A 61-year-old white male presented complaining of floating spots and sparkly things in the center of his right eye for the past month. His ocular history was significant for a subconjunctival hemorrhage in his left eye due to blunt trauma by vegetative matter; this occurred one year earlier. He denied any trauma history involving the right eye.
His medical history was significant for hypertension and hyperlipidemia. He underwent aortic bifemoral bypass in 2001. Medications included lisinopril 20mg q.d., metoprolol 100mg q12h, simvastatin 40mg q.d. and prophylactic baby aspirin 81mg q.d.
Diagnostic Data
Best-corrected visual acuity was 20/150 O.D. and 20/20 O.S. Biomicroscopy revealed a normal cornea, iris, anterior chamber and lens O.U. Pupils, motilities and IOP all appeared normal.
Dilated fundus exam revealed a subtle epiretinal membrane (ERM) with associated traction lines and an unusual elevated appearance in the foveal area of the right eye. There was an incomplete posterior vitreous separation and a subtle area of thickened posterior hyaloid overlying the macula (figure 1). The macula in the left eye was flat and unremarkable.
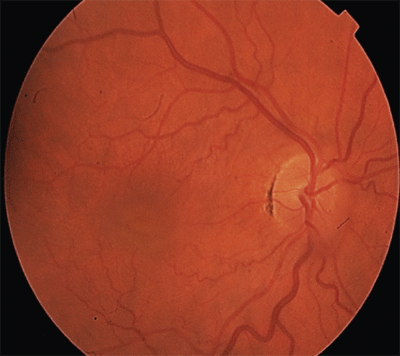 |
| 1. Fundus photo demonstrates subtle thickening of the posterior hyaloid overlying the macula and vitreous O.D. |
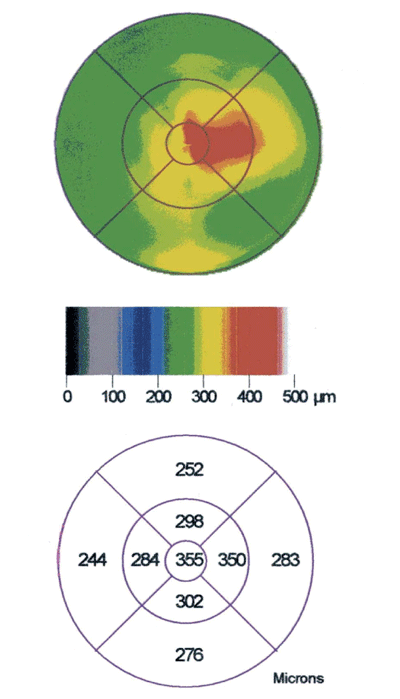
Optical coherence tomography (OCT) revealed a tuft of vitreoretinal tissue that was adherent to the fovea O.D. This caused traction in the macula that dramatically disrupted the foveal architecture (figure 2). OCT also revealed cystic fluid accumulation that appeared as a large, optically transparent intraretinal region in the fovea. Macular thickness analysis demonstrated diffuse thickening O.D. (figure 3).
 |
| 2. OCT demonstrates a tuft of vitreoretinal traction dramatically disrupting the foveal architecture O.D. |
Diagnosis
We diagnosed idiopathic vitreomacular traction syndrome (VMTS). The patients condition is the direct result of tractional forces imparted by an incomplete posterior vitreous detachment (PVD) onto the persistently adherent right macula.
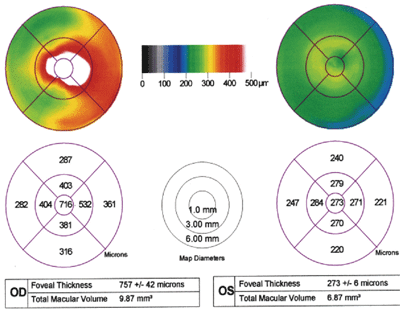 |
| 3. OCT compares the macular thickness between the two eyes. |
Treatment and Follow-up
We referred the patient to a retinal specialist, who initially monitored him for one month for potential spontaneous resolution. Due to the direct tractional etiology of the macular elevation, medical treatment of the cystoid macular edema (CME) was not pursued. The traction was still present at one month, so pars plana vitrectomy with a membrane peel O.D. was performed to release the traction caused by the persistent macular adherence to the posterior hyaloid.
