 |
In 2010, a 38-year-old Caucasian female presented to the office as a new patient with complaints related to visual changes consistent with early presbyopia. She noted that both her distance and near vision had changed gradually over the prior three years, with complaints of strain while performing near tasks.
At this visit, her medications included estradiol QD and Wellbutrin SR QD, and she reported no medication allergies. She reported a family history of glaucoma in her mother, who apparently began drops when she was in her 50s, as reported by the patient. Entering visual acuities through hyperopic astigmatic correction were 20/25- OD and OS.
Examination
A moderate increase in hyperopia was noted on refraction. Best corrected visual acuities were 20/20 OU. Pupils were equal, round and responsive to light and accommodation. We noted no afferent pupillary defect. Extraocular movements were full in all positions of gaze.
A slit lamp examination of her anterior segments was essentially unremarkable. Her angles as estimated at the slit lamp were open grade 2 OU, with no sectoral narrowing. Intraocular pressures (IOP) were 27mm Hg OD and 26mm Hg OS at 10:25am. CCT measured 641µm OD and 635µm OS.
Through dilated pupils, her crystalline lenses were clear in both eyes. Her anterior vitreous was also clear in both eyes. The posterior segment was essentially unremarkable. Specifically, stereoscopic evaluation of her optic discs demonstrated healthy, plush, well perfused neuroretinal rims bilaterally, with relatively small cups estimated to be 0.30 x 0.30 OU. The discs were of normal sizes. The retinal vasculature and macular examinations were all normal, as was the posterior vitreous. Her peripheral retinal evaluation was normal, save for some mild cystoid in each eye.
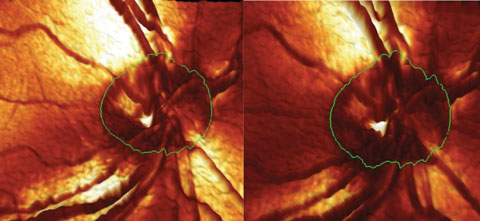 |
| Fig. 1. This image shows the contours of the right optic nerve. Note the change in the neuroretinal rim in the inferotemporal aspect between the baseline (left) and the conversion image (right). |
Monitoring
She was determined to be a glaucoma suspect based on the family history of glaucoma, as well as IOP in the mid-to-upper 20s. As such, baseline HRT 3 images and stereo optic disc photos were obtained as baseline. Given her relatively thick pachs and essentially healthy neuroretinal rims, her risk of converting to frank glaucoma was deemed low. She was scheduled for a yearly follow up, pending the results of a baseline threshold field and repeat IOP measurements after three months. She returned as requested for the field study, at which time her IOP was 26mm Hg OU at noon. SAP fields were normal in both eyes with good reliability indices. She was subsequently scheduled for repeat fields, pressure, HRT 3 imaging and optic nerve photos in one year.
She was compliant with scheduled visits yearly. When seen in August 2013, her IOP had risen to 32mm Hg OD and 27mm Hg OS. Her visual field test results were normal, and HRT 3 imaging demonstrated no changes from baseline. Fields performed at this visit were HEP Flicker Defined Form (FDF) threshold fields. Baseline OCTs were performed this day as well, demonstrating a normal RNFL scan. Given the increase in IOP, a follow up was scheduled in six months. At this follow up visit, IOP measured 33mm Hg OD and 26mm Hg OS. HRT 3 imaging demonstrated a small area of change in the inferotemporal sector of the neuroretinal rim as compared with the baseline. HEP FDF fields were stable. Physical examination of the optic nerves demonstrated no apparent change. She was scheduled for a follow up visit in six months.
In October 2014, she presented with IOPs of 32mm Hg OD and 28mm Hg OS. HEP FDF fields demonstrated a slight change in the superior arcuate area in the right eye, and a completely stable field in the left. The HRT 3 imaging demonstrated further progression of the change in the inferotemporal sector of the right nerve and no change in the left. The RNFL circle scan was stable as compared with the baseline. A slight continued change was noted in the neuroretinal rim appearance (Figure 1).
