 |
A 57-year-old African American woman was referred for evaluation of suspicion of glaucoma secondary to retinal nerve fiber layer (RNFL) defect in the right eye. Her best-corrected visual acuity was 20/20 in both eyes without afferent pupillary defect. Her medical history was positive for HIV, for which her managing physician recently shifted medication from Symtuza (darunavir, cobicistat, emtricitabine and tenofovir alafenamide, Janssen Pharmaceuticals) tablets to Cabenuva (cabotegravir, rilpivirine extended-release injectable suspension, ViiV Healthcare).
Her viral load was undetectable, and her CD4 count was greater than 1000cells/mm3. The patient’s blood pressure was 130/85mm Hg in office. Intraocular pressures were 19mm Hg OD and 21mm Hg OS with central corneal thickness of 543µm OD and 542µm OS. She had split focal RNFL defects superior temporally in the right eye, while the optic discs were sharp and pink with healthy, symmetric neuroretinal rim tissue without focal rim loss.
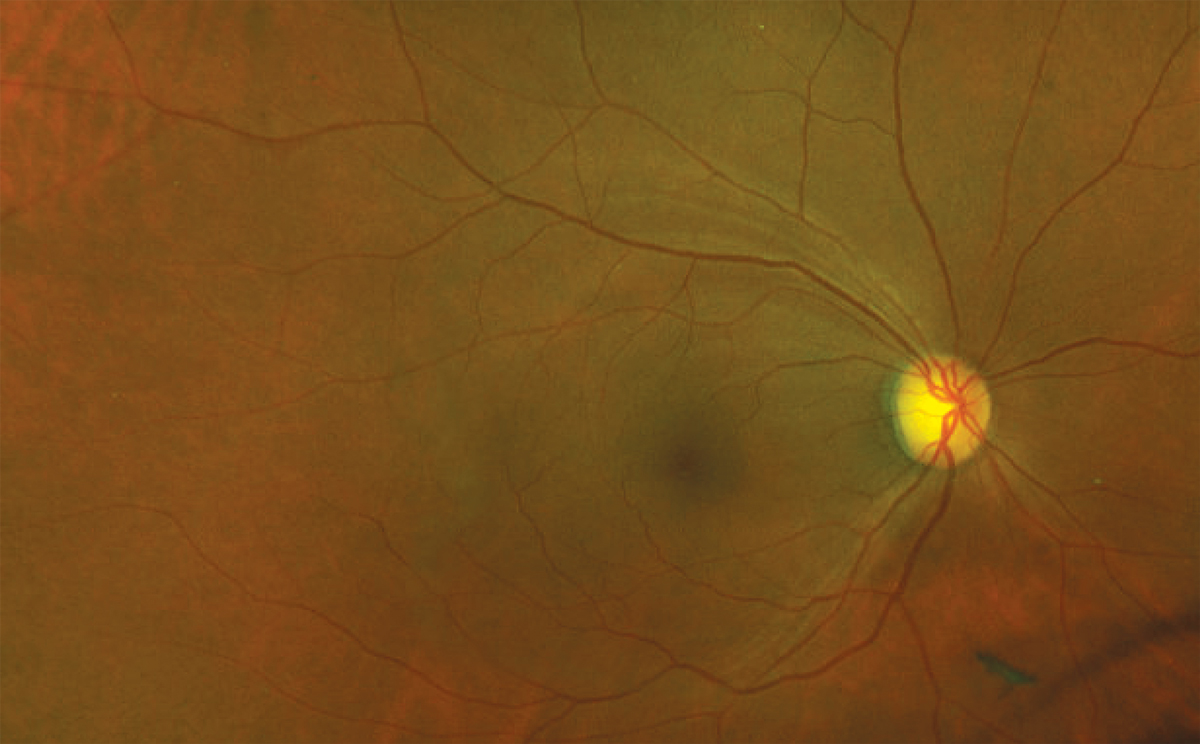 |
| Ultra-widefield image demonstrating superior temporal RNFL defect in the absence of neuroretinal rim notching. A posterior vitreous detachment is visible inferior to the disc. Click image to enlarge. |
RNFL and ganglion cell-inner plexiform layer (GCIPL) imaging fell within the normative range in each eye, as did neuroretinal rim thickness. Automated visual fields were full in each eye. Careful evaluation of the combined RNFL and GCIPL analysis of the right eye reflected the subtle RNFL defect visible clinically and in widefield imaging, which extended toward, without reaching, the optic disc.