|
|
| 4. OCT shows a significant decrease in macular thickness O.D. postoperatively. |
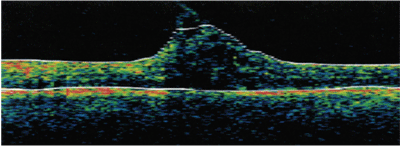
At day one post-op, the patients VA was hand motion at eight feet. OCT demonstrated a significant decrease in macular thickness (figure 4) and release of traction on the foveal architecture (figure 5).
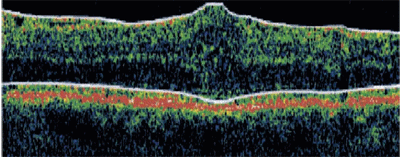 |
| 5. OCT shows a cross-section of the right macula postoperatively. |
Discussion
Idiopathic macular hole, ERM, and VMTS are macular surface disorders that may result in visual morbidity, especially in the elderly. All three conditions have been associated with pathological vitreomacular adhesions.1 Although they share some similarities, these conditions are distinct from one another based on histology, surgical anatomy, clinical characteristics and differing natural courses. Still, diagnosis is sometimes confusing, as there may be overlap in both subjective and objective findings.
VMTS, the least common of these three disorders, is characterized by an incomplete posterior vitreous detachment, in which the vitreous remains attached to the macular area. This results in retinal traction and the development of increased macular thickness and cystoid changes. Patients may experience decreased vision, metamorphopsia, photopsia, and micropsia.2,3
Some individuals have an unusually strong attachment between the posterior vitreous cortex, macula, and peripapillary retina.4-7 One theory: Pre-existing cell migration on the inner retina may cause an unusually strong vitreoretinal adhesion, preventing completion of the subsequent PVD.8 So, individuals with such anomalous adhesions might be at higher risk for developing VMTS during normal age-related vitreal changes; this is what occurred in our patient.
Two distinct clinicopathological features of VMTS suggest different forms of epiretinal fibrocellular proliferation, namely epiretinal membranes interposed in native vitreous collagen, and those composed of single cells or a cellular monolayer proliferating directly on the internal limiting membrane (ILM). Remnants of the cortical vitreous that remain attached to the ILM following posterior vitreous separation may determine the clinicopathological features of the disease. The predominance of myofibroblasts may help explain the increased prevalence of CME and the progressive vitreomacular traction that characterizes VMTS.9
VMTS, ERM and impending macular hole syndrome may present very similarly, particularly in regard to foveal appearance. (See The Other Macular Surface Disorders, below.) Differences include substantial distortion of the retinal vessels, which is more common in macular pucker, and the cystic changes that are more common in VMTS.8
There may be symptomatic overlap in patients who have VMTS and ERM. Patients who have either condition often suffer from vision loss and metamorphopsia. In contrast, patients who have impending macular hole syndrome typically have non-specific symptoms and less vision loss.8
There are some histological differences between the disorders as well. A thickened layer of epiretinal tissue can be dissected and removed from the macular area in both VMTS and ERM. The predominant ultrastructural feature of tissue specimens in VMTS is the presence of fibrous astrocytes; notably absent are retinal pigment epithelial cells, which dominate the cellular features of ERM cases.8
VMTS and impending macular hole are associated with anomalous vitreous detachment, but VMTS is rarely associated with progression to a full-thickness macular hole.8 This difference in the natural course between idiopathic macular hole and VMTS is not fully understood.
