Early detection and management of diabetic retinopathy (DR) is crucial to avoid complications such as vitreous hemorrhage and tractional retinal detachment secondary to neovascularization. Knowing the topography of new vessel growth may help in early detection of proliferative diabetic retinopathy (PDR), a research team from India speculated, investigating this premise in a new study.
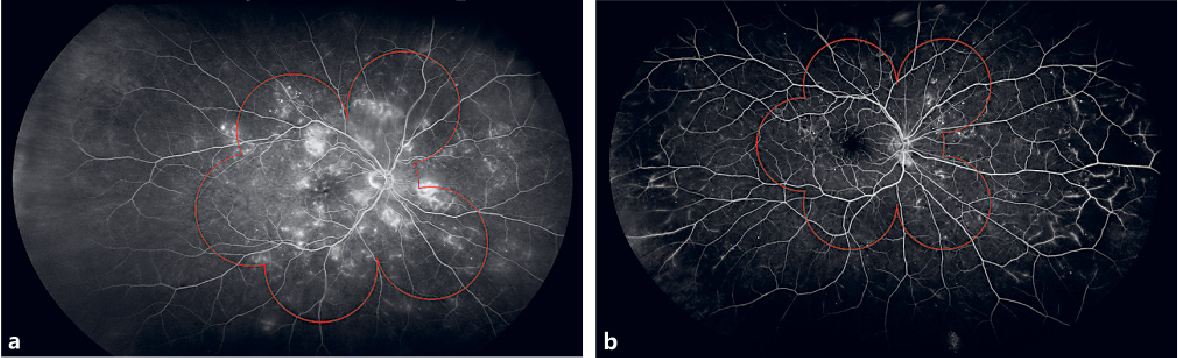 |
| Two comparisons of seven standard field (7SF) fundus imaging (the red overlay) with ultra-wide field fluorescein angiography: (a) right eye of a 48-year-old female patient with moderate DR in which of the pathology is detected with both techniques; (b) right eye of a 64-year-old female patient affected by mild diabetic retinopathy. Microaneurysms are uniformly distributed between 7SF and UWFFA but retinal ischemia and intra-retinal microvascular abnormalities are located outside 7SF boundaries. Photo: Jay Haynie, OD. Click image to enlarge. |
A total of 253 eyes of 187 patients with treatment‑naïve PDR without any fibrovascular proliferation underwent ultra-widefield fluorescein angiography (UWFFA). The number of lesions picked by imaging was compared with fundus imaging (the “seven standard field” montage). The main outcome measure was characteristics of neovascularization, such as the number, location and area of capillary nonperfusion (CNP), measured using angiography, which was considered with 95% confidence intervals.
UWFFA was capable of capturing 24% more “neovascularization elsewhere (NVE)” findings and 37% more nonperfused areas than the fundus images, suggesting that it is a preferred modality for assessment of PDR. The results also showed that such lesions are distributed asymmetrically throughout the retina and are not restricted to the posterior pole.
“The finding in our study that CNP areas are more in the nasal hemisphere and the number of NVEs are more in the temporal hemisphere could mean that vascular endothelial growth factor produced somewhere else in the fundus can lead to neovascularization at some other site,” the authors explained in their study.
The majority of eyes (60%) showed multiple NVEs, which might indicate that neovascularization starts simultaneously at different locations in an ischemic retina. There was a nasotemporal asymmetry in the distribution, the researchers notes, as 55% of lesions were located in the temporal and 45% in the nasal hemisphere.
“The possible hypothesis for this could be because of differential vitreous degeneration and retinal thickness in the temporal and nasal hemispheres,” the authors speculated. “The distribution of NVE in the superior and inferior hemispheres was 53% and 47%, respectively,” which was similar to other reports, citing a study that found 54% of lesions in the superior hemisphere and 46% in the inferior hemisphere.
A higher prevalence of disc neovascularization was reported compared to previous studies, possibly because these patients presented for routine clinical care and may be more likely to have an advanced stage of PDR, the authors noted. “It is our contention that the difference in RNFL thickness and vessel caliber in different quadrants of the retina and differential blood flow makes some vessels more prone to development of NVE,” they added.
The team concluded that topographic distribution of NVE reveals the preferential sites of new vessel formation. “This is important in early detection of neovascularization and its early treatment, particularly in the very early stages of PDR,” the authors surmised.
Nidhi V, Verma S, Shaikh N, et al. Topographic distribution of retinal neovascularization in proliferative diabetic retinopathy using ultra-wide field angiography. August 1, 2023. [Epub ahead of print.] |

