 |
A 17-year-old Black female presented to the office with a chief complaint of blurry vision OU of one month’s duration. She was interested in a new spectacle prescription. She had no prior history of trauma or pain. Her systemic and ocular histories were unremarkable. She denied allergies of any kind.
Diagnostic Data
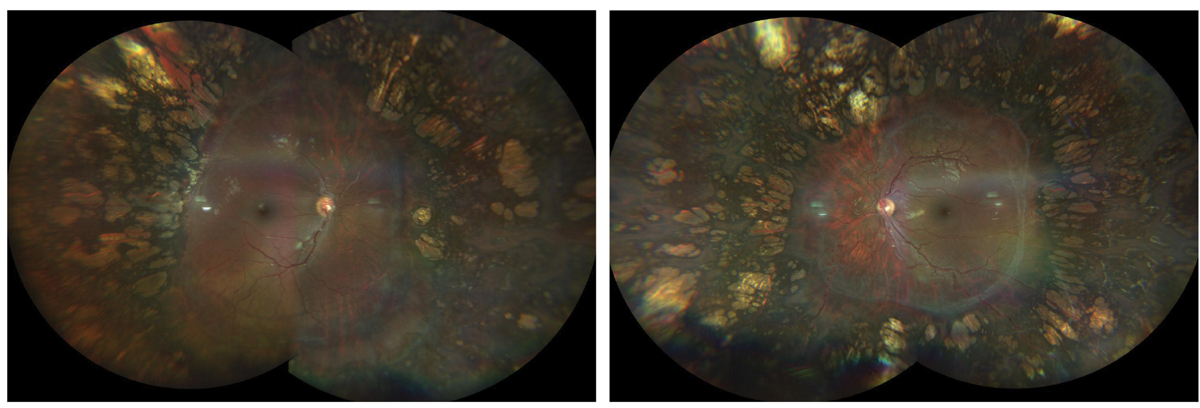
Her best-corrected entering visual acuities were 20/30 OD and 20/30 OS, at distance and near. Her external examination was normal, and there was no afferent pupillary defect. Refraction produced an excellent visual response. Her anterior segment structures were normal and Goldmann applanation tonometry measured 17mm Hg, OU. Her cup-to-disc ratios were 0.2 round OD and 0.25 round OS. The pertinent posterior segment findings are demonstrated in the photographs.
 |
|
The photos above show the right fundus (left image) and left fundus (right image) of our patient. What might cause such a presentation? Click image to enlarge. |
Next Steps
Additional studies that might yield diagnostically pertinent data include 30-2 automated perimetry to look for peripheral constriction, electroretinogram and dark adaptometry looking for rod function suppression, a 5- or 21-line raster optical coherence tomography scan of the macula and widefield color fundus photography to document baseline status.
Correspondence with her general medical team might suggest laboratory work looking for raised plasma levels of ornithine and urinalysis looking for high levels of excretion of lysine and cystine.
