A Closer Look at EyelidsThe eyelids are the first structure examined under the slit lamp microscope and are often quickly evaluated. Yet accurate assessments of common eyelid conditions and appropriate medical management can improve patients' quality of life. The October issue of Review of Optometry gives readers a rundown on best practices regarding eyelid health. |
Meibomian gland dysfunction (MGD) is a common and chronic condition and contributes to nearly 60% of all dry eye disease (DED).1 Although the pathophysiology of MGD is poorly understood, it is characterized by the obstructions of the terminal ducts, cystic dilation, acinar cell dropout and gland dropout caused by hyperkeratinized ductal epithelium (Figures 1 and 2). This leads to qualitative and quantitative changes in the MG secretion and reduced meibum to coat the aqueous layer of the tear film, resulting in tear film instability and evaporative dry eye. The diagnosis of MGD can be made based on the clinical examination, imaging and patient symptomatology; however, more than half of MGD patients do not present with symptoms.2
Just about all optometrists are able to recognize MGD, but there are nuances to the grading of cases that impact management decisions. These finer points may be less familiar and will be discussed below. Management strategies involving in-office interventions are not yet mainstream practice but are increasingly being adopted by optometrists. Thus, the remainder of this feature will detail the procedures and devices in order to help you integrate these into practice should you choose to.
Diagnosis
Traditionally, MGD was identified by assessing the function of the glands by simply pushing on them and asking patients their symptoms. Recent literature shows the value of assessing the morphology of the glands, thus broadening our diagnosis to looking at structure and function. The structure is assessed with photos of the anterior segment—specifically meibography—an infrared image of the glands (Figure 2) or by diffuse/transillumination of the glands with the slit lamp (Figure 3).
Upon structural assessment, evidence of dilation, truncation, tortuosity and/or atrophy of the glands suggests a dysfunction in their ability to produce and/or secrete meibum. Functional assessment entails gentle fingertip expression of the glands to look for evidence of thick, pasty and/or waxy meibum with reduced meibum production, which suggests a dysfunction in the quality and quantity of meibum being released.3
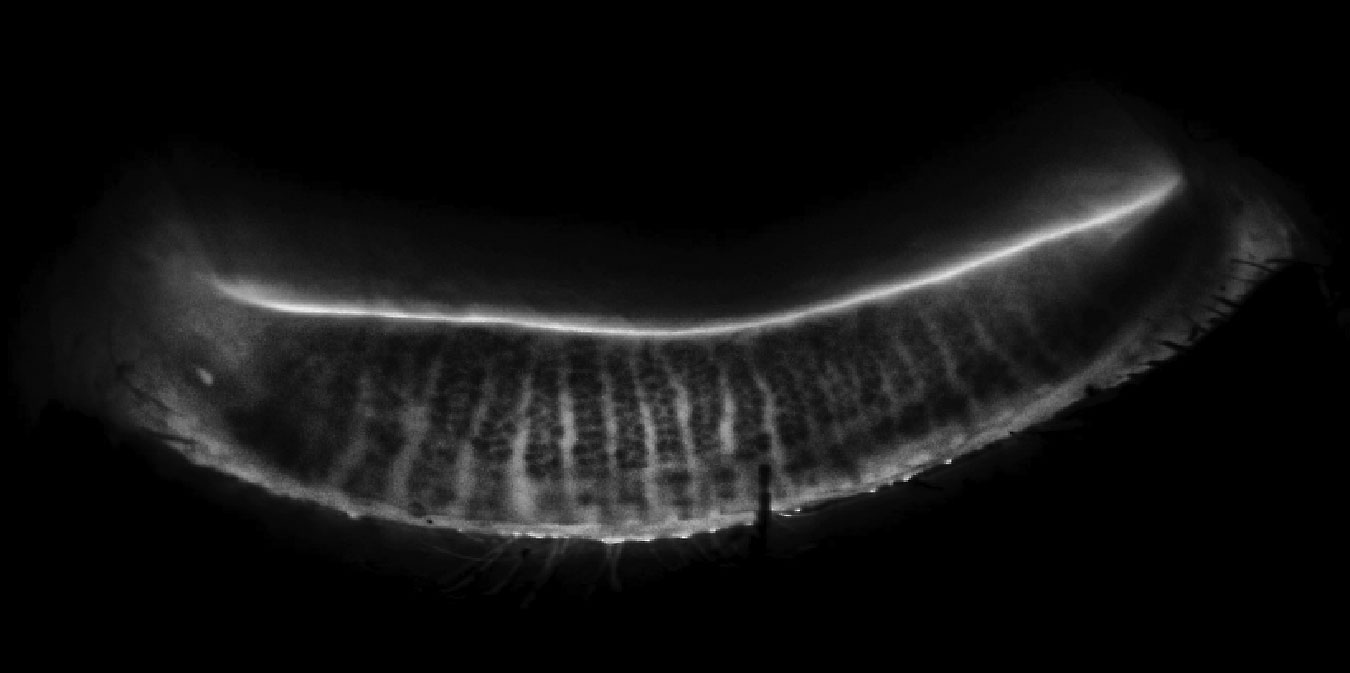 |
|
Fig. 1. Normal MG morphology on LipiScan meibography. Click image to enlarge. |
Grading MGD
In keeping with this approach that considers both structure and function (a paradigm common in glaucoma and many other ocular disease), let’s consider each of these spheres in detail.
