 |
An 85-year-old Hispanic female presented to the office for a routine eye examination with a chief complaint of blurry vision. She had been told by another eye care practitioner that she had cataracts in both eyes, was a glaucoma suspect because her optic nerves were big and that she had a “birthmark” in the back of the right eye, which did not require treatment. Her systemic history was remarkable for hypertension and dyslipidemia, for which she was properly medicated. She denied ocular trauma and had no allergies to medications or environmental factors.
Clinical Findings
Her best-corrected entering visual acuities were 20/25 OD and 20/25 OS at distance and near with no improvement upon the use of pinhole or refraction. Her external examination was remarkable for metamorphopsia and constriction of the superior nasal periphery of the right eye upon facial Amsler, confirmed with Amsler grid. The left eye was normal. All other external findings were normal and there was no afferent pupillary defect.
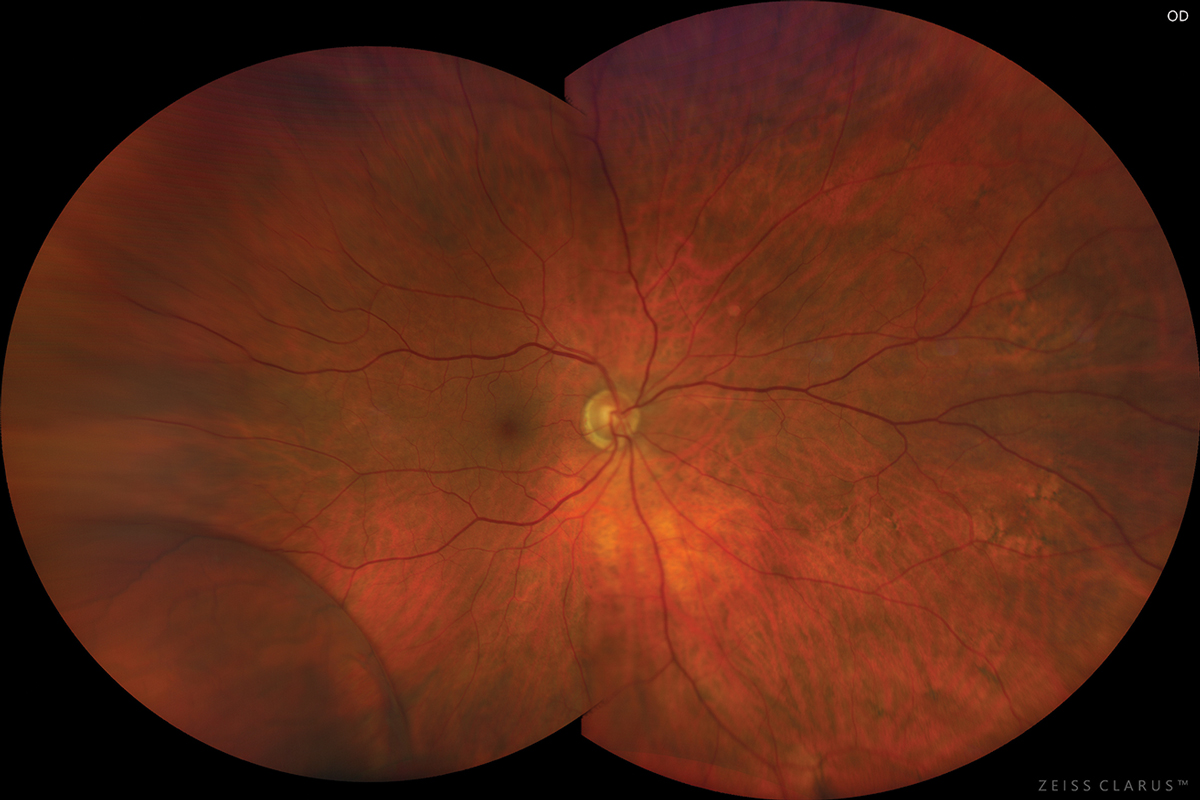
Biomicroscopy of both eyes found normal anterior segment structures and nuclear sclerotic cataracts OU. Her intraocular pressures measured 13mm Hg in both eyes by Goldmann applanation. The pertinent posterior segment finding OD is demonstrated in the photograph.
