 |
Keratoconus (KC) is a chronic, clinically non-inflammatory, bilateral ectasia of the cornea characterized by progressive corneal steepening and apical thinning leading to irregular astigmatism, higher-order aberrations and visual distortion.1 KC classically manifests during puberty and can progress at an unpredictable rate until the third to fourth decade of life. The prevalence varies geographically but is estimated to be 1.38 per 1000 in the world’s population, with a rate of 0.15% in the United States as of 2016.2,3
Other corneal ectasias include pellucid marginal degeneration, keratoglobus and post-refractive ectasia. If left untreated, corneal ectasia can lead to vision loss and an increased probability of corneal transplant. Given the significant risk of affecting social and educational development during young adulthood, the crux of corneal ectasia management lies in halting disease progression with corneal crosslinking (CXL) at its earliest stages to reduce the necessity for invasive corneal surgeries and prevent a lifetime of vision loss.4
CXL was first introduced over 20 years ago by researchers at the University of Dresden as a novel approach to strengthen corneal tissue by more than 300%.5,6 Adopted early and widely used abroad, the FDA approved CXL for progressive KC and corneal ectasia following refractive surgery in 2016. The technique involves the combination of topical riboflavin (vitamin B2) and ultraviolet-A light (UVA) to initiate a chain of chemical reactions that release oxygen free radicals, which generate strong covalent bonds within the stromal collagen, thereby providing structural reinforcement and arresting progression of the ectatic process.
The US Phase III clinical trial demonstrated 1.6D flattening in maximum keratometry after one year.7 Since the approval of CXL, multiple studies have confirmed its efficacy in achieving long-term stabilization of KC in addition to reducing the socioeconomic burden of the disease.8,9 Corneal ectasia was once the most common indication for penetrating keratoplasty; however, with the widespread adoption of CXL and newer contact lens innovations, there has been a decline over the last 10 years, and it has fallen to number six on the list.10 The modern paradigm of corneal ectasia management involves timely intervention with CXL to prevent progression, improving vision and closely monitoring for changes.