Function is assessed by using a clinical grading scale called the Meibomian Glands Yielding Liquid Secretion (MGYLS) score. Gland expressibility is important when assessing the quality and quantity of the secreted meibum. The Meibomian Gland Evaluator (Johnson & Johnson) delivers a standard force of 1.25g/mm2 over an area of 40mm2 (eight glands) that approximates the force applied to the eyelids and MG during a natural blink. Lipid secretion from each gland is assessed to determine the MGYLS score. Eight glands are assessed at a time. For example, if only two thirds of glands on the lower eyelid are producing liquid secretion, the MGYLS is 16/24.
Meibographic imaging can be helpful in conjunction with expressibility scores to determine if an inactive gland is due to pathological or physiological reasons.3
Three types of MG secretions were first described in one study:4
• Clear, oily lipid
• Waxy, cloudy
• Opaque, inspissated
Structure is assessed using the meiboscore grading scale. It is now a well identified and widely used method of assessing morphology:5
• Grade 0 = no loss of meibomian glands
• Grade 1 = less than the one-third loss of the total area
• Grade 2 = less than two-thirds loss of the total area
• Grade 3 = more than two-thirds loss of the total area
Therefore, a careful assessment of the structure and function of the glands is necessary to understand disease severity and help guide treatment.
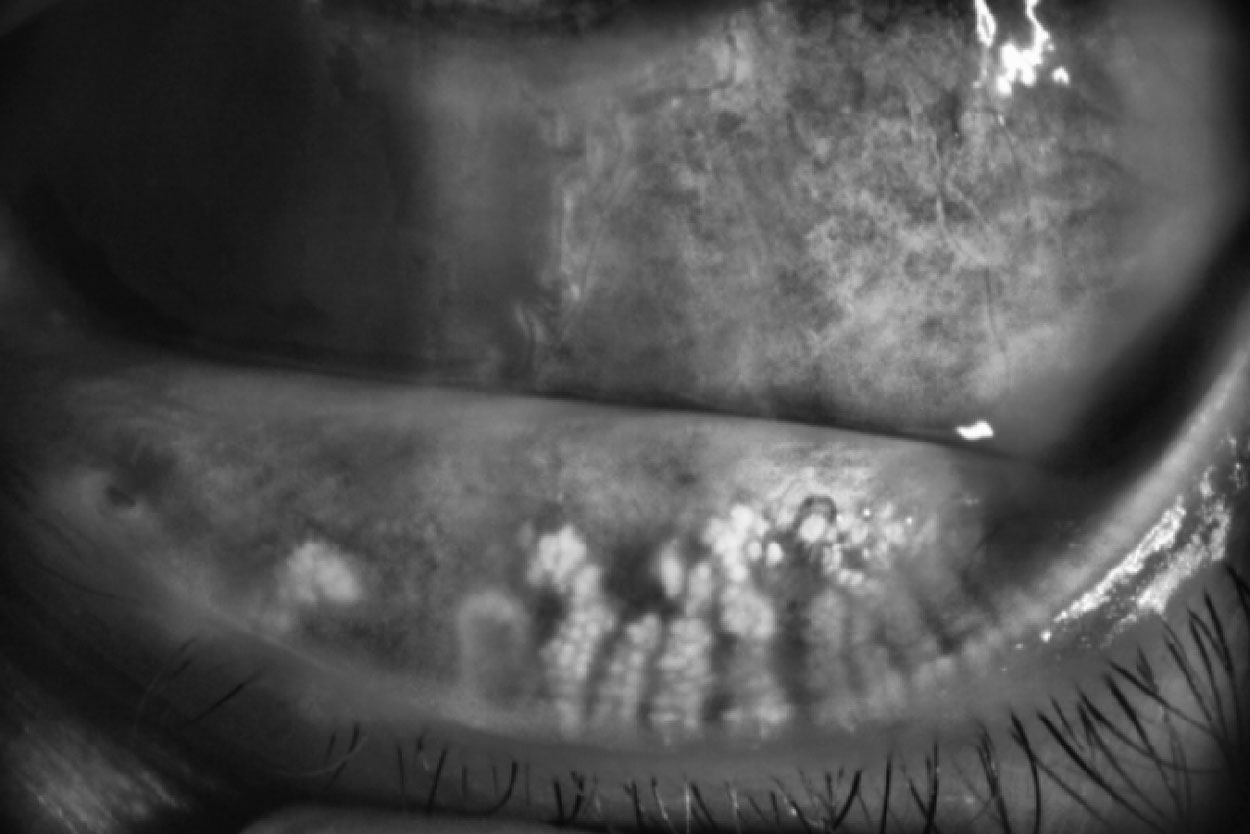 |
Fig. 2. Abnormal MG morphology with truncation and atrophy of glands. Click image to enlarge. |
Appropriate Therapy
As MGD is the leading cause of DED, treating it is critical, but with so many treatment options available, how do we know which one to choose?