Her cup-to-disc ratios measured 0.5/0.5 OU with distinct pink rims. The retinal periphery of the left eye was unremarkable.
 |
|
This photo of a different patient with the same condition shows a comparable presentation. Photo: Jay M. Haynie, OD. Click image to enlarge. |
For More Info
Additional testing included OCT of both to discs to rule out nerve fiber layer loss consistent with glaucoma. OCT was performed on the retinal lesion as well to understand its anatomy. Photodocumentation of both posterior segments was completed. Comparison of photographic records records from the previous managing retina specialist confirmed that the lesion had a stable appearance and position since 2019. Automated perimetry could also be completed to understand the lesion’s impact on function. Finally, B-scan ultrasonography could also be completed if additional data were necessary.
What We Found
The diagnosis in this issue is bullous senile retinoschisis OD. Also known as degenerative or acquired retinoschisis, senile retinoschisis is typically a benign condition characterized by a horizontal separation of the retinal layers typically at the outer plexiform layer (where the axons of the photoreceptors are communicating with the dendrites of the horizontal, bipolar and amacrine cells).1-12
Senile retinoschisis of the peripheral retina typically remains stable and stationary in the majority of cases and therefore rarely produces symptoms or affects function.2,5,6 First described in 1933 by Bartels and later analyzed by Byer and colleagues, senile retinoschisis has no gender predilection, is typically bilateral and most commonly occurs in the inferotemporal retinal periphery.2,5-7,11-12
The underlying mechanism is poorly understood. The bullous form is thought to rise from coalescence of cystic changes in peripheral cystoid degeneration of the retina.4,6
Clinical Work-up
On fundus examination, senile retinoschisis is observed as a smooth and transparent area of elevation identified and bounded by a discrete, curved demarcation line.6,7,12 The roof of the cavity may appear flat or bullous.2,5-7 The overlying retinal vasculature may exhibit sclerosis, telangiectasia or aneurysmal changes.2,5-7 The “floating retinal vessels” inside the bullous zone often demonstrate a “full, firm” appearance, casting shadows on the underlying retinal layers.2,5-7,12 This can be useful to the clinician attempting differentiation from a bullous or rhegmatogenous retinal detachment (RRD).
Differentiating between retinoschisis and RRD can be difficult. It is important to use stereoscopic indirect biomicroscopic techniques, 3-mirror lens examination and binocular indirect ophthalmoscopic evaluation with and without scleral depression to carefully scan the elevated area for full-thickness retinal breaks. White with pressure (upon scleral indentation/depression) is a phenomenon seen when the underlying outer retina in RS is compressed.5-7 When scleral depression is done, the height of the schisis cavity will remain unchanged because there is no retinal break for the intra-cavital fluid to escape during indentation, whereas the cavity collapses in cases of RRD.2,5
Visual field testing can be used to differentiate retinoschisis from retinal detachment; RS shows an absolute visual field defect because “a split neurosensory retina” interrupts the signal from the photoreceptor into the visual pathyway.8 In a retinal detachment the neurosensory retina is intact, typically producing a relative field defect.8,9 In the unlikely circumstance a retinal detachment is highly elevated, chronic or anterior to the equator, an absolute visual field defect can be measured making this test invalid for differentiation.8,10
Posterior segment imaging such as color fundus photography, fundus autofluorescence and OCT are all useful tools for both diagnosing and monitoring retinoschisis.1,3 Standard color fundus photography is helpful for monitoring stability over time. Fundus autofluorescence demonstrates posterior lesion margin hyper-autofluorescence for RRD and hypoautofluorescence in RS.
