The ocular surface is comprised of the cornea, conjunctiva, eyelids, eyelashes, tear film, main and accessory lacrimal glands and meibomian glands.1 The eyelids play a major role in protecting and spreading the moisture over the ocular surface. The lower eyelid supports the tear film, and the glands of Zeiss and Moll, as well as the meibomian glands, secrete lipids. The blinking action stimulates the release of lipids into the tear film as well as moves the tear film towards the puncta. Any disruption in the normal anatomy and physiology of this system can cause the patient to become symptomatic of ocular surface pathology.
The Grand Tour
When scanning the closed eyelids, pay attention to the epidermis (e.g., peeling, scaling) and dermis (e.g., hyperemia, edema, ecchymosis), as well as the position of the eyelids to rule out entropion and ectropion. Additionally, be on the lookout for any lesions that disrupt the proper anatomy or cause inflammatory pathology, such as papillomas, molluscum contagiosum, herpetiform vesicles, hordeola or chalazia. Ask yourself: are the puncta open?
While scanning the eyelashes, direct your attention to any greasy scales, cylindrical dandruff around the roots of the lashes, or discharge. Check for any misdirected lashes rubbing against the ocular surface. Ask the patient to look down, and while pulling up the upper eyelid to scan the eyelid margin. Observe the meibomian gland orifices and the tissue around them. Note any capping or telangectasias, as well as any changes in the normal contour of the eyelid margins. Examine the superonasal, superior and superotemporal bulbar conjunctiva. Blebs created during trabeculectomy will be visible superior to the cornea. Evert the upper eyelid and scan the superior palpebral conjunctiva looking for foreign bodies, concretions, papillae or hyperemia.
Ask the patient to look up, so you can pull down the lower eyelid to scan the eyelid margin. Apply pressure to the glands expressing the meibum and evaluate the appearance of the secretion. Pull down a little farther to examine the lower palpebral conjunctiva looking for cystic changes, concretions, follicles, papillae, hyperemia and foreign bodies. While the patient is still looking upward, scan the inferonasal, inferior and inferotemporal bulbar conjunctiva while asking the patient to change gazes. Scan the nasal canthus then nasal bulbar conjunctiva, for it is not uncommon to note pinguecula and pterygium as you approach the limbus. Scan the temporal bulbar conjunctiva looking for lid-parallel conjunctival folds that could indicate conjunctivochalasis (CCH).
Next, proceed to examine the corneal epithelium, stroma and endothelium, looking for punctate epithelial erosions, limbal neovascularization, dystrophies and degenerations, old scars from foreign bodies, trauma, refractive surgery or previous infections, as well as pannus, infiltrates, edema, guttatae, endothelial pigment and endothelial folds.
