 History
History
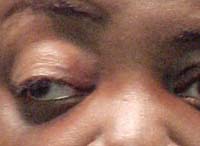
A 54-year-old black male presented emergently to the office with an acute headache and diplopia that had persisted for two days. He suggested that the headache featured significant pain around the temple area of his right eye. While ibuprofen seemed to help with the acute head pain, the headache always returned when the medication wore off.
He reported no history of trauma, and his ocular history was noncontributory. His systemic history was significant for a recent diagnosis of type 2 diabetes mellitus. The patient reported good compliance with metformin use. He reported no allergies of any kind.
Diagnostic Data
His best-corrected visual acuity was 20/25 OD and 20/20 OS via habitual -0.50D spehere/+2.50 bifocals. External examination revealed no evidence of afferent pupillary defect OU. Biomicroscopy uncovered normal and healthy anterior segment structures with open angles OU.
Intraocular pressure measured 16mm Hg OU. Dilated funduscopic examination uncovered quiet grounds, normal posterior poles and distinct optic nerves without evidence of disc edema OU. His cup-to-disc ratio was 0.2 x 0.2 OU.
Your Diagnosis
How would you approach this case? Does the patient require any additional testing? What is your diagnosis? How would you manage this patient? What is the likely prognosis? To find out the patient’s diagnosis, visit Review of Optometry Online,
www.revoptom.com. Click on the “Current Issue” link or on the cover icon, and then click “Diagnostic Quiz” under this month’s table of contents.
Discussion
All patients who present with diplopia should be asked the same battery of questions:
1. Does the double vision disappear when one eye is covered?
2. Is the double vision “up and down” or “side by side?”
3. Is the diplopia greater at distance or near?
4. Is the double vision variable?
5. How long has this been happening?
6. Is there associated head pain or pain upon eye movement?
The diagnosis in this month’s issue is ischemic vascular cranial nerve palsy.
The oculomotor nerve originates in the midbrain. The third nerve passes forward through the tegmentum, red nucleus and substantia nigra, and emerges medially to the cerebral peductal.1,2 It continues through the subarachnoid space and passes between the posterior cerebral and superior cerebellar arteries as it courses forward and parallel to the posterior communicating artery.1,2 The nerve takes a course in close proximity to the tentorium cerebelli. It then enters the middle cranial fossa by penetrating the dura mater both in front of and lateral to the posterior clinoid process, and runs along the lateral wall of the cavernous sinus above the trochlear nerve.1,2
The oculomotor nerve divides into a superior and inferior division as it enters the orbit through the superior orbital fissure. The superior division travels to innervate the superior rectus muscle and the levator palpebrae superioris muscle. The inferior division of cranial nerve (CN) III is divided into three branches, supplying the medial rectus, inferior rectus and inferior oblique. The nerve that supplies the inferior oblique muscle passes through the ciliary ganglion and contains parasympathetic fibers. These will synapse in the ciliary ganglion, which gives rise to postganglionic fibers that travel along the short ciliary nerves to innervate the papillary sphincter for pupillary constriction.1,2
Paresis of CN III can result from any insult along its course from the midbrain to the orbit.3,4 Nuclear lesions are rare. At the level of the midbrain in the brainstem, damage to the oculomotor fascicles causes incomplete paralysis due to the anatomic separation between the fasciles.4 Fascicular CN III palsy usually occurs secondary to ischemic conditions or metastatic lesions.3,4 Here, because of the proximity of adjacent structures, syndromic presentations are common.
Benedikt’s syndrome involves a lesion of the red nucleous and substantia nigra.4 Its symptoms include ipsilateral CN III palsy and contralateral hemichorea, hemiballismus and loss of sensation.4 Nothnagel’s syndrome effects CN III fascicles as they exit the cerebellar peduncle, causing ipsilateral CN III palsy, dysmetria and contralateral ataxia.4
Claude’s syndrome is a combination of Benedikt’s and Nothnagel’s syndromes. Webber’s syndrome involves the ipsilateral CN III fascicles and the descending pyramidal tract, and causes ipsilateral CN III palsy and contralateral hemiparesis.4
In adults over 50 years-of-age, where a systemic diagnosis of hypertension or diabetes exists, pupil-sparing CN III palsy is assumed to be secondary to ischemic vascular (vasculopathic) etiology—until proven otherwise.5,6
The clinical sign of “pupil sparing” is considered the hallmark finding in ischemic vascular CN III palsy.5,6 Ischemic vascular lesions are a result of microvascular disease, where the vascular architecture of the vasa nervorum––which supplies nourishment to the deeper axons of the third nerve that are bound for the recti and levator muscles––are damaged. The resulting lost function results in an eye that assumes a posture that is down and out with a ptotic lid.5
Because the superficial axons that subserve pupillary constriction are fed by another blood supply, they are left unaffected, preserving the pupilomotor response to light.5 Such findings are predisposed in patients with poorly controlled diabetes, hypertension, anemia and/or increased cholesterol levels.6 In isolation, this nonsurgical third cranial nerve palsy tends to resolve uneventfully within three to six months.5 If the palsy fails to resolve within this timeframe, the MRI––along with ancillary imaging––should be repeated to rule out other etiologies.5,6
In the subarachnoid space, CN III is vulnerable to compressive insult from trauma or uncal herniation (and rarely microvascular disease and infections). The pupil is usually involved. Acute subarachnoid bleeding typically causes severe headache and reduced consciousness.7,8
The posterior communicating artery, located at the junction of the carotid artery within the Circle of Willis, is the most frequent location of aneurysms that induce painful CN III palsy.7,8 Typically, aneurysms and subarachnoid hemorrhages cause pupil-involved third nerve palsy secondary to external compressive and expansive effects that impinge on CN III axons.7,8
In the intracavernous space, CN III travels in close proximity with CN IV, CN VI, and the ophthalmic and maxillary divisions of CN V, as well as sympathetic fibers. Lesions in this region usually result in multiple-nerve involvement. Intracavernous sinus lesions may result from cavernous sinus fistula, pituitary apoplexy, aneurysm, tumors, inflammation or infection.7,8
Superior orbital fissure syndrome presents similarly to cavernous sinus syndrome––except exophthalmos is less likely to occur, and CN II and pupil-involved CN III palsies are more common.7,8
Lesions located in the orbit usually are accompanied by signs of inflammation, exophthalmos and/or mechanical restriction. Common etiologies include trauma, neoplasm and infection. These lesions must be differentiated from muscle-affecting diseases, such as thyroid-related ophthalmopathy, ocular myasthenia gravis and ocular myopathy.7,8
The treatment of CN III palsy depends on the underlying cause. Tumors, for example, will require resection or irradiation. Strokes demand ischemic vascular therapy.
