 |
The use of radiologic studies in the ophthalmic setting is becoming increasingly more common, especially for neuro-ophthalmic disorders. Many eye care providers now routinely order various types of neuroimaging studies that provide valuable and detailed information on neural visual pathways not easily obtained through clinical examination alone.
The most commonly ordered diagnostic test is magnetic resonance imaging (MRI) because it allows imaging of the orbital apex and optic nerve despite the dense bone surrounding these areas.1
Imaging Basics
The most common indications for neuroimaging are vision or visual field loss, pupil abnormalities, ptosis, proptosis, diplopia or ophthalmoplegia, nystagmus and certain optic disc abnormalities. MRI studies are based on the signal detection of the interaction between hydrogen molecules within a magnetic field.
When an MRI is ordered, both T1 and T2 studies are performed routinely for the imaging of the brain and orbits. Studies that are weighted towards T1 are best for the observation of normal anatomy, while T2 studies better distinguish pathology by enhancing the signaling differences and contrast in various tissues. In T1 images, fluids such as cerebrospinal fluid (CSF) and vitreous appear dark, or hypointense, whereas in T2-weighted images, fluids appear bright. For example, when viewing the CSF within the optic nerve sheath in suspected papilledema, a T2 image offers better contrast because it will appear hyperintense.
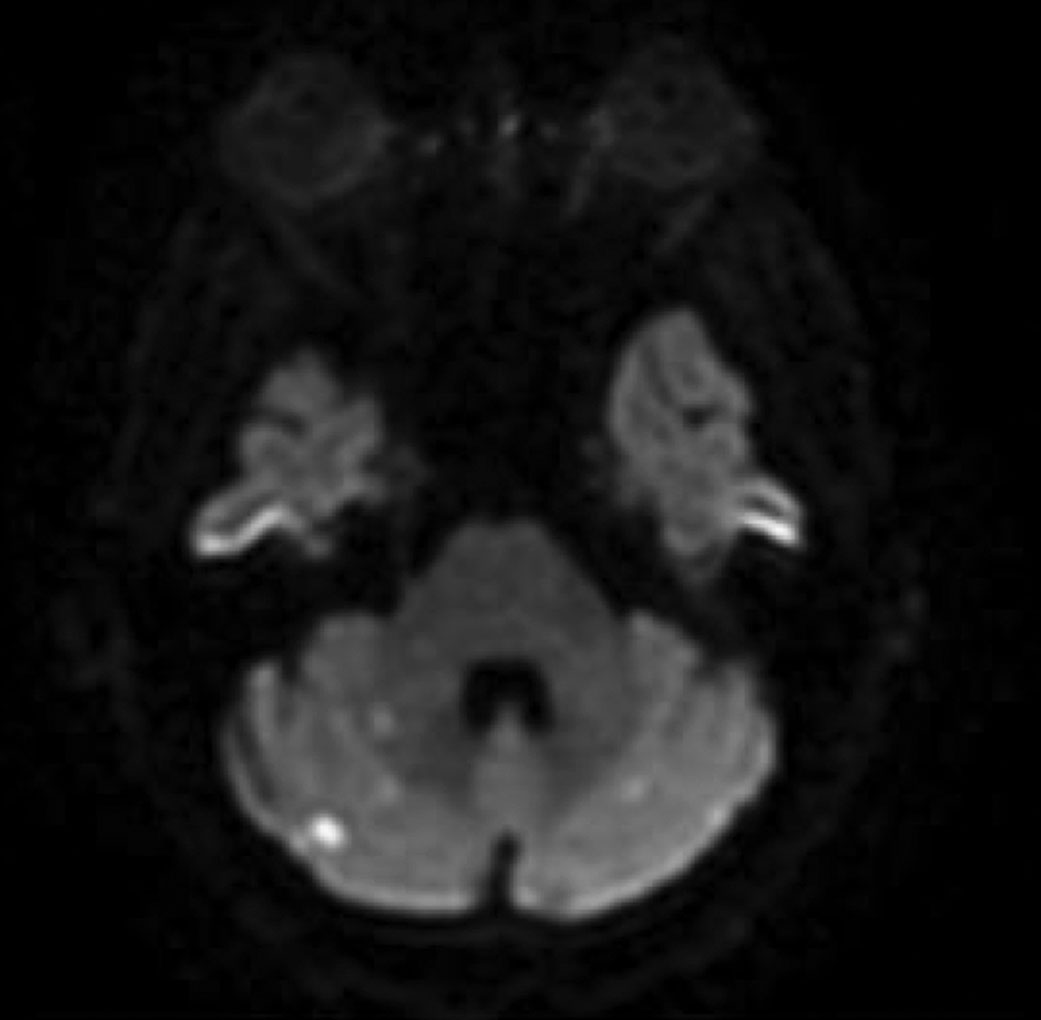 |
| DWI of the brain reflecting areas of hyperintensity that correlate with lesions of acute infarct. Click image to enlarge. |
MRI studies also include the use of contrast material, gadolinium, as well as specialized sequences such as fluid attenuation inversion recovery (FLAIR), fat suppression MRIs, gradient echo, magnetic resonance angiography and magnetic resonance spectroscopy (MRS) (Table 1). The different uses and sequences of MRI alone are vast, but the focus of this article will be on diffusion-weighted imaging (DWI) studies and their application in ophthalmic practice.2
DWI Drilldown
This sequence is obtained in most routine MRI brain examinations. DWI MRI works by detecting the free diffusion of water molecules. In a normal, healthy brain, there is free diffusion of water molecules along a concentration gradient, from regions of higher concentration to regions of lower concentration. A number of disease processes may impair this free motion, resulting in areas of restricted diffusion that appear “bright” on DWI.3