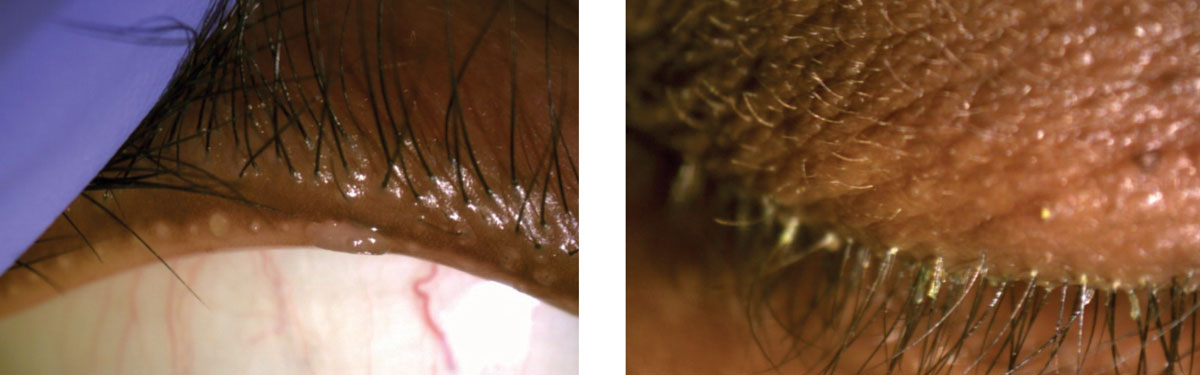 |
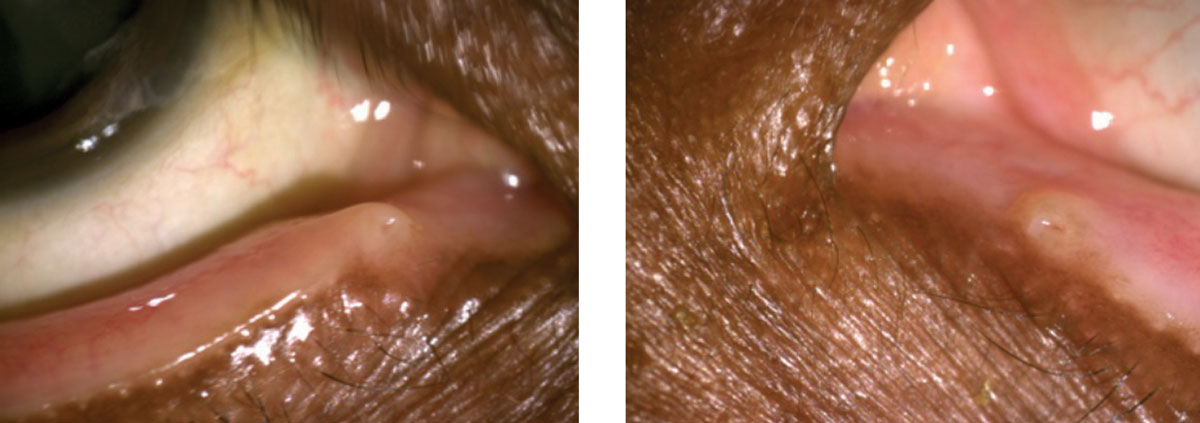
| At left, this anterior segment photo depicts severe meibomian gland capping and a small squamous cell papilloma of the eyelid margin. The image on the right shows cylindrical dandruff around the roots of the lashes. |
Lid Abnormalities
In most countries, life expectancy is increasing, so an expected increase in ophthalmic conditions due to involutional changes are expected as well. It is not uncommon to diagnose involutional entropion or ectropion in middle-aged and older adults. Although increasing age is not the sole cause of lid anomalies, such as entropion and ectropion, involutional changes are deemed the most common.2 It is not uncommon to find more women to be afflicted with involutional entropion than men.3 In addition to involutional causes, entropion and ectropion can also stem from a cicatricial etiology.
Trichiasis
This is a condition in which lashes can grow or be misdirected toward the eye. Every time the patient blinks or even rubs their eyes, the possibility of the lashes scratching against the ocular surfaces increases. Trichiasis can cause irritation, tearing and pain. Trichiasis is commonly caused by trachoma, which can lead to significant corneal scarring. However, trachoma is uncommon in the United States, so cases here are usually deemed idiopathic or secondary to a traumatic etiology, ocular cicatrical pemphigoid, Stevens-Johnson syndrome, chemical burn or severe blepharitis.4
It is not uncommon for the signs and symptoms of punctate epithelial erosions, along with tearing, foreign body sensation and redness to be shared between trichiasis and entropion. Left untreated, trichiasis may result in serious ocular sequelae such as corneal ulcers, punctate keratopathy, abrasions and scarring.4 The quality of these patients’ vision, as well as the ocular surface, can be severely impacted in these cases, so it is imperative to be aware of misdirected lashes when evaluating ocular health. Because some patients may be asymptomatic, clinicians must have astute observation skills.
Entropion
During the initial assessment of the ocular health, you may notice inward turning of the eyelid. If you do, you must determine if there is only trichiasis (an inward turning of the eyelid margin).
Entropion always presents with trichiasis (assuming the patient has eyelashes), but trichiasis can occur without entropion. Due to the frictional forces that occur with blinking, both conditions can cause significant punctate erosions on the cornea, which may cause the patients to present with complaints of redness, foreign body sensation, irritation and tearing.