Finally, the OCT of a senile retinoschisis demonstrates a thin inner neurosensory layer with bridging fibers connecting the inner and the outer retinal layers (varying in number between a lot and a few).8 The outer neurosensory layer attached to the RPE will show an irregular surface.8
Retinal detachment can occur; this is referred to as a “schisis-detachment.”1-12 They are rare and rarely progress beyond the posterior margin of the retinoschisis.2,5,6 These are not likely because it is theorized that the schisis cavity fluid is too viscous to easily migrate into the subretinal space through the outer layer retinal cavities (holes).2,5,6 Malagola and colleagues concluded that only one in 300 untreated asymptomatic schisis-detachment lesions progressed to symptomatic retinal detachments.2
Monitoring as Management
Since the vast majority of retinoschisis cases remain unchanged, asymptomatic and uncomplicated by RRD, most cases are monitored annually without treatment.1-12 Some cases spontaneously regress; there are some reports in the literature of posterior progression following prophylactic treatment.2,5,6
The management for “basic uncomplicated retinoschisis” is observation.1-10 For asymptomatic RS or lesions evolving into peripheral schisis-detachment with or without outer retinal layer holes, observation is recommended unless the case is considered high risk (i.e. evidence of progression on multiple visits).1-10
For progressive, symptomatic schisis-retinal detachment, treatment is required.2,5,6 Due to the rare nature of schisis-related complications, there is no widely agreed-upon gold standard treatment.2,5,6 There are a number of studied methods with varying degrees of measured success. These methods include scatter laser photocoagulation of the entire area of schisis followed by laser barrier at the posterior margin, scleral buckling, pneumatic retinopexy followed by laser retinopexy and pars plana vitrectomy with or without “unroofing” of the inner schisis retinal layer.1-10 The “unroofing” method may present an advantage by relieving traction without worsening functional vision.2,5-7 This is possible because retinoschisis already causes absolute scotoma in the affected area.2,5,6 Since treatment recommendations vary greatly between retinoschisis, schisis-detachment and symptomatic retinoschisis-related retinal detachment, correct and prompt diagnosis by a retinal specialist is key.
As this patient had “basic uncomplicated retinoschisis without symptoms,” observation was recommended by both our primary care team and the retina specialist at least once a year.
Dr. Gurwood thanks Dr. Korey Patrizi for contributing this case.
Dr. Gurwood is a professor of clinical sciences at The Eye Institute of the Pennsylvania College of Optometry at Salus University. He is a co-chief of Primary Care Suite 3. He is attending medical staff in the department of ophthalmology at Albert Einstein Medical Center, Philadelphia. He has no financial interests to disclose.
|
1. Navaratnam J, Salvanos P, Vavvas DG, Bragadóttir R. Ultra‐widefield autofluorescence imaging findings in retinoschisis, rhegmatogenous retinal detachment and combined retinoschisis retinal detachment. Acta Ophthalmol. 2020;99(2):195-200. 2. Ness S, Subramanian M L, Chen X, Siegel N H. Diagnosis and management of degenerative retinoschisis and related complications. Surv Ophthlamol. 2022;67(4):892-907. 3. Rachitskaya AV, Yuan A, Singh RP et al. Optical coherence tomography of outer retinal holes in senile retinoschisis and schisis-detachment. Br J Ophthalmol. 2016;101(4):445-8. 4. Slean GR, Fu A D, Chen J, Kalevar A. Neovascularization of the iris in retinoschisis. American Journal of Ophthalmology Case Reports. 2017: 7(1):99–101. 5. Xue K, Muqit MM, Ezra E, et al. Incidence, mechanism and outcomes of schisis retinal detachments revealed through a prospective population-based study. Br J Ophthalmol. 2017;101(8):1022-6. 6. Mandura RA. Acquired senile retinoschisis of the peripheral retina imaged by spectral domain optical coherent tomography. Cureus. 2021;13(4):e14540. 7. Byer NE. Clinical study of senile retinoschisis. Arch Ophthalmol. 1968;79(1):36-44. 8. Stehouwer M, Tan SH, van Leeuwen TG, Verbraak FD. Senile retinoschisis vs. retinal detachment, the additional value of peripheral retinal OCT scans (SL SCAN-1, Topcon). Acta Ophthalmol. 2014;92(3):221-7. 9. Kylstra JA, Holdren DN. Indirect ophthalmoscope perimetry in patients with retinal detachment or retinoschisis. Am J Ophthalmol. 1995;119(4):521-2. 10. Ip M, Garza-Karren C, Duker JS, et al. Differentiation of degenerative retinoschisis from retinal detachment using optical coherence tomography. Ophthalmology. 1999;106(3):600-5. 11. Über die Entstehung von Netzhautablösungen. Bartels M. Klin Mbl Augenheilkd. 1933;91:437. 12. Byer NE. Long-term natural history study of senile retinoschisis with implications for management. Ophthalmology. 1986;93(9):1127-37. |