Treatments range from age-old (warm compresses and eyelid hygiene) to brand new (Miebo, the perfluorohexyloctane drops just launched by Bausch + Lomb that target evaporation). Miebo, in conjunction with MG evacuation treatments, could be beneficial in controlling signs and symptoms of MGD, as perfluorohexyloctane helps to reduce the evaporation rate and stabilizes the tear film, therefore improving the homeostasis of the ocular surface.
Other pharmaceutical options include topical or oral antibiotics and steroidal or nonsteroidal anti-inflammatories, as well as nutraceuticals. Direct manipulation of the affected area is yet another avenue for treatment, as physical interventions like blepharoexfoliation, thermal evacuation, light-based therapies and intraductal probing all have a potential role to play.
It is established that MGD is associated with changes in the composition and structure of meibum, which contributes to the vicious cycle of impaired tear film quality. The physical changes of the meibum are associated with an increase in lipid melting point, starting at 35°C in obstructive MGD compared with 32°C in normal eyes.6 In-office treatments aim to raise the temperature in MGs, melt the stagnant meibum and evacuate it by applying pressure. Subsequently, lid margins should be evaluated for telangiectasia, biofilm, eyelid wiper epitheliopathy, blepharitis and hyperkeratinized epithelial cells obstructing the MG secretion (Figure 4).
The meiboscore scale may be used as a guide to treatment with mild-moderate cases that would benefit most from thermal evacuation procedures, as well as more MGD and atrophy that would benefit procedures.5
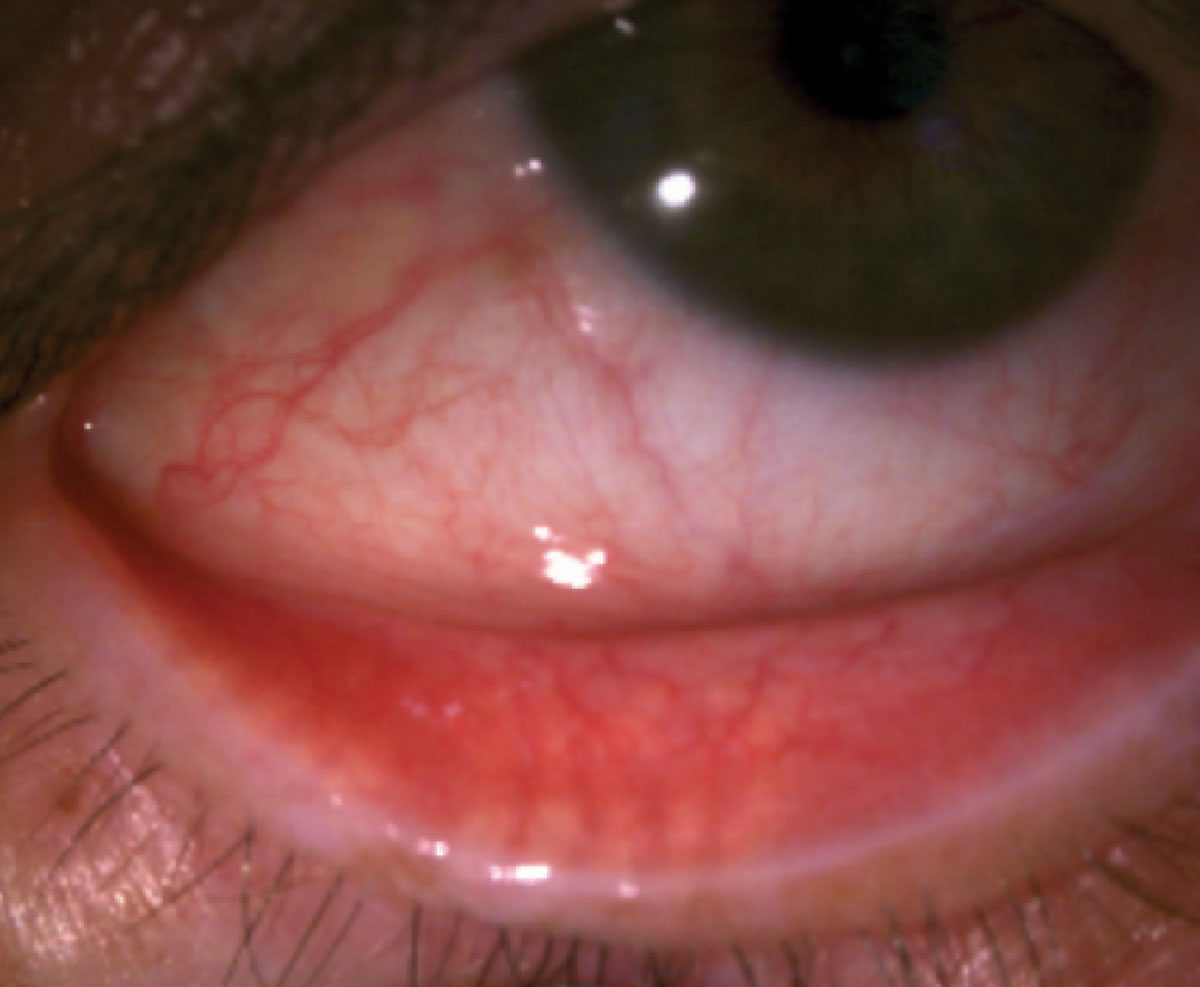 |
Fig. 3. Diffuse illumination of MG structure. Click here to enlarge image. |
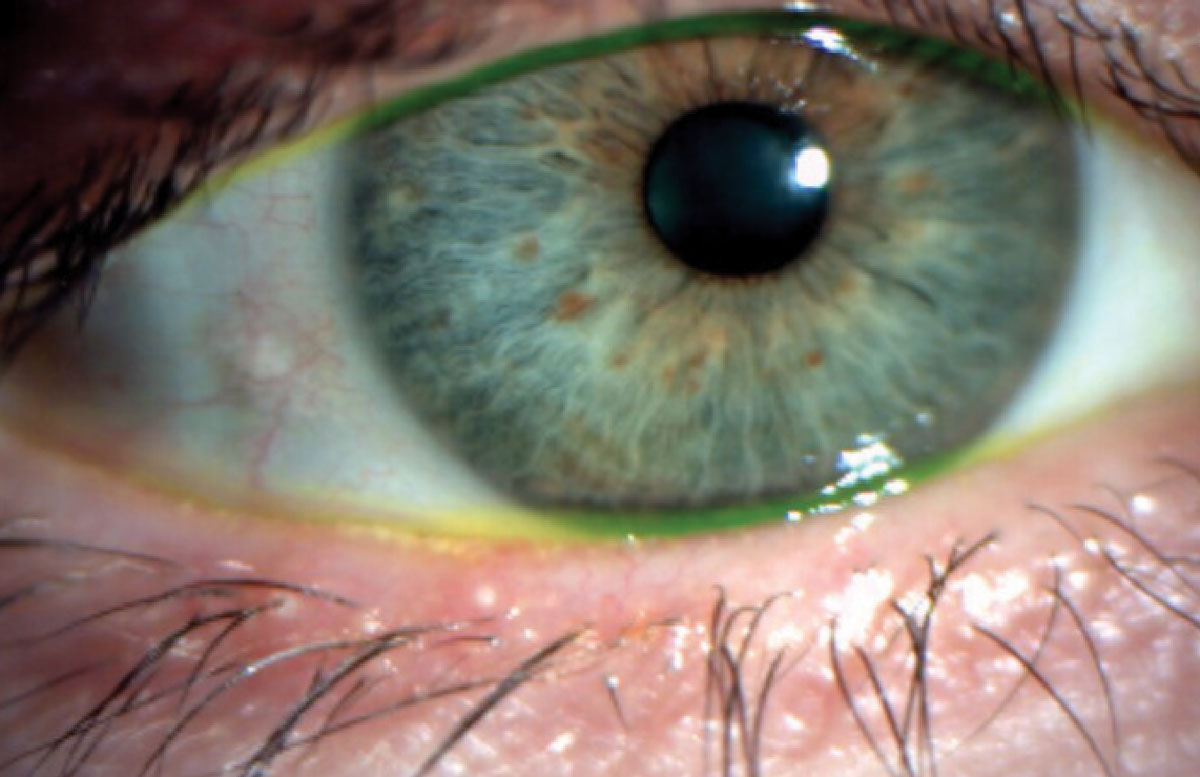 |
Fig. 4. Hyperkeratinization along the eyelid margin. Click image to enlarge. |
Blepharoexfoliation
Blepharitis is commonly associated with MGD and DED, with over 40% of patients in primary eye care having the condition, of which more than half are due to overpopulation of Demodex mites.7 Patients often complain of itchy and irritated eyes, especially in the morning, as the mites tend to be more active at night. To diagnose the condition, have the patient look down to examine the top eyelashes to look for crusts, flakes or collarettes. The latter are pathognomonic for Demodex (Figure 5).