OCT can play a critical role in diagnosing VMTS and appears more sensitive than slit lamp biomicroscopy in identifying vitreoretinal adhesions. In one study, such adhesions were identified only 8% of the time using biomicroscopy vs. 30% of the time with OCT.10
OCT can help confirm the diagnosis of VMTS when clinically invisible.11 It can also help explain the pathogenesis of the disease and objectively assess anatomical improvement of secondary macular changes following surgery.12 On OCT, VMTS is characterized by the appearance of a perifoveal vitreous detachment with focal adhesion to the fovea; it is often associated with cystoid macular edema (CME).13
Vitrectomy effectively resolves traction in VMTS syndrome and improves visual acuity in most cases.1,11,14-19 This is demonstrable with OCT, which reveals release of traction and reduction in macular thickness postoperatively.11,14,15 It is the treatment of choice to prevent deterioration of visual function before irreversible damage occurs to the retina.18,19 However, approximately 10% of VMTS patients will experience spontaneous resolution of cystoid macular changes following posterior vitreous detachment, making surgery unnecessary.20 For this reason, some have suggested it is prudent to wait several months before attempting vitrectomy.21
Factors that limit visual improvement include accelerated development of nuclear sclerotic cataracts, recurrence of epiretinal membrane, persistent macular edema and macular hole development.15-17,22 Of note, one study differentiated between two types of incomplete PVD; one type appeared as a V-shaped detachment, the other as a partial detachment temporal to the fovea but attached nasally. Patients treated in the former group had a much more favorable post-surgical outcome than those in the latter.22 Such prognostic differences further illuminate the importance of careful evaluation with OCT in caring for patients with VMTS.
The rationale for vitrectomy is supported by a study of the natural course of VMTS; patients with symptomatic VMTS experienced worsening of visual acuity during the course of the study unless spontaneous, complete PVD occurred.23 The Other Macular Surface Disorders Two additional macular surface disordersidiopathic macular hole and epiretinal membrane (ERM)may adversely affect vision.
There are many similarities and differences among VMTS, impending macular hole and ERM. Specific risk factors and pathogenic features of these conditions are not entirely understood. Recognizing each disorders distinguishing features allows for a more precise diagnosis, more appropriate prognostic patient counseling and appropriate referral for surgical intervention, when indicated.
J. Donald M. Gass, M.D., described the stages of macular hole formation based on biomicroscopic findings.24 OCT findings have since been correlated to the original stages he proposed.25 His classification, still commonly used, is as follows:24
Impending, or Stage 1A macular hole. The development of a 250m to 300m yellowish spot, which represents foveolar detachment, is the earliest sign of macular hole formation. This stage is clinically subtle and may be asymptomatic; therefore, it is often missed and is most often detected in patients who have a full-thickness hole in the fellow eye.
Stage 1B hole. As the yellowish spot enlarges, it is replaced by a yellow halo. This halo enlarges further and is associated with additional loss of the foveal depression.
Stage 2 macular holes. This stage represents actual retinal dehiscence (early macular hole).
Stage 3 hole. This diameter of the hole has enlarged to about 485m. At this stage, the vitreous usually remains attached, but a vitreofoveal separation may develop. A Stage 3 hole may occur with or without an operculum. This stage is accompanied by a positive Watzke-Allen sign at the slit lamp.
Stage 4 macular hole. This is characterized by complete posterior vitreous separation accompanied by the presence of a Weiss ring in front of the optic nerve head.
OCT allows for differentiation of foveal cysts and partial- (lamellar) and full-thickness macular holes. Also, research utilizing OCT helps describe the clinical stages and progression of macular holes. An additional stage, Stage 0, has been proposed based on a study involving OCT examination of fellow eyes with unilateral idiopathic macular holes. Significant vitreoretinal interface abnormalities were identified in certain patients and were associated with a sixfold increased risk of macular hole formation.26 Another study attempting to correlate Dr. Gass" biomicroscopic classification with OCT findings found one main difference between the two: the appearance of a pseudocyst instead of a foveolar detachment in the early stages.24
When a patient presents with a macular hole, it is important to classify the stage of development. Treatment is not recommended for Stage 1 holes, as spontaneous closure can occur. Once visual acuity is reduced to 20/40 to 20/400, surgical intervention may be advised, depending on the duration and onset of visual symptoms. Options include pars plana vitrectomy, membrane peel, air-fluid exchange and gas injection for holes of recent onset in Stages 2 to 4 and of less than one year in duration.27
Epiretinal Membrane
ERM has been associated with various ocular pathologies, however, most cases are idiopathic and seem to occur only in association with PVD.28-30 It is the most common of the macular surface disorders, occurring in 12% of elderly patients.31
ERM appears as a yellow, glistening, reflective membrane within the macular area. It may be accompanied by the appearance of a pseudohole, a reddish foveal or perifoveal lesion that is often oval in shape and has a slightly irregular margin. The pseudohole represents a central defect or contracture in the ERM that creates a steep drop-off around the fovea, simulating a true macular hole.3,32 On OCT, a pseudohole appears as a steep foveal pit with thickened edges and with intact retinal tissue at the base of the depression.13 The local vasculature may become distorted, and traction-induced retinal striae may be evident.