Indications and Contraindications
CXL is indicated for progressive keratoconus and post-refractive ectasia. The definition of progression is at the discretion of the physician and usually involves topometric, pachymetric and refractive changes.11 Insurance carriers often require documentation of these specific criteria within 24 months:12
 |
|
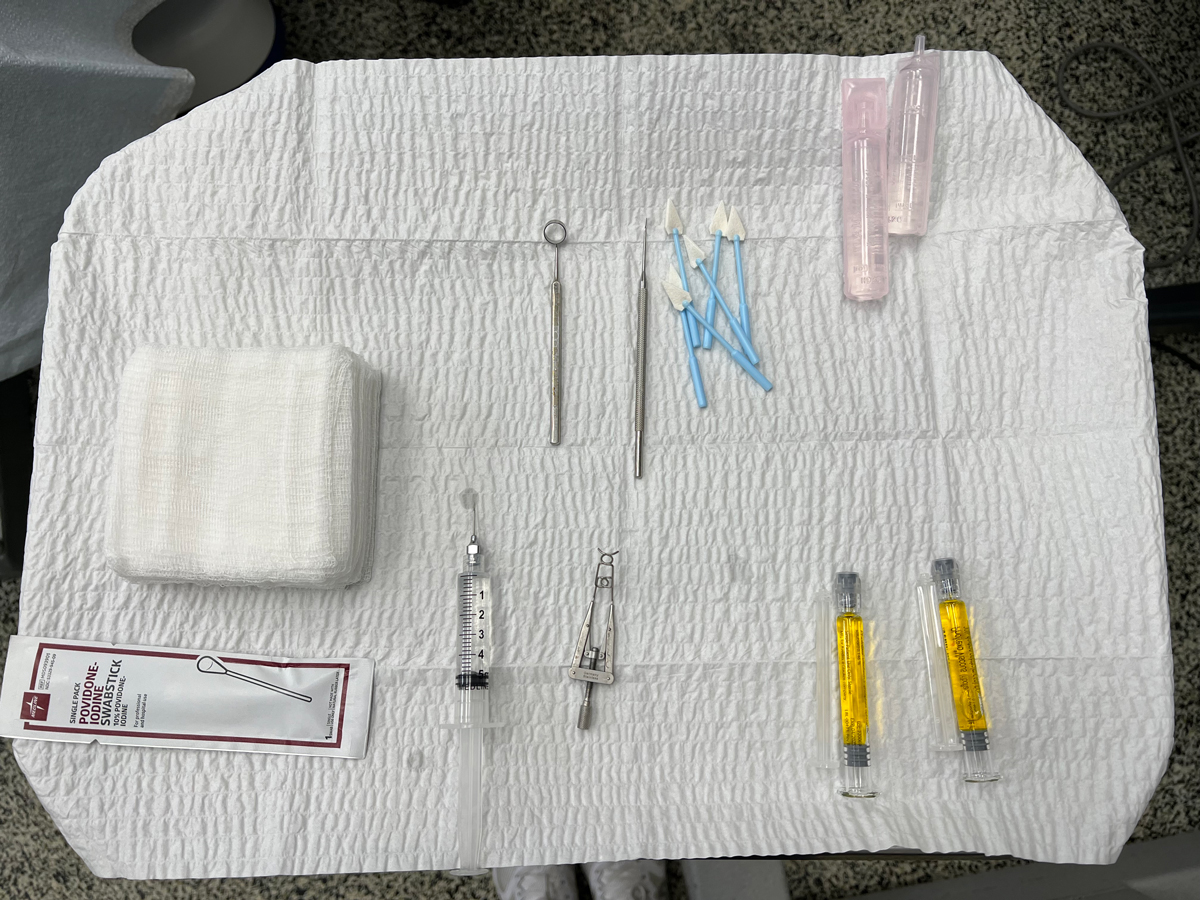
Fig. 1. CXL instrumentation set up on a Mayo stand. Click image to enlarge. |
- >1D change in the steepest keratometry value (Kmax)
- >1D change in cylinder value in subjective refraction
- >0.1mm back optic zone radius change in gas permeable contact lens parameters
While there is no specific age range in the indication statement for CXL, the FDA-approval clinical trial included patients aged 14 to 65.7 Nevertheless, for the vulnerable, faster-progressing younger individuals, closer follow-up and a lower threshold for CXL should be adopted for patients under 17 years old and steeper than 55D Kmax.13
CXL is contraindicated in pregnant and nursing mothers as the safety has not been evaluated. Corneal stromal thickness of less than 400µm is contraindicated to prevent endothelial cell damage; however, hypo-osmolar riboflavin solution can be used to swell the cornea at the time of the procedure. Another proposed alternative is placing a riboflavin-soaked soft contact lens on the eye during UVA irradiation.
UV light exposure may reactivate herpes simplex keratitis, so caution should be taken for patients with a history of herpetic ocular disease, which may include antiviral prophylaxis.8
Informed Consent Considerations
A written description of the crosslinking procedure in lay language, its indications, alternative treatments, postoperative expectations and possible adverse events should be read and signed by the patient, clinician and a witness.14 It must be stressed that the primary purpose of CXL is to stop progression of corneal ectasia, not use as a refractive procedure. However, there are additional studies demonstrating CXL treatment was able to decrease the steepness of the cone, consequently improving both uncorrected and corrected visual acuity.15