In-office procedures of blepharoexfoliation may offer a more efficient treatment and faster symptom relief, followed by home eyelid hygiene for maintenance. Blepharoexfoliation should be combined with in-office MG expressions to remove biofilm and hyperkeratinized epithelial cells along the eyelid margin.
There are several options for in-office blepharoexfoliation:
• Zocular Eyelid System Treatment (ZEST), a manual eyelid cleaning system containing gel with activated okra polysaccharide complex.
• BlephEx (Alcon), a handheld device to mechanically remove debris and exfoliate the eyelid margin.
• NuLids Pro (NuSight Medical), an in-office version of the home eyelid cleaning device NuLids. This is a great tool for patients to use at home as part of their lid hygiene maintenance after an in-office procedure.
• Lid debridement with tools like a Karpecki debrider or golf spud, which removes biofilm and excess keratin from the lid margins. However, unlike the aforementioned tools, these methods would not address blepharitis.
Once the eyelids are cleared of debris and margins are exfoliated, a thermal or light-based treatment followed by MG expressions can be performed. Blepharoexfoliation should be repeated as often as needed based on symptom relief and appearance, as well as severity of eyelid hyperkeratinization and blepharitis.
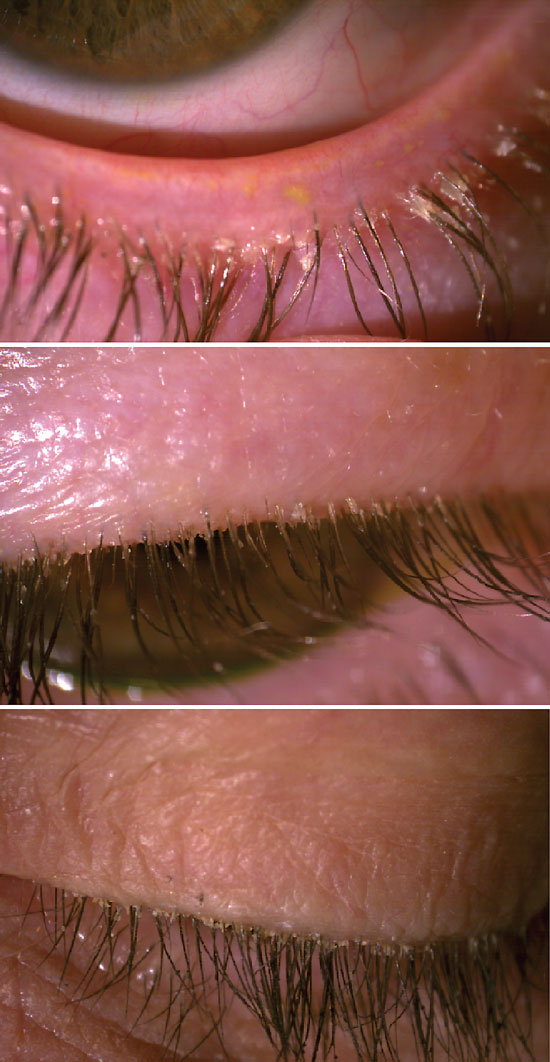 |
|
Fig. 5. Collarettes in Demodex blepharitis. Click image to enlarge. |
Thermal Treatments
This category of intervention has seen newer entrants in recent years, giving optometrists several systems to choose from.
• LipiFlow (Johnson & Johnson Vision). The oldest and most familiar of such systems, this device delivers consistent heat at 42.5°C to the inner eyelids and pulsating pressure of 5.2psi to melt and evacuate stagnant meibum. The unit consists of a desktop console that regulates the heat and pressure delivered to the patient’s eyelids via sterile single-use applicators that vault over the cornea.