ERMs are likely to develop following the transient vitreomacular traction that occurs during posterior vitreous detachment. Dehiscence in the internal limiting membrane occurs, allowing migration and proliferation of glial cells on the inner retinal surface.31,33-35
Most cases of ERM are non-progressive. Surgery is indicated only for cases that have clinically significant visual loss (<20/60 visual acuity or intractable symptoms).36 In such instances, pars plana vitrectomy with membrane peel is recommended.27
Dr. Convertino completed a residency at Malcom Randall VA Medical Center in Gainesville, Fla., and currently practices at The Low Vision Institute in Fort Lauderdale, Fla. Dr. Marcus is residency director and Dr. Wong is director of optometry clinical operations at Malcom Randall VA Medical Center.
1. Koerner F, Garweg J. Diseases of the vitreo-macular interface. Klin Monatsbl Augenheilkd 1999 May;214(5):305-10.
2. Maumenee AE. Further advances in the study of the macula. Arch Ophthalmol 1967 Aug;78(2):151-65.
3. Gass JD. Macular dysfunction caused by vitreous and vitreoretinal interface abnormalities: vitreous traction maculopathies. In: Gass JD.. Stereoscopic Atlas of Macular Diseases: Diagnosis and Treatment, 3rd ed. Vol. 2. St Louis: Mosby-Year Book, 1987:676-93.
4. Sebag J. Age-related differences in the human vitreoretinal interface. Arch Ophthalmol 1991 Jul;109(7):966-71.
5. Sebag J. Anatomy and pathology of the vitreo-retinal interface. Eye 1992;6(Pt 6):541-52.
6. Foos RY, Wheeler NC. Vitreoretinal juncture. Synchysis senilis and posterior vitreous detachment. Ophthalmology 1982 Dec;89(12):1502-12.
7. Smiddy WE, Green WR, Michels RG, de la Cruz Z. Ultrastructural studies of vitreomacular traction syndrome. Am J Ophthalmol 1989 Feb 15;107(2):177-85.
8. Smiddy WE, Michels RG, Green WR. Morphology, pathology, and surgery of idiopathic vitreoretinal macular disorders. A review. Retina 1990;10(4):288-96.
9. Gandorfer A, Rohleder M, Kampik A. Epiretinal pathology of vitreomacular traction syndrome. Br J Ophthalmol 2002 Aug;86(8):902-9.
10. Gallemore RP, Jumper JM, McCuen BW 2nd, et al. Diagnosis of vitreoretinal adhesions in macular disease with optical coherence tomography. Retina 2000;20(2):115-20.
11. Johnson MW. Tractional cystoid macular edema: a subtle variant of the vitreomacular traction syndrome. Am J Ophthalmol 2005 Aug;140(2):184-92.
12. Munuera JM, Garcia-Layana A, Maldonado MJ, et al. Optical coherence tomography in successful surgery of vitreomacular traction syndrome. Arch Ophthalmol 1998 Oct; 116(10):1388-9.
13. Schuman JS, Puliafito CA, Fujimoto JG. Optical Coherence Tomography of Ocular Diseases. 2nd ed. Thorofare, N.J.: Slack Inc., 2004:58.
14. Larsson J. Vitrectomy in vitreomacular traction syndrome evaluated by ocular coherence tomography (OCT) retinal mapping. Acta Ophthalmol Scand 2004 Dec;82(6):691-4.
15. Petropoulos IK, Stangos AA, Brozou CG, et al. Vitrectomy for vitreo-macular traction syndrome. Klin Monatsbl Augenheilkd 2003 Mar;220(3):122-6.