 |
|
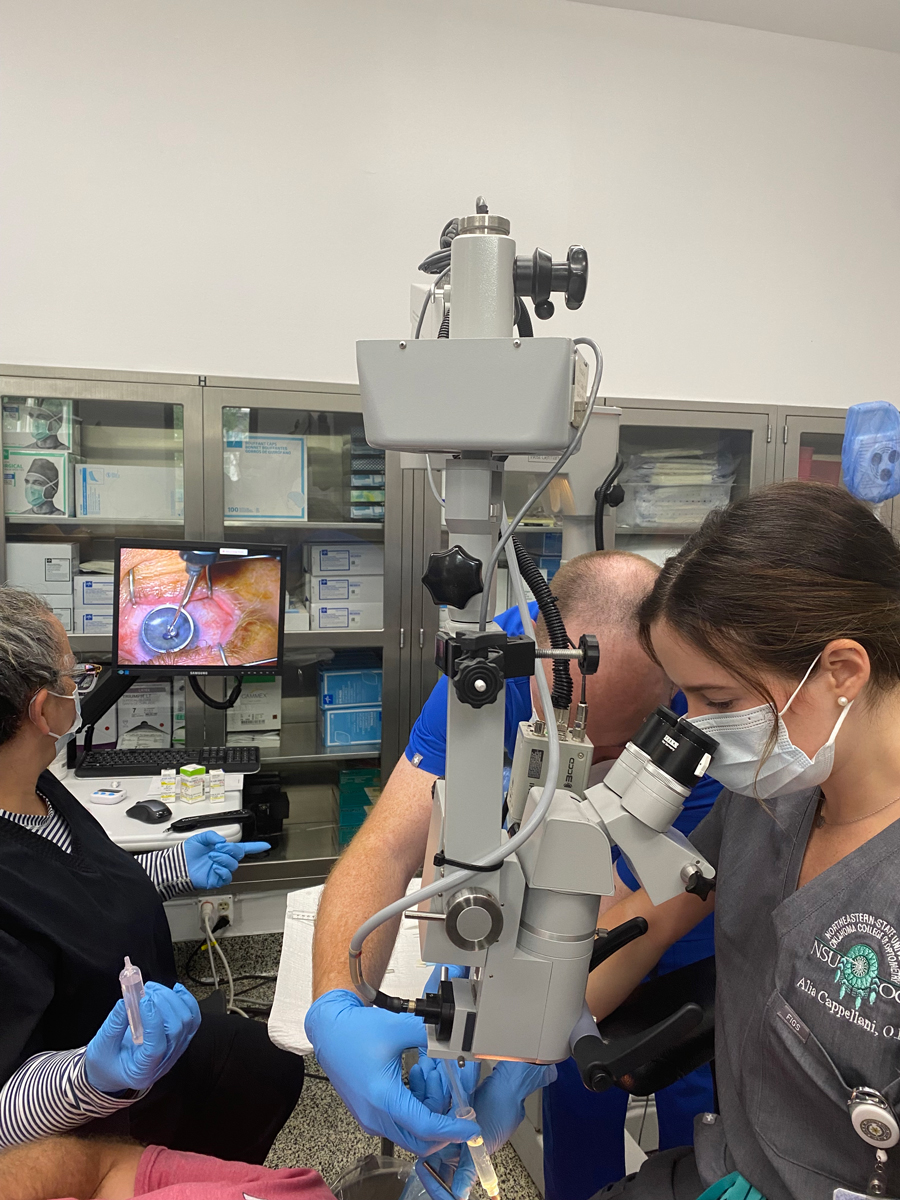
Fig. 2. Instillation of a diluted alcohol solution into the well to soften up the epithelium prior to debridement. Click image to enlarge. |
Procedure
Currently, the standard epithelium-off Dresden protocol is the only FDA-approved CXL method (iLink, Glaukos). It entails a 60-minute monocular treatment as follows:7
1. Positioning and alignment. The patient is positioned in a supine position with the surgical microscope focused.
2. Pre-op lid sterilization. The eyelids and periorbital area should be thoroughly cleaned with povidone-iodine. Topical anesthetic eye drops are instilled three to four times over five-minute intervals in the surgical eye to aid in softening the epithelium, and one drop in the other eye for blink control. An eyelid speculum is placed so the lids and lashes will not interfere with surgery. An instrument tray showing the proper instrumentation needed for the CXL procedure is shown in Figure 1.
3. Epithelial removal. A well is placed onto the cornea, centered around the cone and a diluted ethanol solution is dripped in the well for 30 seconds to loosen the epithelium prior to debridement (Figure 2). A dry surgical sponge is used to soak up the ethanol, the well is removed and the eye is rinsed with a balanced salt solution. The epithelium is removed using a surgical sponge and spatula to clear the circular 7mm to 9mm treatment zone.
4. Riboflavin baste. One drop of Photrexa Viscous (riboflavin 5’-phosphate in 20% dextran ophthalmic solution) is instilled topically every two minutes for 30 minutes. The eye is examined with a slit lamp to confirm the presence of yellow flare in the anterior chamber.
If flare is not present, additional Photrexa Viscous should be instilled at a rate of one drop every two minutes for an additional two to three drops until there is yellow flare seen in the anterior chamber.
5. Pachymetry check.
Ultrasound pachymetry is performed to ensure the corneal thickness is a minimum of 400µm. If corneal thickness is less than 400µm, two drops of Photrexa (riboflavin 5’-phosphate ophthalmic solution) should be instilled every five to 10 seconds until 400µm is achieved.