The ocular surface and pathologic eyelid findings associated with involutional entropion include lateral canthal tendon laxity (78%), dry eye (72%), superficial punctate keratopathy (62%), lower retractor laxity (53%), chronic blepharitis (49%), chronic conjunctivitis (23%), and medical canthal tendon laxity (15%).3
Ectropion
On the other hand, during examination you may notice an outward turning of the eyelid (ectropion). This has several possible causes, but an age-related or involutional etiology is the most common. Like involutional entropion, ectropion has a gender bias as well. Older males are more likely to be afflicted with involutional ectropion.3
Because of the ectropion, you may notice punctate epithelial erosions secondary to rapid tear evaporation and chronic exposure of the ocular surface. Ocular surface and eyelid abnormalities associated with involution ectropion include lateral canthal tendon laxity (80%), dry eye (52%), chronic blepharitis (43%), chronic conjunctivitis (40%), lower retractor laxity (40%).3
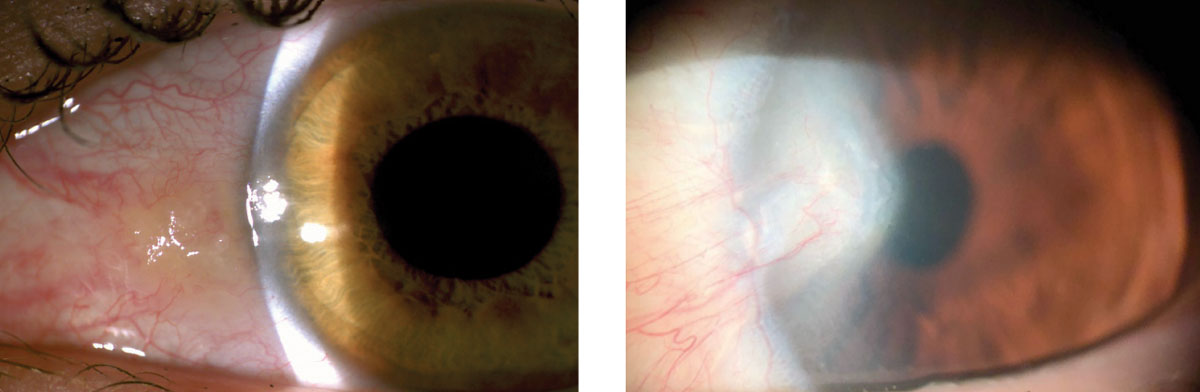 |
| These photos show patients with pinguecula (left) and pterygium (right). |
Punctal Stenosis
Punctal stenosis may be easy to overlook during the clinical exam—in fact, if you quickly gloss over the punctum during your biomicroscopic exam, stenosed or closed puncta may not even be observed. However, in patients older than 80, it is common.
Clinically, patients are diagnosed with dry eye disease due to their epiphora complaints; however, instilling artificial tears in a patient with punctal stenosis only exacerbates their complaints. In cases such as these, it is best to observe a stenosed puncta when the patient has complaints of epiphora. Interestingly, punctal stenosis, also known as external punctal stenosis (EPS), results from one of two possible etiologies, acquired and age-related. Acquired causes can be due to topical or systemic medication use, various infections, lid malposition, trauma or tumors.5-7 Age-related changes are mostly due fibrosing of the tissue surrounding the puncta.8
In cases of acquired punctal stenosis, practitioners should be aware of the possible relationship between EPS and long-term use of topical anti-glaucoma medications. Because practitioners commonly encounter patients with ectropion, they should also be aware of the relationship between ectropion and punctal stenosis. It has been postulated that the cause of punctal stenosis in relationship to lid ectropion is due to underuse of an external punctum unopposed to the tear meniscus or secondary to inflammation.9
Patients with punctal stenosis may also present with dry eye disease. Punctal plugs are commonly used in the management of patients with dry eye disease. Clinically, stenosed puncta function similarly to punctal plugs, so patients, especially the elderly, may benefit from having stenosed puncta. One study suggested not to promote surgical intervention in cases of punctal stenosis in order to prevent disruption of the suspected protective mechanism.10 However, practitioners should remember that punctal plugs and stenosed punctum may be contraindicated in cases of inflammatory-based dry eye disease due to the possibility of exacerbating the condition from retained inflammatory cytokines in the tear film.