RNFL Loss
Observation of a focal RNFL defect that is representative of axonal loss of retinal ganglion cells either clinically or by fundus photography is an important structural finding in glaucomatous optic neuropathy; however, these defects are not pathognomonic for glaucoma. In nonglaucomatous eyes, RNFL loss may develop as a result of underlying neurological, systemic or retinal disease, or as a result of certain systemic medications and treatments. This means that their presence requires careful ophthalmic examination and review of medical history, and may require further medical or neurologic investigation.1-6
In the case of the incidental finding of focal RNFL defect in a nonglaucomatous, otherwise healthy eye, the most common cause is a past microvascular anomaly, or resulting axonal loss following a focal ischemic event leading to disruption of axoplasmic transport: a resolved cotton wool spot. These entities are common ocular manifestations of diabetes mellitus and hypertension—and may also occur in the context of HIV.1,2,4-6 Following resolution of a cotton wool spot, an RNFL defect may be apparent due to the local disruption to axonal transport, resulting in axonal death, and a corresponding subtle, nonprogressive visual field defect or microperimetry abnormality may also be detectable.1,5
HIV Retinopathy
This condition is classified as either infectious, which includes cytomegalovirus retinitis, herpetic necrotizing retinitis and other opportunistic infections, or non-infectious. Non-infectious HIV retinopathy, or HIV microangiopathy, is evidenced through the presence of cotton wool spots or, following their resolution, focal RNFL defects. It is estimated to occur in up to 60% of untreated HIV-positive patients.6 OCT angiography has also been demonstrated to be useful in detecting subclinical microvascular abnormality in individuals with HIV who undergo antiretroviral therapy (ART) in the absence of infectious retinitis.7
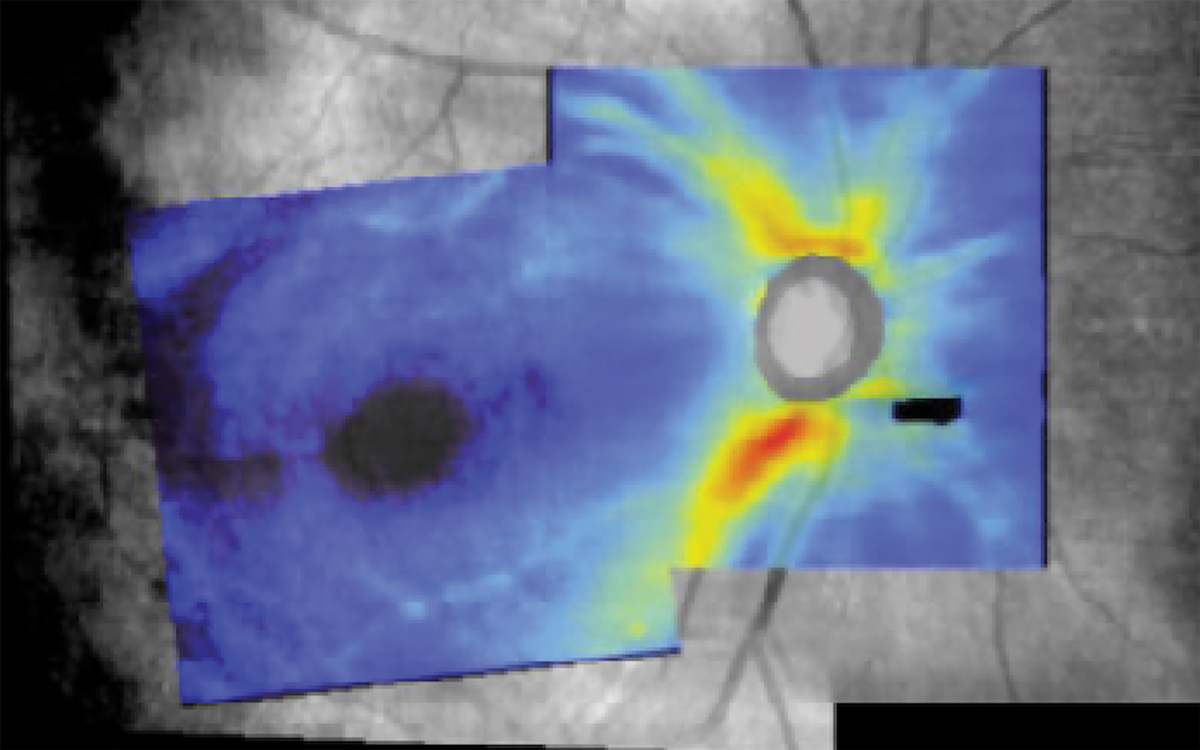 |
|
Combined RNFL and ganglion cell analysis map of the right eye demonstrating subtle superior temporal RNFL loss not extending to the optic disc. The area of missing data inferior nasal to the optic disc correlates to the posterior vitreous detachment. Click image to enlarge. |
HIV Treatment
Advancements in HIV prevention, treatment and cure research have continued at a rapid pace since the initial approval of the first ART in 1996. Currently, antiretroviral therapy is recommended to be initiated as soon as possible following HIV diagnosis with the goal of reducing plasma HIV RNA to an undetectable level, ultimately reducing disease-related morbidity and mortality as well as preventing viral transmission.8 Initial therapy generally includes three oral medications from two or more HIV drug classes.8
Real-world challenges related to ongoing, long-term viral suppression include the requirement of excellent adherence to often complex oral medication regimens, adverse effects related to therapy and access to treatment. Therapeutic and prevention strategies for individuals diagnosed with or at risk of developing HIV are central to long-term public health strategies to reduce new infections.8
Recently approved HIV therapies aim to improve adherence to therapy through longer acting agents and to address multidrug resistance. Cabenuva, approved in 2021, is indicated for individuals who have achieved viral suppression on their current ART as a replacement for oral therapy and is administered subcutaneously every one to two months in a physician’s office. Sunleca (lenacapavir, Gilead Sciences), approved in 2022, is used in addition to other antiretroviral therapies in patients who have demonstrated multidrug resistance.