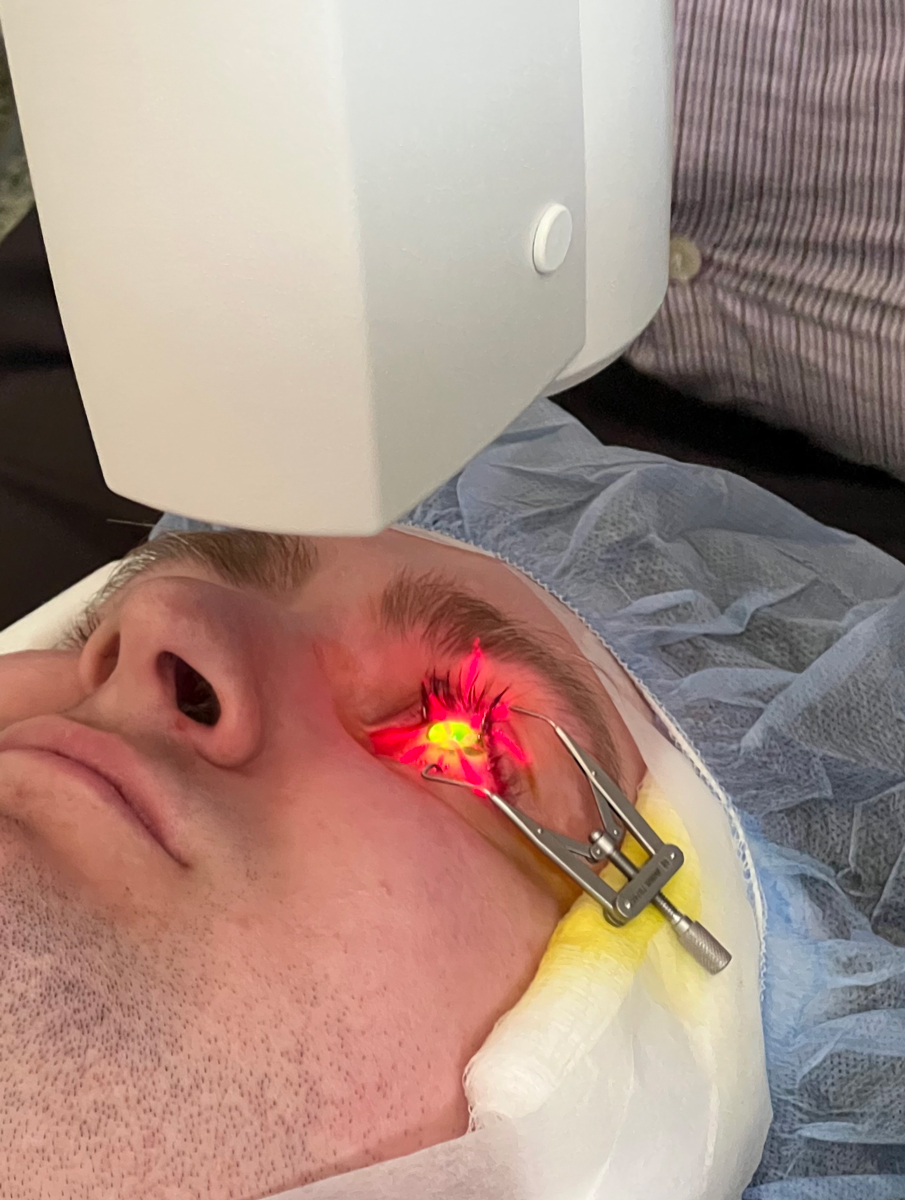
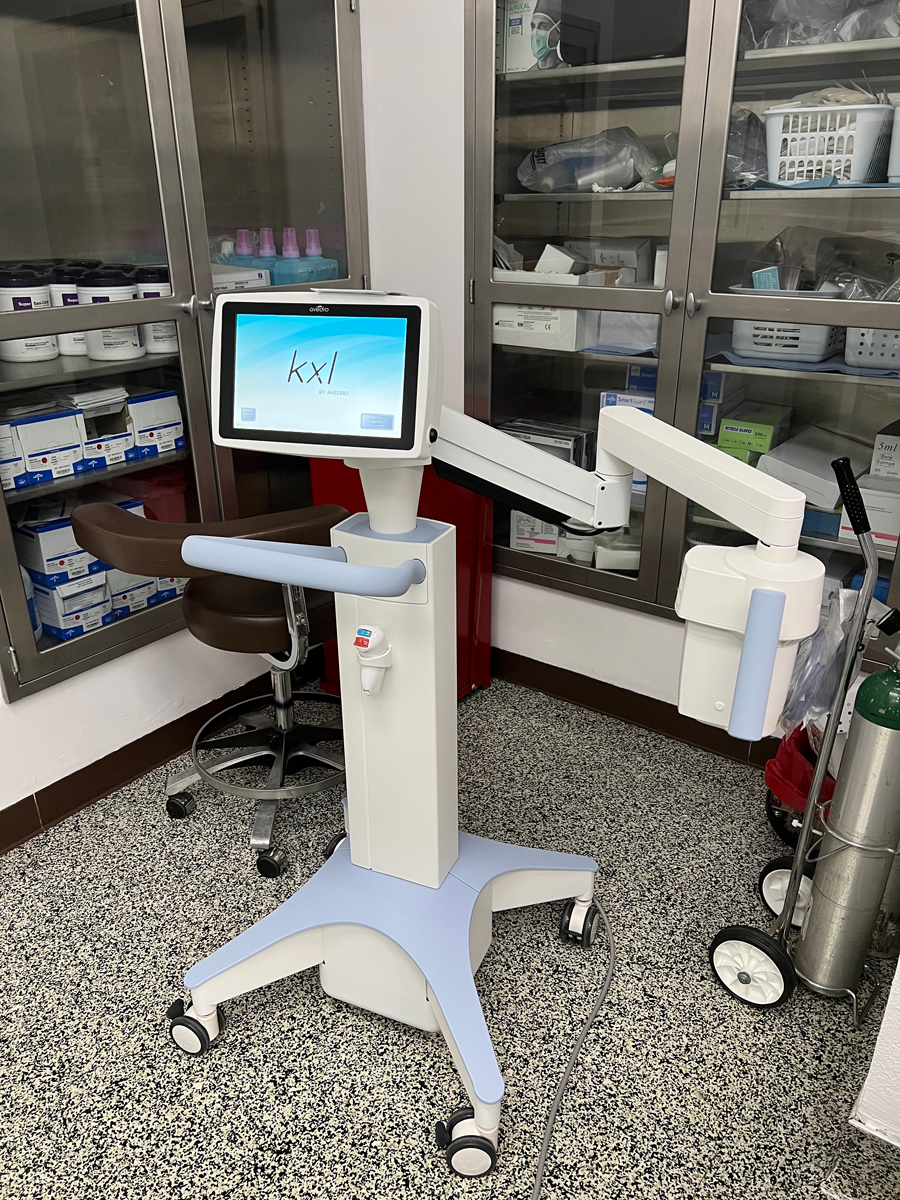
6. UV-A irradiation. Once the yellow flare is visualized and the 400µm threshold thickness is met, corneal irradiation is performed for 30 minutes (Figure 3). The KXL system delivers a metered dose of 365nm UV-A light, 3mW/cm2 exposure centered over the cornea (Figure 4). The instillation of Photrexa Viscous is continued at a rate of one drop every two minutes throughout the 30-minute irradiation period. This results in a total treatment dose of 5.4J/cm2.
7. Therapeutic contact lens. A bandage contact lens (BCL) is applied to the eye after the procedure to promote epithelial healing and decrease pain.
Post-op Care
Topical antibiotics are prescribed three to four times per day until re-epithelialization, and topical steroids are tapered over a month. Often, an NSAID is prescribed for the first few days after the procedure to help with pain. An oral narcotic can also be considered in the two to five days after the procedure to help with post-op pain. Preservative-free artificial tears are used for lubrication and comfort, and no eye rubbing is strongly encouraged.
 |
|
Fig. 3. Irradiation with UVA light following saturation of the cornea with riboflavin. Click image to enlarge. |
Follow-up/Comanagement
A typical postoperative follow-up schedule entails an exam at day one, days four to seven, one month, three months and six to 12 months.
Day one: The eye is evaluated for normal healing and a properly positioned BCL. Blurry vision and discomfort are expected.
Days four to seven: The corneal epithelium is monitored for closure, and the BCL is removed once fully reepithelialized.
Month one: Visual acuity and refraction can be assessed more accurately, although these will continue to change. Corneal haze and a demarcation line is expected. Contact lens wear can be resumed at this time.
Month three: Corneal haze is often resolved. Patients are continually monitored for stability with corneal topography every six to 12 months.