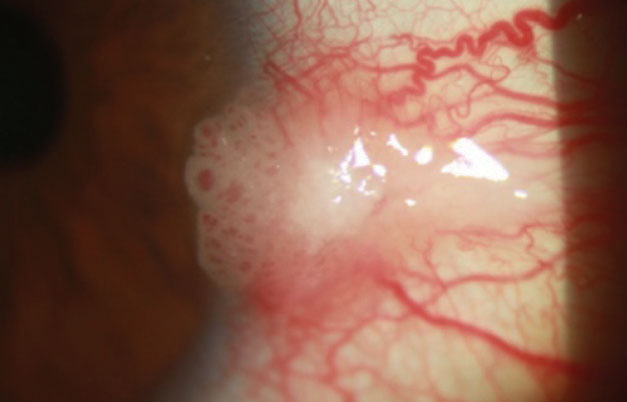 |
| This patient has a pink, raised, limbal lesion extending over the cornea with tortuous dilated feeder vessels and keratinized central surface plaque. |
Blepharitis and Demodicosis
Meibomian gland dysfunction (MGD) plays a major role in ocular surface pathology. Terminal duct obstruction and qualitative as well as quantitative changes in the glandular secretion result in altered tear film.
The spectrum and severity of the signs associated with blepharitis depends on the location and the degree of inflammation. In one form of anterior blepharitis, the skin of the eyelids, the base of the eyelashes, and the eyelash follicles are affected by Staphylococcus, leading to scaling, crusting and erythema of the eyelid margin with collarette formation at the base of the cilia, which can cause eyelash loss and corneal punctate epithelial erosions, marginal infiltrates and neovascularization. In the seborrheic type of anterior blepharitis, greasy, foamy scales called scurf surround the bases of the cilia. The patient may also present with signs of both types as well as with co-existing meibomianitis.11
In posterior blepharitis, the inflammation affects the meibomian glands and their orifices. Prominent blood vessels crossing the mucocutaneous junction, frothy discharge along the eyelid margin, pouting or plugging of meibomian orifices, expression of meibomian secretions that range from turbid fluid to thick, cheese-like material, thickening and scalloping of the eyelid margin, trichiasis and chalazia may all be observed.11
Ocular demodicosis, characterized by cylindrical dandruff around the root of the eyelashes, is often associated with blepharitis, chalazia and keratitis.12 Both Demodex follicularum and Demodex brevis can cause chronic and recurrent inflammation of the eyelid margin, as well as trichiasis, distichiasis and madarosis.12 While the larger D. follicularum mites congregate in the hair follicles, the smaller D. brevis mites reside in the sebaceous glands.12
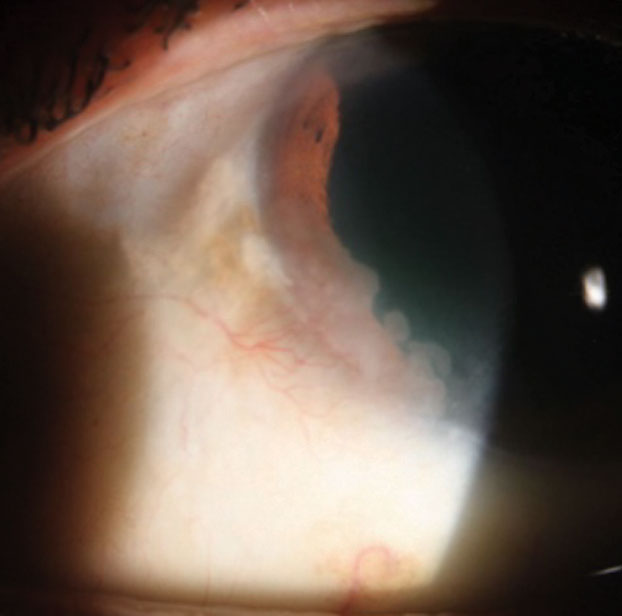 |
This patient portrays an opalescent cornea from 7 o’clock to 9 o’clock with feeder vessels. |
Dry Eye Disease
In 2017, the Tear Film & Ocular Surface Society’s International Dry Eye Workshop II set out to create an evidence-based definition and a contemporary classification system for dry eye disease.1 As a result, the following definition was accepted:
“Dry eye is a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.”1
Clinical signs include diffuse conjunctival hyperemia as well as corneal punctate epithelial erosions. Tissue staining with fluorescein, lissamine green or rose bengal are clinically beneficial in determination of the severity of ocular surface dryness.