The advantages of this treatment are that it is hands-free and automated, both eyes are treated at the same time for 12 minutes and the applicator insertion and removal can easily be delegated to a trained technician. The downside is that applicators come in one size and the treatments are not customizable. When selecting patients, those with mild to moderate MGD show the best results with LipiFlow. Although less effective in advanced MGD, a single LipiFlow treatment session has been shown to be effective in improving meibomian gland secretion for up to three years in patients with mild to moderate MGD.8
• Systane iLux (Alcon). This handheld device uses single-use sterile tips to deliver LED-based therapeutic heat to the eyelids while applying pressure to evacuate the meibum. The advantages are that the unit is portable and small, and treatments are customizable if more pressure or retreatment is needed because the MG orifices and meibum excretions are visible during the procedure. The downsides are that each eye is treated separately and it can be time consuming for the doctor or practice. A single treatment with iLux has been shown to improve signs and symptoms at two weeks, lasting up to 12 weeks, and its efficacy was shown to be noninferior to LipiFlow.9
• TearCare (Sight Sciences). This is a blink-assisted thermal delivery device that consists of a small portable hub with temperature and time control and what the company calls Smart Lids, which adhere externally to the top and bottom eyelids. The treatment temperature is 45°C for 15 minutes and both eyes are treated at the same time. Patients can keep their eyes open and are instructed to do blinking exercises during treatments. MG evacuation is performed after thermal treatment using MG expressor forceps. The treatments are customizable, as the temperature and treatment time are adjustable. The effectiveness of a single TearCare treatment has been shown to last up to six months and, with retreatment, up to 12 months.10
• MiboFlo (Mibo Medical Group). This thermal delivery system contains a console that controls temperature and a wand with two reusable tips that can be disinfected. Ultrasound gel is used over the skin, allowing the tips to glide over the lids to distribute the temperature of 42.5°C for 12 minutes, followed by manual MG expressions. Both time and temperature are adjustable, therefore treatments can be customized. With a new dual handpiece option, the treatments can be performed by a clinician monocularly or binocularly. The efficacy of MiboFlo treatment can last up to six months, although retreatment is often needed.11
• Radiofrequency (RF) devices. Ablative RF has been used for decades in numerous surgical procedures, whereas non-ablative RF is used for cosmetic procedures such as skin tightening and wrinkle reduction and prevention. Radio waves at 4MHz are delivered with a single electrode in a monopolar RF, which requires a grounding pad or between two electrodes in bipolar RF. The electric current then generates heat up to 45°C in the tissue, which stimulates collagen and elastin formation. The temperature can be adjusted to safe levels when applied over the lids in a circular motion, which heats the MGs to melt thick and stagnant meibum. Plastic corneal shields can protect the ocular surface and apply backpressure on the lids to help with meibum evacuation during a monopolar RF treatment. Corneal shields are not necessary for bipolar RF.
Additionally, MG expression can be performed with MG expressor forceps after RF treatment. The benefit of this approach is that, while treating MGD, it also offers aesthetic benefits like skin tightening and wrinkle reduction in treated areas. Skin tightening around the eyelids can also result in a more adequate, full blink, assisting in the mitigation of MGD’s effects. In one study comparing the effects of RF treatment to LipiFlow, both groups showed improvements in MG expressions, wax plug scoring and SPEED scores.12
Light-based Therapies
These procedures have a somewhat steeper learning curve than thermal pulsation but are increasingly embraced within eyecare. Note that ODs may not be permitted to perform these procedures in every state; be sure to check with your board before proceeding.
• Intense pulsed light (IPL). In 1995, this technology was cleared by the FDA to treat telangiectasia of the skin; by happenstance, dermatologists noticed that treatments near the eyes led to improvements in dry eye symptoms, and it has been used in eyecare for the past two decades. IPL uses broad wavelength non-coherent light and a series of filters ranging from 515nm to 715nm to target the skin’s three main chromophores (hemoglobin, water and melanin). The proposed mechanisms of action are thrombosis of abnormal telangiectatic blood vessels on lid margin and liquefaction of meibum. The increase in skin temperature from IPL may be modest and transient. Treatment effects include reducing Demodex infestation, stimulating the mitochondria and thus inducing photobiomodulation, increasing the intracellular activity of the MGs and improving the microstructure and macrostructure of MG acini.13-14
Four sessions of IPL followed by MG expressions performed every two weeks are effective in treating patients with ocular rosacea and associated MGD; this protocol has been shown to significantly improve signs and symptoms.15
 |
|
Click image to enlarge. |
Treatment protocols vary; some treat the eyelid directly with corneal shields in place (off-label), while the on-label use of the OptiLight (Lumenis) is tragus to tragus white sparing the eyelids (Figure 6).
Typically, patients need at least four treatments, each separated by two to six weeks and may need one to two treatments a year for maintenance. More advanced MGD cases may need a greater number of application and a combination of IPL with thermal treatments mentioned above to effectively evacuate obstructed glands.