Diagnosis
The diagnosis in this case is gyrate atrophy (GA) bilaterally. This condition is a rare genetic disease of autosomal recessive inheritance that affects both the choroid and retina.1-5 Mutations in the ornithine aminotransferase (OAT) gene located on chromosome 10 results in a decrease or absence of the activity of the enzyme OAT.1-5
Deficiency of OAT creates a 10- to 20-fold increase in the plasma level of the amino acid ornithine (hyperornithinemia).1-5. The ocular result is characterized by the development of chorioretinal atrophic patches that start in the mid-peripheral retina in the first decade of life and progress centrally to the macular area; myopia, cystic macular edema, epiretinal membrane and posterior subcapsular cataract are commonly associated findings.1-10 In cases where maculopathy exists in the absence of angiographic or OCT demonstration of fluid formation, foveoschisis with macular pseudohole has been suggested as plausible.4,5
Gyrate atrophy, according to the literature, is particularly prevalent in Finland. However, it has been reported in many other countries of the world, including the United States, Japan, Germany, United Kingdom, India, China, Australia, France, Tunisia, Egypt, Korea, Brazil, Nepal and Turkey.8 In Finland, the prevalence is estimated to be around one in 50,000 with no predilection for gender.8,9
GA is caused by point mutations in the gene encoding of ornithine δ-aminotransferase (OAT), a tetrameric pyridoxal 5'-phosphate-dependent enzyme known to catalyze the transamination of l-ornithine and α-ketoglutarate to glutamic-γ-semialdehyde and l-glutamate in mitochondria.2 More than 50 OAT variants have been identified.2
Some forms of the disease respond to peroxidine administration (vitamin B6 at 300mg/day to 500mg/day) in the setting of an arginine-restricted diet.2,3,5-7 In some instances, creatinine—an endogenous compound synthesized from arginine, glycine and methionine—has been tried as well.6 Most patients do not respond to this therapy.2,3 Cases of macular edema can be treated with posterior sub-Tenon’s triamcinolone acetonide injection.10
This patient was referred to the retina clinic so that the diagnosis could both be confirmed and discussed with regard to new and traditional interventions. Retinology did confirm the diagnosis and the patient and parents were counseled regarding the potential for worsening of the condition over time.
Dr. Gurwood is a professor of clinical sciences at The Eye Institute of the Pennsylvania College of Optometry at Salus University. He is a co-chief of Primary Care Suite 3. He is attending medical staff in the department of ophthalmology at Albert Einstein Medical Center, Philadelphia. He has no financial interests to disclose.
1. Jasani KM, Parry NRA, Black G, Kelly SP. Unique case of gyrate atrophy with a well-preserved electroretinogram (ERG). BMJ Case Rep. 2018;2018:bcr2016217556. 2. Montioli R, Desbats MA, Grottelli S, Doimo M, Bellezza I, Borri Voltattorni C, Salviati L, Cellini B. Molecular and cellular basis of ornithine δ-aminotransferase deficiency caused by the V332M mutation associated with gyrate atrophy of the choroid and retina. Biochim Biophys Acta Mol Basis Dis. 2018;1864(11):3629-38. 3 Cui X, Jauregui R, Park KS, Tsang SH. Multimodal characterization of a novel mutation causing vitamin B6-responsive gyrate atrophy. Ophthalmic Genet. 2018;39(4):512-6. 4. Tripathy K, Chawla R, Sharma YR, Gogia V. Ultrawide field fluorescein angiogram in a family with gyrate atrophy and foveoschisis. Oman J Ophthalmol. 2016;9(2):104-6. 5. Zhioua Braham I, Ammous I, Maalej R, Boukari M, Mili Boussen I, Errais K, Zhioua R. Multimodal imaging of foveoschisis and macular pseudohole associated with gyrate atrophy: a family report. BMC Ophthalmol. 2018;18(1):89. 6. Kim SJ, Lim DH, Kim JH, Kang SW. Gyrate atrophy of the choroid and retina diagnosed by ornithine-δ-aminotransferase gene analysis: a case report. Korean J Ophthalmol. 2013;27(5):388-91. 7. Tanzer F, Firat M, Alagoz M, Erdogan H. Gyrate atrophy of the choroid and retina with hyperornithinemia, cystinuria and lysinuria responsive to vitamin B6. BMJ Case Rep. 2011;2011:bcr0720103200. 8. Elnahry AG, Tripathy K. Gyrate atrophy of the choroid and retina. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020(7):PMID: 32491691. 9. Mäntyjärvi M, Tuppurainen K. Colour vision in gyrate atrophy. Vision Res. 1998;38(21):3409-12. 10.Alparslan Ş, Fatih MT, Muhammed Ş, Adnan Y. Cystoid macular edema secondary to gyrate atrophy in a child treated with sub-tenon injection of triamcinolone acetonide. Rom J Ophthalmol. 2018;62(3):246-9. |