Following initial treatment, which includes oral and injectable formulations of lenacapavir over either a two- or 15-day pre-specified course, long-term maintenance dosing calls for subcutaneous injection every six months.8 For individuals at increased risk of HIV infection, pre-exposure prophylaxis (PrEP) can reduce the risk of HIV infection by up to 99%. Currently in the United States, there are two oral medications and one long-acting injectable medication approved for PrEP.9
Diagnosis
After a thorough review of systems without positive findings, we discussed the patient’s medical history in more detail. The patient had access to her recent complete metabolic panel and complete blood count (with platelets), which were negative for evidence of underlying infection, inflammation, hematologic or metabolic concern. She reported no history of positive COVID-19 test and, despite her elevated systolic blood pressure in office, she reported typical greater concern for low blood pressure by her managing physician.
She shared that her symptoms that led her to seek medical care where she was ultimately diagnosed with HIV eight years prior were very severe: rapid, significant weight loss, lethargy and flu-like symptoms. At the time of diagnosis, she recalled that her CD4 count was “very low.”
The patient described multiple therapeutic challenges since diagnosis including adverse effects of nausea, weight gain, lethargy and brain fog until the change to her current long-term injectable therapeutic strategy. Now, for the first time since being diagnosed with HIV, she reported having the energy and ability to think clearly that she recalled from prior to her HIV diagnosis. Clinical stability of the RNFL defects with stable RNFL and GCIPL imaging has been demonstrated over a 12-month period without intervention.
In the absence of optic neuropathy, nonprogressive RNFL defects may indicate previous presence of a cotton wool spot or microvascular insult related to underlying treatable systemic conditions including HIV.
Dr. Steen is an associate professor at Nova Southeastern University College of Optometry where she serves as director of the Glaucoma Service, coordinator of the Primary Care with Emphasis in Ocular Disease Residency and teaches courses in glaucoma and ocular pharmacology. Her financial disclosures include Bausch & Lomb, Santen, Ocuphire and Carl Zeiss Meditec.
1. Ioannides A, Georgakarakos ND, Elaroud I, et al. Isolated cotton-wool spots of unknown etiology: management and sequential spectral domain optical coherence tomography documentation. Clin Ophthalmol. 2011;5:1431-3. 2. Brown GC, Brown MM, Hiller T, et al. Cotton-wool spots. Retina. 1985;5(4):206-14. 3. Marinho PM, Marcos AAA, Romano AC, et al. Retinal findings in patients with COVID-19. Lancet. 2020;395:1610. 4. Freeman WR, McCutchan AJ, Arevalo JF, et al. HNRC Group. The relationship between AIDS retinal cotton wool spots and neuropsychological impairment in HIV-positive individuals in the pre-highly active antiretroviral therapy era. Ocul Immunol Inflamm. 2004;12(1):25-33. 5. Shin, JY, Lee, J, Lee, CJ et al. Association between localised retinal nerve fiber layer defects and cardiovascular risk factors. Sci Rep. 2019;9(1):19340. 6. Abu EK, Abokyi S, Obiri-Yeboah D, et al. Retinal microvasculopathy is common in HIV/AIDS patients: a cross-sectional study at the cape coast teaching hospital, Ghana. J Ophthalmol. 2016;20:8614095. 7. Du KF, Huang XJ, Chen C, et al. Macular changes observed on optical coherence tomography angiography in patients infected with human immunodeficiency virus without infectious retinopathy. Front Med (Lausanne). 2022;9:820370. 8. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in adults and adolescents with HIV. Department of Health and Human Services. 2023. clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-arv. Accessed December 20, 2023. 9. US Public Health Service. Preexposure prophylaxis for the prevention of HIV infection in the United States-2021 Update. A clinical practice guideline. 2021. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf. Accessed December 20, 2023. |