Patient selection is important as IPL can cause skin photosensitivity or pigmentary changes; it is contraindicated for patients on certain medications like doxycycline, those with skin conditions like melasma or darker skin types like Fitzpatrick V and VI. However, with the right patient selection, there are many advantages of IPL, as it targets the root cause of DED by improving MG function and reducing inflammation, and it has been shown to be effective even in the most advanced cases of MGD. In addition, an off-label IPL treatment of chalazion has been shown to have similar resolution rates as excision surgery, while also preventing the recurrence of gland obstruction and improving MG function (Figure 7).16
• Low-level-light therapy (LLLT). This procedure delivers red or near-infrared light from a low-power LED-based light source to treat MGD through photobiomodulation. LLLT has been used in dermatology as an adjunct to aesthetic procedures to promote wound healing, decrease inflammation and reduce pain. Equinox (Marco) offers a specially designed mask that delivers three light options to the eyelids and upper two thirds of the face: red (633nm), used for photobiomodulation of MGs, wound healing and collagen and elastin production; blue light (400nm to 450nm), which has a bacteriostatic effect used to treat blepharitis; and yellow light (560nm to 580nm), which is used to stimulate cells’ metabolism and relieve swelling.
Non-ophthalmic units adopted from aesthetic companies like Celluma are an affordable alternative used by clinicians as a standalone treatment or add-on to IPL. We find that LLLT is a great option for the pediatric population with blepharitis, chalazion and MGD, or for those who may not be a good candidate or tolerate IPL. Two treatments a week for three weeks have been shown to reduce signs and symptoms of MGD.17
Putting it All Together
Many unique challenges exist in diagnosing and managing MGD, from navigating insurance benefits to choosing appropriate treatments and perfecting your techniques in gland expressions. Let’s put it all together in a sample patient encounter:
1. Consider implementing MG imaging when establishing your dry eye clinic. Many slit lamps now offer imaging and video capabilities. There are portable meibography devices and combination units of imaging and topography available. Meibography is an invaluable tool for achieving patient “buy-in” to in-office procedures and allows them to have a tangible result in following the efficacy of their treatment plan and disease progression.
2. Address MGD at a medical visit that is billed to the patient’s medical insurance carrier. The diagnosis can be discussed briefly during a routine exam, but any related testing cannot be billed to a vision insurance plan. Anterior segment imaging or meibography is either billed for reimbursement or patients pay out-of-pocket. Offices that do not bill medical insurance carriers may opt to set an out-of-pocket fee for a DED evaluation that includes a set of imaging, such as meibography and/or corneal topography if the patient is pre-surgical. The provider may choose to include a follow-up in this fee or charge separately for each visit.
3. Patients should receive a validated dry eye questionnaire at every visit, such as SPEED, DEQ-5 or OSDI; this includes their baseline visit. A quantifiable assessment of the patient’s symptoms is important for the provider to summarize a baseline of symptom severity and a tangible and standardized method of following improvements with treatments. Patients should also commit to one symptom that bothers them the most; the progress of this symptom should be assessed at every visit.
4. Assess structure and function. Perform meibography and/or anterior segment imaging of the glands and anterior segment with vital dyes. Assessment of MG function is very important and should be documented at the initial visit; i.e., inspissated, waxy or clear lipid secretions.
5. Implement a treatment plan and follow the patient closely, within three to four weeks. Assess compliance and disease state progression at each visit.
6. Upon follow-up, perform anterior segment imaging with vital dyes and MG expressibility scores. Questionnaires are performed at each visit.
Honing Manual MG Expression
We always recommend heating the MGs to the appropriate therapeutic melting temperature for a sustained period of time prior to expressing glands in-office. As previously reported, a warm compress will not achieve the therapeutic melting temperature of the meibum; rather, in-office heating should occur with a standardized device such as the LipiFlow, TearCare, iLux or RF.5 Expressions are then performed within minutes of heating the glands.
MG expressor forceps, such as the Mastrota Meibomian Paddle, can be used to gently express the glands on each of the four lids. This is typically performed in the slit lamp after anesthetizing the eye. Sometimes it’s easier to reach the top lids by reclining the patient at a 45° angle in the chair. All four lids should be expressed. Cotton tip applicators can certainly be used for expressions, but a dedicated paddle will allow for an easier experience for the patient and provider. When holding the paddle, we recommend a vertical/45° angle to the lid margin to access the distal portion of the glands. Circular and rectangular shapes of paddles are available.
With an especially inspissated gland, providers should sustain pressure for a few seconds longer than easily expressed glands. More force is not necessary; rather, sustained pressure and passing the gland multiple times is more effective. Debridement of the eyelid margin can provide great relief to symptomatic patients before and after manual expressions in-office.
 |
Takeaways
MGD treatment is not only rewards patients and providers but also is an excellent practice builder. Start by understanding the literature on the pathophysiology of MGD and educate your patients on the disease state. Then, consider offering one blepharoexfoliation and thermal/light treatment. Augment your in-office offerings with at-home products for sale and focus on improving patient symptoms with your treatment regimen. As your dry eye clinic grows, you can add more devices and expand your services.