Although the details and the physical properties of the technique are complex, there are generally two equal but opposite gradient pulses applied to the tissue in question: if there is no free movement of water (i.e., restricted diffusion), the effects of the dephasing gradient and the equal but opposite rephrasing gradient cancel each other out, producing a hyperintense signal on DWI; if there is normal, free movement as in healthy tissue, the effects of the dephasing and rephrasing gradients do not cancel each other out and the tissue will produce an isointense signal on DWI.3,4
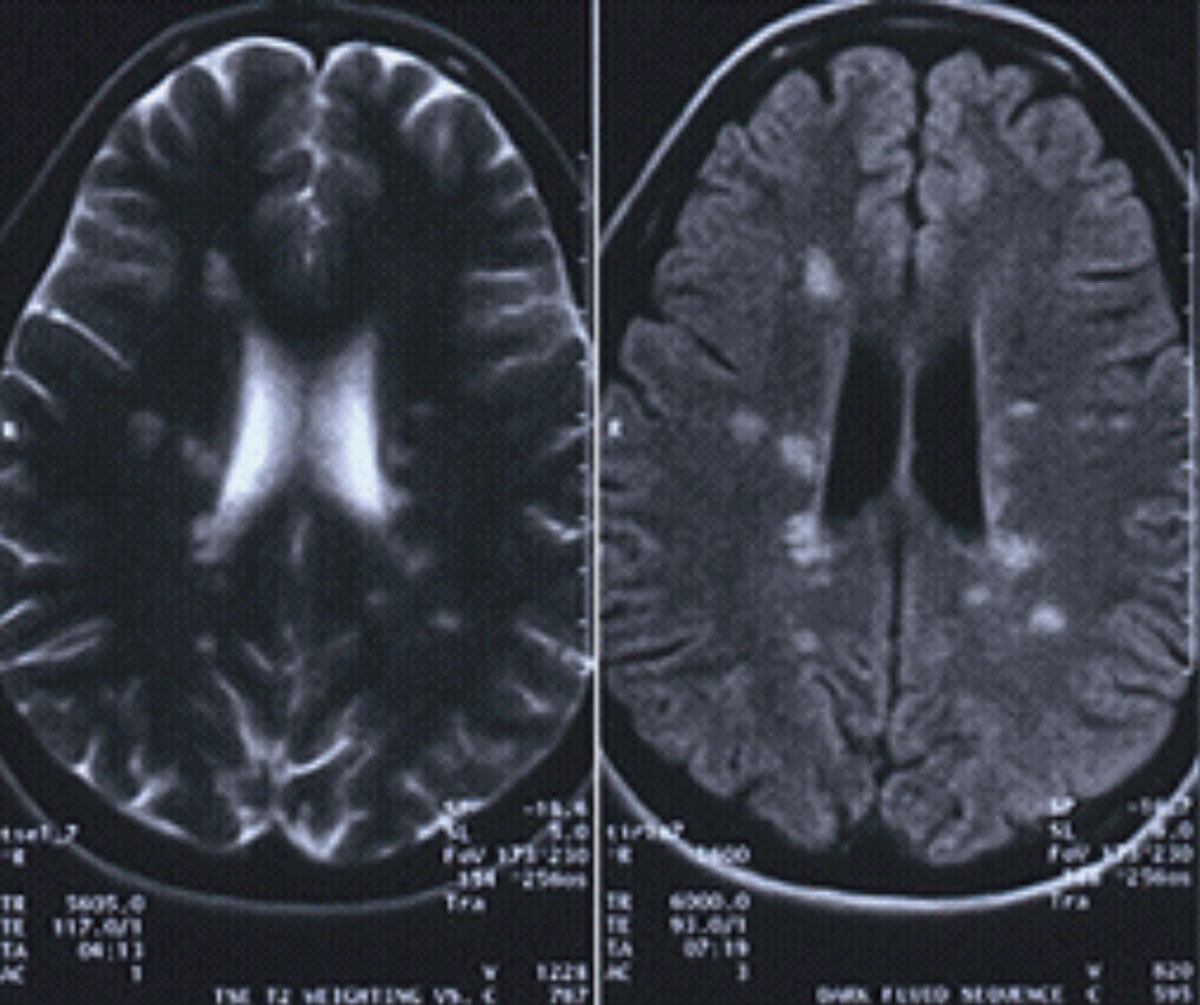 |
| This patient’s MRI reveals classic white matter lesions perpendicular to the lateral ventricles, consistent with MS. Image: Christopher L. Suhr, OD. Click image to enlarge. |
Clinical Applications
Restricted diffusion occurs in cytotoxic damage from ischemia, inflammation, trauma or tumor. Most commonly, the DWI sequence is used in the diagnosis and assessment of acute ischemic stroke.5 As vision and visual field loss can be the initial presenting sign of a cerebral infarct, clinicians should be familiar with this imaging modality and be able to order and understand its basic principles, as this is often required in emergent cases. The DWI can identify acute infarct as early as three to five minutes following the onset of clinical symptoms and remains positive for up to 14 days.5-7 This makes it a crucial tool to determine if thrombolytic therapy should be administered, where timeliness is an essential factor in stroke treatment.5
Acute strokes appear as high signal, bright lesions on DWI. These areas correspond with a hypoperfused brain. As such, when this area regains perfusion, DWI abnormalities may reverse. This is referred to as pseudonormalization because the disappearance of the DWI signal does not imply a return of normal cellular function—that will remain abnormal on conventional MRI. Therefore, chronic infarcts will appear hypo or isointense on DWI.5
Table 1. Brief Summary of Different MRI Sequences | |
| MRI Sequence | General Use |
| T1 “fat suppression” | Visualization of orbit |
| FLAIR | Demyelinating disease |
| Gradient echo | Hemorrhage in patients with underlying vascular malformations, intracerebral hemorrhage and traumatic brain injury |
| MRI with contrast (gadolinium) | Identification of areas where there is breakdown of the blood-brain barrier |
| MRS | Diagnosis and evaluation of treatment response of brain tumors |
| DWI | Hyperacute ischemic lesions, areas of cytotoxic edema |
Additional Ophthalmic Uses
The utility of DWI in acute stroke diagnosis and management is well studied and understood. In addition to these cases, DWI can be useful in other areas of ocular disease. Specifically, DWI is helpful in distinguishing an arachnoid cyst from an epidermoid tumor. Normally, these lesions have similar features on T1 and T2 MRIs. However, arachnoid cysts contain cerebrospinal fluid, whereas epidermoid tumors are solid masses. This results in a higher DWI signal on solid tumors, where water mobility is low or more restricted.2,5