16. Gribomont AC. Surgical prognosis in idiopathic vitreomacular syndrome and epiretinal membrane. J Fr Ophthalmol 2005 Sep;28(7):739-42.
17. Jiang YR, Ma Y, Li XX. Analysis of the effect of surgical management on vitreomacular traction syndrome. Zhonghua Yan Ke Za Zhi 2004 Oct;40(10):670-3.
18. Smiddy WE, Michels RG, Glaser BM, de Bustros S. Vitrectomy for macular traction caused by incomplete vitreous separation. Arch Ophthalmol 1988 May;106(5):624-8.
19. McDonald HR, Johnson RN, Schatz H. Surgical results in the vitreomacular traction syndrome. Ophthalmology 1994 Aug;101(8):1397-403.
20. Kusaka S, Saito Y, Okada AA, et al. Optical coherence tomography in spontaneously resolving vitreomacular traction syndrome. Ophthalmologica 2001 Mar-Apr;215(2):139-41.
21. Rodriguez A, Infante R, Rodriguez FJ, Valencia M. Spontaneous separation in idiopathic vitreomacular traction syndrome associated with contralateral full-thickness macular hole. Eur J Ophthalmol 2006 Sep-Oct;16(5):733-40.
22. Yamada N, Kishi S. Tomographic features and surgical outcomes of vitreomacular traction syndrome. Am J Ophthalmol 2005 Jan;139(1):112-7.
23. Hikichi T, Yoshida A, Trempe CL. Course of vitreomacular traction syndrome. Am J Ophthalmol 1995 Jan;119(1):55-61.
24. Johnson RN, Gass JD. Idiopathic macular holes: observations, stages of formation, and implications for surgical intervention. Ophthalmology 1988 Jul;95(7):917-24.
25. Azzolini C, Patelli F, Brancato R. Correlation between optical coherence tomography data and biomicroscopic interpretation of idiopathic macular hole. Am J Ophthalmol 2001 Sep;132(3):348-55.
26. Chan A, Duker JS, Schuman JS, Fujimoto JG. Stage 0 macular holes: observations by optical coherence tomography. Ophthalmology 2004 Nov;111(11):2027-32.
27. Kaiser PK, Friedman NJ, Pineda R 2nd. The Massachusetts Eye and Ear Infirmary Illustrated Manual of Ophthalmology. 2nd ed. Philadelphia: W.B. Saunders, 2004:341.
28. Wise GN. Relationship of idiopathic preretinal macular fibrosis to posterior vitreous detachment. Am J Ophthalmol 1975 Mar;79(3):358-62.
29. Wiznia RA. Posterior vitreous detachment and idiopathic preretinal macular gliosis. Am J Ophthalmol 1986 Aug 15; 102(2):196-8.
30. Hirokawa H, Jalkh AE, Takahashi M, et al. Role of the vitreous in idiopathic preretinal macular fibrosis. Am J Ophthalmol 1986 Feb 15;101(2):166-9.
31. Roth AM, Foos RY. Surface wrinkling retinopathy in eyes enucleated at autopsy. Trans Am Acad Ophthalmol Otolaryngol 1971 Sep-Oct;75(5):1047-58.
32. Allen AW Jr, Gass JD. Contraction of a perifoveal epiretinal membrane simulating a macular hole. Am J Ophthalmol 1976 Nov;82(5):684-91.
33. Smiddy WE, Maguire AM, Green WR, et al. Idiopathic epiretinal membranes. Ultrastructural characteristics and clinicopathologic correlation. Ophthalmology 1989 Jun; 96(6):811-21.
34. Appiah AP, Hirose T, Kado M. A review of 324 cases of idiopathic premacular gliosis. Am J Ophthalmol 1988 Nov 15;106(5):533-5.
35. Ciulla TA, Pesavento RD. Epiretinal fibrosis. Ophthalmic Surg Lasers 1997 Aug;28(8):670-9.
36. Fraser-Bell S, Guzowski M, Rochtchina E, et al. Five-year cumulative incidence and progression of epiretinal membranes: the Blue Mountains Eye Study. Ophthalmology 2003 Jan;110(1):34-40.