Potential Complications
CXL is considered a minimally invasive procedure with a low complication rate; however, potential complications can arise, mainly as a result of epithelial debridement. Postoperative pain, infection, stromal haze, inability to heal and reduced visual acuity are the most commonly reported adverse events.16
 |
|
Fig. 4. The iLink remains the first and only FDA-approved crosslinking device. Click image to enlarge. |
On the Horizon
In an attempt to circumvent the possible side effects, investigational modifications to the Dresden protocol involve transepithelial or epithelium-on (epi-on) CXL. Various protocols aimed at delivering riboflavin across an intact epithelium remain an active subject of research and development, including molecules to enhance penetration, riboflavin-soaked sponges, pulsed UV light and oxygen goggles.4
Takeaways
Corneal ectasia is progressive and irreversible; thus, the long-term prognosis hinges on the ability to diagnose and manage the condition in its early stages. In the last two decades, CXL has emerged as an effective method to halt corneal ectasia leading to vision preservation, better contact lens fitting, cornea transplant prevention and, ultimately, improved quality of life. Optometrists are on the front lines of diagnosing corneal ectasias and are actively involved in performing CXL across multiple states.
In regions where this procedure falls outside the current scope of practice, a collaborative comanagement approach fosters valuable professional relationships with our ophthalmology colleagues, ensuring optimal care for our patients.
Dr. Cappellani is a residency-trained optometrist with clinical emphasis on specialty contact lenses, myopia management and ocular disease at Mission Eye Care in Calgary, AB, Canada. She is a fellow of the American Academy of Optometry and Scleral Lens Society, as well as a diplomate of the American Board of Optometry. She has no financial disclosures.
Dr. Lighthizer is the associate dean, director of continuing education and chief of specialty care clinics at the NSU Oklahoma College of Optometry. He is a founding member and immediate past president of the Intrepid Eye Society. Dr. Lighthizer’s full disclosure list can be found here.
1. Wagner H, Barr JT, Zadnik K. Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study: methods and findings to date. Cont Lens Anterior Eye. 2007;30(4):223-32. 2. Hashemi H, Heydarian S, Hooshmand E, The prevalence and risk factors for keratoconus: a systematic review and meta-analysis. Cornea. 2020;39(2):263-70. 3. Munir SZ, Munir WM, Albrecht J. Estimated prevalence of keratoconus in the United States from a large vision insurance database. Eye Contact Lens. 2021;47(9),505-10. 4. Angelo L, Gokul Boptom A, McGhee C, Ziaei M. Corneal crosslinking: present and future. Asia Pac J Ophthalmol (Phila). 2022;11(5):441-52. 5. Spörl E, Huhle M, Kasper M, Seiler T. Artificial stiffening of the cornea by induction of intrastromal crosslinks. Der Ophthalmologe. 1997;94,902-06. 6. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-A–induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-7. 7. Hersh PS, Stulting RD, Muller D, et al; United States Crosslinking Study Group. United States multicenter clinical trial of corneal collagen crosslinking for keratoconus treatment. Ophthalmology. 2017;124(9):1259-70. 8. Mohammadpour M, Masoumi A, Mirghorbani M, et al. Updates on corneal collagen cross-linking: Indications, techniques and clinical outcomes. J Curr Ophthalmol. 2017;29(4):235-47. 9. Lindstrom RL, Berdahl JP, Donnenfeld ED, et al. Corneal crosslinking vs. conventional management for keratoconus: a lifetime economic model. J Med Econ. 2021;24(1):410-20. 10. Mathews P, Benbow A, Corcoran K, et al. 2022 Eye Banking Statistical Report—executive summary. EBCT. 2023;2(3):e8-12. 11. Gomes JA, Tan D, Rapuano CJ, et al. (2015). Global consensus on keratoconus and ectatic diseases. Cornea. 2015:34(4):359-69. 12. iLink. Understanding insurance coverage for cross-linking in the US. Glaukos. www.glaukos-ilink.com/blogs/understanding-insurance-coverage-for-cross-linking-in-the-u-s/. Accessed November 8, 2023. 13. Ferdi AC, Nguyen V, Gore DM, et al. (2019). Keratoconus natural progression: a systematic review and meta-analysis of 11,529 eyes. Ophthalmology. 2019;126(7):935-45. 14. Chu YR, Heckman J. Corneal cross-linking. In: Freisber L, Lighthizer N, Skorin L, et al., eds. The Ophthalmic Laser Handbook. Lippincott Williams & Wilkins; 2021:165-75. 15. Meiri Z, Keren S, Rosenblatt A, et al. (2016). Efficacy of corneal collagen cross-linking for the treatment of keratoconus: a systematic review and meta-analysis. Cornea. 2016;35(3):417-28. 16. Agarwal R, Jain P, Arora R. (2022). Complications of corneal collagen cross-linking. Indian J Ophthalmol. 2022;70(5):1466-74. |