Cerebral abscesses are also readily identified on DWI because there are often central areas of cellular necrosis, cysts or both. These areas restrict water movement and result in bright signals on DWI. Additionally, blood products that may be subtle on conventional MRI contribute to low signal intensity on DWI, aiding in its identification.5
Acute white matter changes in multiple sclerosis (MS) also appear on DWI. While they will also arise on conventional MRI and FLAIR, the DWI is particularly helpful in dating MS plaques, which is useful when evaluating responses to therapy.5
Finally, in cases of central retinal artery occlusion (CRAO), DWI studies reveal multiple areas of restricted diffusion in affected retinas, suggestive of infarct.8 One study also found a correlation between the severity of papilledema and optic nerve hyperintensity on DWI in patients with idiopathic intracranial hypertension (IIH). These findings suggest a possible surrogate marker for the identification and severity of papilledema secondary to IIH.9
DWI is an essential tool in the evaluation of a patient presenting emergently with acute vision loss. Clinicians must understand the principles of this unique sequence to apply it in practice—and properly identify everything from stroke and tumors to MS and CRAO.
1. Kakaria AK. Imaging in neuro-ophthalmology: An overview. Oman J Ophthalmol. 2009;2(2):57-61 2. Lee AG, Brazis PW, Garrity JA, White M. Imaging for neuro-ophthalmic and orbital disease. Am J Opthalmol. 2004;138(5):852-62. 3. Schaefer PW, Grant PE, Gonzalez RG. Diffusion-weighted MR imaging of the brain. Radiology. 2000;217(2):331-45. 4. Bammer R. Basic principles of diffusion-weighted imaging. European J Radiol. 2003;45(3):169-84. 5. Mukherji SK, Chenevert TL, Castillo M. Diffusion-weighted magnetic resonance imaging. J Neuro-Ophthalmol. 2002;22(2):118-22. 6. Allen LM, Hasso AN, Handwerker J, Farid H. Sequence-specific MR imaging findings that are useful in dating ischemic stroke. Radiographics. 2012;32(5):1285-97. 7. Reith W, Hasegawa Y, Latour LL, et al. Multislice diffusion mapping for 3-D evolution of cerebral ischemia in a rat stroke. Neurology. 1995;45(1):172-7. 8. Alsinaidi O, Shaikh AG. Diffusion-weighted magnetic resonance imaging in acute retinal pathology. Neuro-Ophthalmology. 2018;42(3):191-93. 9. Salvay DM, Padhye LV, Huecker JB, et al. Correlation between papilledema grade and diffusion-weighted magnetic resonance imaging in idiopathic intracranial hypertension. J Neuro-Ophthalmol. 2014;34(4):331-5. |

