-27 These may occur spontaneously from preexisting conditions or as a result of ocular trauma.1-3 Some of the common entities associated with RRD include lattice degeneration, flap tears, atrophic holes, operculated retinal breaks and acquired retinoschisis with both inner and outer holes.1,3,4,41 As the retinal tissue loses its connection to the RPE, it becomes edematous and dysfunctional.22 The detached retina loses its oxygen supply and relies on anaerobic pathways to metabolize glucose.22 Long-duration retinal detachments feature increased lactic acid and dextrose concentrations.23 Phospholipids are also increased in the SRF, reflecting retinal organelle degradation.23 Eventually, photoreceptor death occurs within 48 hours to 72 hours unless surgical intervention is employed.23,40
ERD occurs in association with subretinal disorders which damage the RPE layer.5,28-34 Transudation of fluid from the choroidal reservoir through Bruch’s membrane and a breach in the RPE overcomes the eye’s natural mechanisms for deturgessing the plasma solution, causing it to build under the photoreceptors. When the threshold is reached it causes them to disinsert from the RPE.5,23,28-34 Affected by gravity, as the fluid accumulates, the detachment will shift with eye and head movements. However, since the density of fluid affecting the retina changes with movement, no particular area of the retina is continuously affected.5 This may explain why patients with ERD have final functional outcomes that are better than those with RRD or TRD.5,28-34
TRD occurs in the presence of proliferative vitreoretinopathies.1,35-39 The etiology of TRD involves fibrotic scaffolding of the vitreous along proliferative vascular networks, which through vitreal shrinkage, induce strong anterior tractional forces.39,42-44 RPE cell proliferation and migration are believed to play a role in the pathogenesis.43 Findings suggest that the vitreous contributes modulators that stimulate RPE cells along with macrophages, fibroblasts and glial cells to interact with constituents of the extracellular matrix such as fibronectin, vitronectin, and factor XIII.43,44 These mechanisms induce the formation of membranes that capture the sensory retina and forcibly separate it from the underlying RPE.39,42-44 Unlike rhegmatogenous or exudative retinal detachments which tend to occur acutely, TRD often develop slowly. When positioned peripherally TRD may not be noticed by the patient until visual acuity is compromised by the underlying disease process.
Retinal detachments demand repair and treatment of both the retina and the underlying cause.1-49 Patients presenting with an acute onset RRD involving or threatening the macula warrant an immediate and emergent referral to a retinal surgeon. Fresh RRD should be repaired within 24 to 48 hours; chronic or long-standing RRD or RRD that do not threaten the macula should be addressed within one week of diagnosis.50,51 Small peripheral RRD secondary to atrophic holes or RRD secondary to small tears displaying minimal SRF may be managed with barrier laser photocoagulation or cryopexy.1,45-49 While cryopexy has been reported to provoke a more aggressive post operative inflammatory response, its outcomes over time compared to laser barrier treatment are similar.49 An advantage of cryopexy over laser procedures is that it is generally less expensive and does not have to be repeated.49 Larger RRD require surgical repair using procedures that include vitrectomy, scleral buckling, needle aspiration, laserpexy, cryopexy, pneumatic retinopexy and intraocular silicone oil tamponade.13,52-55
Vitrectomy has been investigated as a principle treatment method for RRD.56-58 Vitrectomy seems to allow improved control of more complicated situations.57 The Scleral Buckling vs. Primary Vitrectomy in Rhegmatogenous Retinal Detachment Study (SPR study) is a prospective, randomized, multicenter study comparing primary vitrectomy with or without additional scleral buckling to scleral buckling alone.56,58 In the pseudophakic subgroup, no difference in functional outcome was seen; however, better anatomical results with a lower rate of retina-affecting re-operations was observed in the vitrectomy group.56-58 Based on this data, primary vitrectomy combined with a scleral buckle is the method of choice in complicated retinal detachment in pseudophakic patients. In contrast, primary vitrectomy does not seem to offer an advantage over scleral buckling in phakic patients.56,58 The primary drawback of vitrectomy is its significant propensity to create cataract and postpone complete visual recovery.58
Scleral buckling is accomplished under general anesthesia where a soft silicone sponge or hard silicone band is used to indent the eye at the location of detachment.59 The intent of the buckle (explant-on top of the sclera, implant-placed into a scleral dissection) is to eliminate the vitreoretinal traction that induced the retinal tear and to prevent fluid seepage underneath the retinal break.1,59 This process also encourages RPE pumping to eliminate the SRF. Drainage of SRF via syringe is controversial with some believing it is not necessary and others believing it is crucial.58 Raised IOP, choroidal detachment, diplopia, macular edema and macular pucker are all potential complications.59
Pneumatic retinopexy employs an intravitreal gas bubble (usually perfluoropropane, C3F8 or sulfahexafluoride) to achieve reattachment of the retina for RRD.60-62 This technique is performed under local anesthesia and is more common for treating smaller, superiorly located RRD.60 Careful eye and head positioning are important post-operatively to ensure resolution.60 In certain instances, silicone oil tamponade may be favorable to either of the aforementioned techniques.63 The use of polydimethylsiloxane (PDMS) as a silicone oil endotamponade has become a standard in retinal surgery.63 In cases of complicated inferior and posterior retinal detachment heavy silicone oils are sometimes considered.63 A randomized prospective clinical trial (HSO study) comparing heavy and standard silicone oil in patients with PVR of the lower retina have failed to demonstrate superiority of a heavy oil tamponade.64
Exudative detachments, because of their nature, generally require less intervention than RRD.5,7 ERDs may resolve spontaneously or following management of the underlying condition.5,7,23,28-34 This may involve oral antibiotics in cases involving infection, high dose oral or vitreal injectable/implantable corticosteroids in the case of inflammatory disorders, oral acetozolomide in cases of ICSC and radiation therapy and/or local resection in the case of intraocular neoplasms.65-67 In cases involving choroidal neovascularization laser photocoagulation, photodynamic therapy and vascular endothelial growth factor inhibitor injections may all be used.65,68
Tractional retinal detachments are more difficult to manage than either RRD or ERD. Managing the underlying cause is an essential precursor.39,35-37 Endolaser scatter photocoagulation must be employed directly to the retina to correct the underlying inciting retinal cause for new vessel growth.39 Surgical repair of TRD involves pars plana vitrectomy to remove the traction and the fibrovascular membranes along with the inciting vitreal cytokines. Gas or oil tamponade is used to promote retinal reattachment.63,64,69
Summarizing, all patients presenting with symptoms of retinal detachment or a predisposing history (peripheral retinal thinning or breaks, blunt ocular trauma, proliferative diabetic vitreoretinopathy, etc.) must undergo a thorough dilated fundus evaluation, with scleral indentation where appropriate, fresh rhegmatogenous detachments should be immediately referred for evaluation of surgical intervention, the effect of gravity increases the risk for superior detachments to spread. conservative surgical management may be indicated for partial or sectoral RRD (laser barrier).
The patient was referred for immediate barrier laser intervention. This was completed and there were no complications.
|