Conjunctivochalasis
Conjunctival folds, also known as conjunctivochalasis (CCH), are more prevalent in the elderly, and only increase with age.13-15 Patients with CCH may experience epiphora or dry eye disease symptoms such as irritation, burning and foreign body sensation.16-18
Clinically, you should look for a delayed tear clearance and tear film instability. Due to anatomical obstruction of the punctum by the redundant conjunctival folds, destruction of the lacrimal lake and impediment of the tear flow by the conjunctival folds, the patient may experience delayed tear clearance.19-23 As the flow of tears is impeded, the conjunctival folds cause instability of the tear film, which can lead to ocular surface inflammation.18,19,24
The clinical suspicion of conjunctival folds is confirmed by biomicroscopic examination and use of vital dyes. While examining the anterior segment with the slit lamp, you may find redundant conjunctival folds over the inferior eyelid margin, which move with blinking. During your clinical exam, you can change the appearance of the folds based on how you manipulate the eyelids. If you press upward on the lower lid, you’ll notice worsening of the folds. Conversely, pulling the lid away from the globe while the patient looks up will cause the folds to disappear.18
Vital dyes can help the clinician further assess the redundant folds as well as the tear film. Fluorescein staining will show the classic precorneal tear film as well as punctate erosions. Additionally, it will show the folds along with the interrupted or decimated tear meniscus. Unlike dry eye disease where rose bengal and Llssamine green reveal staining in the exposure zone of the conjunctiva, these two dyes will stain non-exposure zones of the conjunctiva as well as detect punctate erosions over the redundant conjunctival folds.18
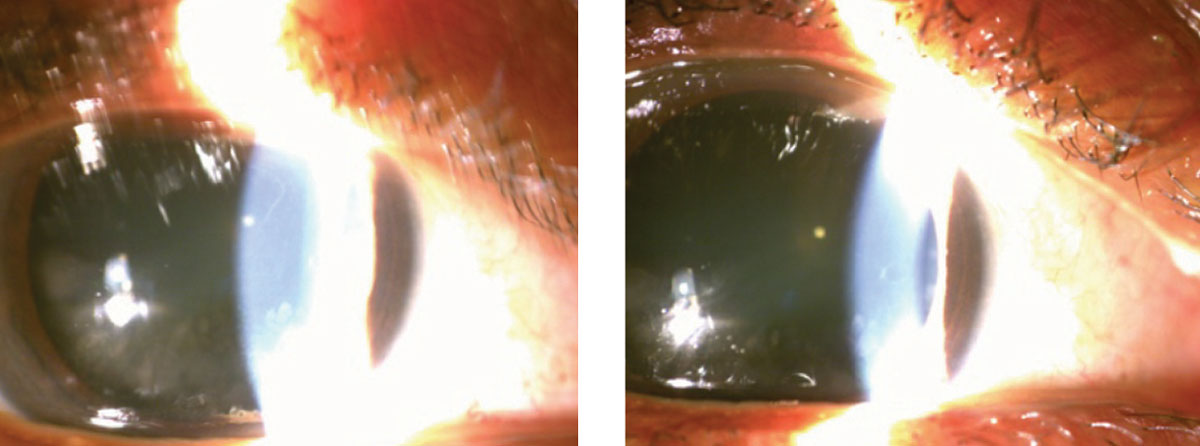
 |
This patient displays punctal stenosis in both the right (at left) and left eyes. |
Pinguecula, Pterygium and Squamous Neoplasia
Approaching the limbus while scanning the nasal and temporal bulbar conjunctiva within the palpebral fissure, the clinician will often observe a round, yellowish elevation. Pinguecula, which is more commonly found on the nasal aspect of the limbus, is a condition of abnormal differentiation characterized by squamous metaplasia with proliferation.25 While the condition is benign, its presence results in tear film instability that may lead to symptoms of irritation and signs of inflammation.