Currently, there are two treatment options for aneurysms: surgical clipping or endovascular coiling (EVC).9,10 Surgical clipping is the optimal choice for inducing aneurysm base closure. However, when it is not surgically possible to reach the lesion, EVC is required.
The EVC procedure is accomplished via passing a platinum catheter coil through the vascular system into the aneurysm. The coiling technique within the enlarged anomaly’s lumens initiates blood clotting through a thrombotic reaction, which stabilizes the malformation and prevents it from rupturing. 9,10 The risks associated with surgical clipping and endovascular coiling, such as stroke or death, are the same. Mortality rates are as low as 1% to 5 %.9,10
Vasculopathic CN III palsy often resolves spontaneously within three months. But, if it doesn’t, further workup should include MRI, a vasculitis workup and CSF examination to rule out infectious diseases or overproduction anomalies (e.g., pseudotumor cerebri, hydrocephalus, aqueductal stenosis, meningitis). In any CN III palsy involving the pupil, an MRI and MRA are emergently indicated to rule out the presence of emerging aneurysm.9,10
As for our patient, we recorded and documented his extraocular muscle motilities in primary gaze and the four cardinal positions of gaze (up, down, left and right). We compared versions and ductions, which were found to be equivalent. After a review of the patient’s history, we determined that the forced duction test was unnecessary.
Next, we made a stat neuro-ophthalmologic referral to rule out infection, inflammation, space-occupying lesion and aneurysm. We recommended a comprehensive blood panel, including a complete blood count with differential and platelets, lipid panel, angiotensin converting enzyme, fluorescent treponemal antibody absorption test, reactive plasma regain, westergren sedimentation rate and c-reactive protein, as well as emergent neuroimaging via MRI and MRA to rule out aneurysm and mass.
We confirmed the diagnosis of exclusion when the balance of the neurologic work-up returned negative. We referred the patient back to his primary care team, and suggested that his diabetic regimen should be evaluated and adjusted. The condition resolved after six months of follow-up by both our staff and the neuro-ophthalmology department.
Thanks to Candice Tolud, OD, of Moorestown, N.J., Krutti Shah, OD, of Sparta, N.J., Ahn Ty Nugyen, OD, of Tampa, Fla., and Mashel Nehema, OD, Philadelphia, Pa., for their contributions to this case.
1. Snell RS, Lemp MA. Cranial Nerves, Part I. Those Nerves Directly Associated with the Eye and Orbit. In: Snell RS, Lemp MA (eds.). Clinical Anatomy of the Eye, 2nd ed. Philadelphia: Blackwell; 1998:294-328.
2. Moore KL, Dalley AF, Agur AM. Clinically Oriented Anatomy, 6th ed. Philadelphia: Lippincott Williams & Wilkins; 2010:902-7.
3. Shechtman DL, Woods AD, Tyler JA. Pupil sparing incomplete third nerve palsy secondary to a cavernous sinus meningioma: challenges in management. Clin Exp Optom. 2007 Mar;90(2):132-8.
4. Herzau V. Infranuclear Disorders of ocular motility. In: Schiefer U, Wilhelm H, Hart W (eds.). Clinical Neuro-Ophthalmology: A Practical Guide. New York: Springer; 2007:137-54.
5. Yanovitch T, Buckley E. Diagnosis and management of third nerve palsy. Curr Opin Ophthalmol. 2007 Sep;18(5):373-8.
6. Jacobson DM, McCanna TD, Layde PM. Risk factors for ischemic ocular motor nerve palsies. Arch Ophthalmol. Jul 1994;112(7):961-6.
7. Rai S, Rattan V. Traumatic superior orbital fissure syndrome: Review of literature and report of three cases. Natl J Maxillofac Surg. 2012;3(2):222-5.
8. Evans HH, Wurth BA, Penna KJ. Superior orbital fissure syndrome: a case report. Craniomaxillofac Trauma Reconstr. 2012;5(2):115-20.
9. King JT Jr, Berlin JA, Flamm ES. Morbidity and mortality from elective surgery for asymptomatic, unruptured, intracranial aneurysms. J Neurosurg. 1994 Dec;81(6):837-42.
10. Yanoff M, Duker J. Ophthalmology, 2nd ed. Philadelphia: Mosby; 2004:1324-34.