Discussion
It appears that both structural and functional changes are occurring in the patient’s right eye, whereas the left is remaining stable. We each have our own threshold-to-treat levels, but the evidence at this point is strong that the right eye is beginning to convert. Primarily based upon the patient’s relatively young age, I chose to defer treatment, on the outside chance that the changes were merely artifactual. While I didn’t necessarily believe this to be the case, I was not yet comfortable committing this patient to a lifetime of treatment. As such, I explained my findings to the patient, couching the conversation in the context of ‘appearing to develop glaucoma, but let’s be 110% certain before putting you on medications.’ Generally, patients are reluctant to begin medications without a compelling reason.
Follow Up
When the patient returned six months later, IOPs remained stable in both eyes, the FDF field remained stable, as did the OCT, but the HRT 3 imaging continued to demonstrate progression (Figure 2). At this point, I was convinced that I had enough evidence to warrant initiating therapy in the right eye. Accordingly, the patient was medicated with Travatan Z (travoprost, Alcon) HS in the right eye only. Her post-treatment IOP was 20mm Hg OD and 25mm Hg OS at 2pm two weeks after initiating therapy. One month following, her IOP was 21mm Hg OD and 26mm Hg OS. Reassured, it appears as though we are achieving a consistent reduction in IOP in the medicated right eye.
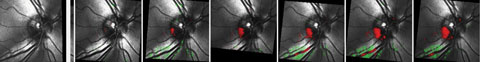 |
| Fig. 2. Progressive structural changes to the inferotemporal neuroretinal rim in the right eye. |
In August 2015, her IOP was measured at 20mm Hg OD and 24mm Hg OS. FDF fields demonstrated a slight increase in the field defect previously seen. The OCT remained stable. However, the HRT showed further change in the suspect area of the right optic nerve.
The possibility exists that the continued progression of the structural inferotemporal defect in her right eye may simply be a result of the time lapse from initiation of therapy until the HRT 3 image obtained in August 2015. But, it may also indicate that, although IOP seems to be responding nicely to Travatan Z, we might not necessarily be achieving enough of an IOP reduction to stave off further damage. As is always the case in managing glaucoma, it is imperative that we are certain that things are stable, both structurally and functionally, before we can reassure the patient that they are, in fact, stable. While I was comfortable in thinking that we were on the right track, I was not convinced that the situation was completely stable.
Of course, the benchmark in determining stability is the analysis of structural and functional testing, using whatever specific instruments you are comfortable with or have access to. Not surprisingly, I did not make any changes to her therapy at the August 2015 visit, but did ask her to return in December for repeat testing.
Medications
About one week after the August visit, the patient called the office mentioning that, due to changes in insurance coverage, her branded medication would have a much higher co-pay, and requesting a change to generic latanoprost. Understanding the reality that medications can be cost prohibitive and, subsequently, compliance prohibitive, I OK’d the change to latanoprost. However, this was an inopportune time to change medications, as I was still in the process of determining whether she was stable. I also had not been given ample time to determine what her IOP variances were post treatment.
Much would hinge on the December 2015 visit: if all was stable, structurally, functionally and with her IOP, then it would be reasonably prudent to continue with the generic medication and move forward with regularly scheduled visit. Conversely, if all was not stable, would it be because the new medication is not controlling IOP as well as the branded medication, or because the disease progressed despite reasonable IOP control?
Unfortunately, there appears to be continued progression of the structural defect in the December 2015 scan (Figure 2). And unfortunately, I am back to the same position I was a few months ago in assessing the stability of the situation. It does not appear to be stable. But is she unstable because of the new medication, or would the disease have progressed anyway? As of yet, I don’t have the answer to that crucial question.
But this case highlights the potential can of worms that may be opened when insurance companies, through financial strong-arming, dictate which medications a patient (reasonably and affordably) can take, independent of the clinical judgment of the prescribing physician. Just as I was sorting this case out (and many glaucoma cases take a rather lengthy time to ascertain stability), a wrench is thrown into the gears, requiring further evaluation.
As expected, the patient has been scheduled for yet another structural and functional series of tests to determine, yet still, stability.
Wouldn’t it have simply been better to just leave the patient and the doctor alone? I think so.