Another commonly encountered finding in the nasal limbal area, especially in patients exposed to UV radiation, is a pterygium.26 This wing-like hypertrophy of the subconjunctival connective tissue and overlying epithelium drags the conjunctival vessels as it crosses the limbus and approaches the visual axis. These lesions may be thin and flat or elevated and gelatinous. They may be quiet or inflamed. Stocker’s line, a punctate, brownish subepithelial iron line passing vertically in front of the invasive apex of the pterygium, may often be observed and is a sign of chronicity.27 Pterygia may induce astigmatism, decrease visual acuity as they obscure the visual axis, cause symptoms of irritation when inflamed, and be cosmetically displeasing to the patient.
It is important to examine limbal lesions closely to differentiate them from ocular surface squamous neoplasia (OSSN). A pink, gelatinous lesion along the limbus with tortuous, dilated feeder vessels and sometimes with keratinized plaques on its surface is likely to be a form of OSSN.28 These typically show abrupt onset and rapid progression may present as opalescence on the cornea or chronic conjunctivitis.28
 |
Here, a patient shows map-dot-fingerprint dystrophy. |
Ocular Surface Conditions
Disruption to the ocular surface has various etiologies. Because treatment of each etiology varies, it is imperative for the practitioner to carefully evaluate the ocular surface and the cause of damage to the surface before initiating treatment. Corneal epithelial defects are a frequent cause of ocular irritation, foreign body sensation, tearing, pain, redness and, quite possibly, photophobia. While performing biomicroscopy, the astute clinician may detect corneal irregularities beneath the epithelial layer. With the use of sodium fluorescein, the practitioner may notice negative staining on the cornea, which helps to delineate the corneal irregularities commonly associated with epithelial basement membrane dystrophy (EBMD), also known clinically as anterior basement membrane dystrophy or map-dot-fingerprint dystrophy.
It is not uncommon for the clinician to notice corneal irregularities during biomicroscopy or decreased visual acuity because of a compromised ocular surface. If you suspect EBMD, ask the patient if they have ever had symptoms of recurrent corneal erosion (RCE), because approximately 10% of EBMD patients experience symptoms of pain or decreased vision secondary to corneal surface irregularity.29,30 Ocular surface dryness can have a tremendous impact on patients with EBMD, as they are highly likely to rub their epithelial surface off if they rub their eye aggressively or have their epithelial surface adhere to their superior palpebral conjunctival when they open their eyes in the morning. Because EBMD patients are at increased risk of RCE due to their corneal irregularities, judicious use of artificial tears, lubricating ointment or 5% NaCl ointment at bedtime may help minimize the risk of erosion.
Dr. Demeritt is an assistant professor at Nova Southeastern University College of Optometry.
Dr. Lewandowska is an assistant professor at Nova Southeastern University College of Optometry.
1. Craig J, Nichols K, Akpek E, Et al. TFOS DEWS II Definition and Classification Report. Ocul Surf. 2017;15:276-283. 2. Hintschich C. Correction of entropion and ectropion. Dev Ophthalmol. 2008;41:85-102. 3. Damasceno R, Osaki M, Dantas P, Belfort R. Involutional entropion and ectropion of the lower eyelid: prevalence and associated risk factors in the elderly population. Ophthalmic Plast Reconstr Surg. 2011;27(5):317-20. 4. Choo P. Distichiasis, trichiasis, and entropion: advances in management. Int Ophthalmol Clin. 2002 Spring;42(2):75-87. 5. Seiff S, Shorr N, Adams T. Surgical treatment of punctal‐canalicular fibrosis from 5‐fluorouracil therapy. Cancer. 