Dr. Ioussifova graduated from the New England College of Optometry and completed a residency program in community health and ocular disease in Boston. She is a fellow of the American Academy of Optometry, previously served as an adjunct clinical faculty at the Pacific University College of Optometry and was an examiner for the NBEO. Dr. Ioussifova owns South Waterfront Eye Center, a practice with special interests in advanced dry eye treatments, nutritional counseling and aesthetic services.
Dr. Kataria graduated from the New England College of Optometry, completed her clinical rotations at Bascom Palmer Eye Institute as well as a residency in primary care and ocular disease at the Baltimore VAMC. After practicing medical optometry and advanced ocular surface disease as an associate, she now owns a private practice near Los Angeles called Avant Eyes Optometry & Advanced Dry Eye Center. They have no financial disclosures.
1. TCY Chan, SSW Chow, KHN Wan, HKL Yuen. Update on the association between dry eye disease and meibomian gland dysfunction. Hong Kong Medical Journal. 2019;25(1):38-47. 2. Arita R, Fukuoka S, Kawashima, M. Proposed algorithm for management of meibomian gland dysfunction based on noninvasive meibography. J Clin Med. 2021;10(1):65. 3. Tomlinson A, Bron AJ, Korb DR, et al. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. Invest Ophthalmol Vis Sci. 2011;30;52(4):2006-49. 4. Bron AJ, Benjamin L, Snibson GR. Meibomian gland disease. Classification and grading of lid changes. Eye (Lond). 1991;5(Pt 4):395-411. 5. Arita R, Itoh K, Iinoue K, Amano S. Noncontact infrared meibography to document age-related changes of the meibomian glands in a normal population. Ophthalmology. 2008;115(5):911-5. 6. Geerling G, Tauber J, Baudouin C, et al. The international workshop on meibomian gland dysfunction: report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2011;52(4):2050-64. 7. Trattler W, Karpecki P, Rapoport Y, et al. The prevalence of Demodex blepharitis in US eye care clinic patients as determined by collarettes: a pathognomonic sign. Clin Ophthalmol. 2022;16:1153-64. 8. Greiner JV. Long-term (three-year) effects of a single thermal pulsation system treatment on meibomian gland function and dry eye symptoms. Eye Contact Lens. 2016;42(2):99-107. 9. Wesley G, Bickle K, Downing J, et al. Systane iLux thermal pulsation system in the treatment of meibomian gland dysfunction: a post-hoc analysis of a 12-month, randomized, multicenter study. Clin Ophthalmol. 2022;16:3631-40. 10. Gupta PK, Holland EJ, Hovanesian J, et al. TearCare for the treatment of meibomian gland dysfunction in adult patients with dry eye disease: a masked randomized controlled trial. Cornea. 2022;41(4):417-26. 11. Gomez ML, Afshari NA, Gonzalez DD, Cheng L. Effect of thermoelectric warming therapy for the treatment of meibomian gland dysfunction. Am J Ophthalmol. 2022;242:181-8. 12. Jaccoma EH, Litherland C, Jaccoma A, Ahmed AH. Pelleve vs. LipiFlow MGD-related dry eye treatment study: the ThermaLid procedure. A pilot study comparing the efficacy of the Pellevé System to LipiFlow for the treatment of dry eye due to meibomian gland dysfunction. J Dry Eye Ocu Surf Dis. 2018;1(1):e11-21. 13. Craig JP, Chen YH, Trunbull PR. Cumulative effect of intense pulsed light therapy for meibomian gland dysfunction confirmed in prospective, double-masked, placebo controlled trial. Invest Ophthalmol Vis Sci. 2015;56(7):6194. 14. Cote S, Zhang AC, Ahmadzai V, et al. Intense pulsed light therapy for the treatment of meibomian gland dysfunction.Cochrane Database Syst Rev. 2020;3(3), CD013559. 15. Toyos R, Desai NR, Toyos M, Dell SJ. Intense pulsed light improves signs and symptoms of dry eye disease due to meibomian gland dysfunction: a randomized controlled study. PLoS One. 2022;17(6):e0270268. 16. Zhu Y, Zhao H, Huang X, et al. Novel treatment of chalazion using light-guided-tip intense pulsed light. Sci Rep. 2023;13(1):12393. 17. Park Y, Kim H, Kim S, Cho KJ. Effect of low-level light therapy in patients with dry eye: a prospective, randomized, observer-masked trial. Sci Rep. 2022;12(1):3575. |