1985;56(8):2148-9. 6. Tabbara, K. F., & Bobb, A. A. (1980). Lacrimal system complications in trachoma. Ophthalmol. 1980;87(4):298-301. 7. Brink H, Beex L. Punctal and canalicular stenosis associated with systemic fluorouracil therapy. Documenta Ophthalmologica. 1995;90(1):1-6. 8. Kristan R. Treatment of lacrimal punctal stenosis with a one-snip canaliculotomy and temporary punctal plugs. Arch Ophthalmol. 1988;106(7):878-9. 9. Soiberman U, Kakizaki H, Selva D, Leibovitch I. Punctal stenosis: definition, diagnosis, and treatment. Clin ophthalmol. 2012;6:1011-8. 10. Ulusoy M, Kıvanç S, Atakan M, Akova-Budak B. How important is the etiology in the treatment of epiphora? Journal of Ophthalmology. www.hindawi.com/journals/joph/2016/1438376/. 11. American Academy of Ophthalmology (AAO) Cornea/External Disease Preferred Practice Pattern Panel. Preferred practice pattern guidelines. Blepharitis. www.aao.org/preferred-practice-pattern/blepharitis-ppp--2013. October 2013. Accessed October 24, 2018. 12. Luo X, Li J, Chen C, et al. Ocular demodicosis as a potential cause of ocular surface inflammation. Cornea. 2017;36:S9-S14. 13. Hashemi H, Rastad H, Emamian M, Fotouhi A. Conjunctivochalasis and related factors in an adult population of Iran. Eye & contact lens. 2018;44:S206-S209. 14. Watanabe A, Yokoi N, Kinoshita S, et al. Clinicopathologic study of conjunctivochalasis. Cornea. 2004;23(3):294-8. 15. Zhang X, Li Q, Zou H, et al. Assessing the severity of conjunctivochalasis in a senile population: a community-based epidemiology study in Shanghai, China. BMC Public Health. 2011;11(1):198. 16. Balci O. Clinical characteristics of patients with conjunctivochalasis. Clin. Ophthalmol. 2014;8:1655-60. 17. Chhadva P, Alexander A, McClellan A, et al. The impact of conjunctivochalasis on dry eye symptoms and signs. Invest Ophthalmol Vis Sci. 2015;56(5):2867-71. 18. Meller D, Tseng S. Conjunctivochalasis: literature review and possible pathophysiology. Survey of ophthalmology, 1998;43(3):225-32. 19. Di Pascuale M, Espana E, Kawakita T, Tseng S. Clinical characteristics of conjunctivochalasis with or without aqueous tear deficiency. Bri J Ophthalmol. 2004;88(3):388-92. 20. Erdogan-Poyraz C, Mocan M, Irkec M, Orhan M. Delayed tear clearance in patients with conjunctivochalasis is associated with punctal occlusion. Cornea. 2007;26(3):290-3. 21. Huang Y, Sheha H, Tseng S. Conjunctivochalasis interferes with tear flow from fornix to tear meniscus. Ophthalmol. 2013;120(8):1681-7. 22. Liu D. Conjunctivochalasis. A cause of tearing and its management. Ophthalmic plastic and reconstructive surgery. 1986;2(1):25-8. 23. Prabhasawat P, Tseng S. Frequent association of delayed tear clearance in ocular irritation. Bri J Ophthalmol. 1998;82(6):666-75. 24. Yokoi N, Komuro A, Nishii M, et al. Clinical impact of conjunctivochalasis on the ocular surface. Cornea. 2005;24(8):S24-S31. 25. Dong N, Li W, Lin H, et al. Abnormal epithelial differentiation and tear film alteration in pinguecula. Invest Ophthalmol Vis Sci. 2009;50(6):2710-5. 26. Moran D, Hollows F. Pterygium and ultraviolet radiation: a positive correlation. Bri J of Ophthalmol. 1984;68(5):343-6. 27. Hansen A, Norn M. Astigmatism and surface phenomena in pterygium. Acta ophthalmologica. 1980;58(2):174-81. 28. Cicinelli M, Marchese A, Bandello F, Modorati G. Clinical management of ocular surface squamous neoplasia: A review of the current evidence. Ophthalmol Ther. link.springer.com/article/10.1007/s40123-018-0140-z. July 20, 2018. Accessed October 24, 2018. 29. Waring III G, Rodrigues M, Laibson P. Corneal dystrophies. I. Dystrophies of the epithelium, Bowman’s layer and stroma. Surv ophthalmol. 1978;23(2):71-122. 30. Werblin T, Hirst L, Stark W, Maumenee I. Prevalence of map-dot-fingerprint changes in the cornea. Bri J Ophthalmol. 1981;65(6):401-9. |

