Bright Ideas for Better ExamsThe February 2023 issue of Review of Optometry is our annual diagnostic skills and techniques issue. In each article, eyecare experts offer practical tips and advice to help you brush up on exam techniques that you can put to use everyday in your office. Check out the other articles featured in this issue:
|
As it stands, relatively few patients are diagnosed with keratoconus in the modal eyecare practice, even though we now know the disease is far more prevalent than previously thought. New technologies allow for earlier and more frequent diagnosis. Given there are now enhanced treatment alternatives, including modern scleral contact lenses and corneal crosslinking (CXL), early detection represents an imperative. With such advances come new trends, challenges and controversies. Here, we investigate five in particular.
Challenge:
Early Identification an Imperative
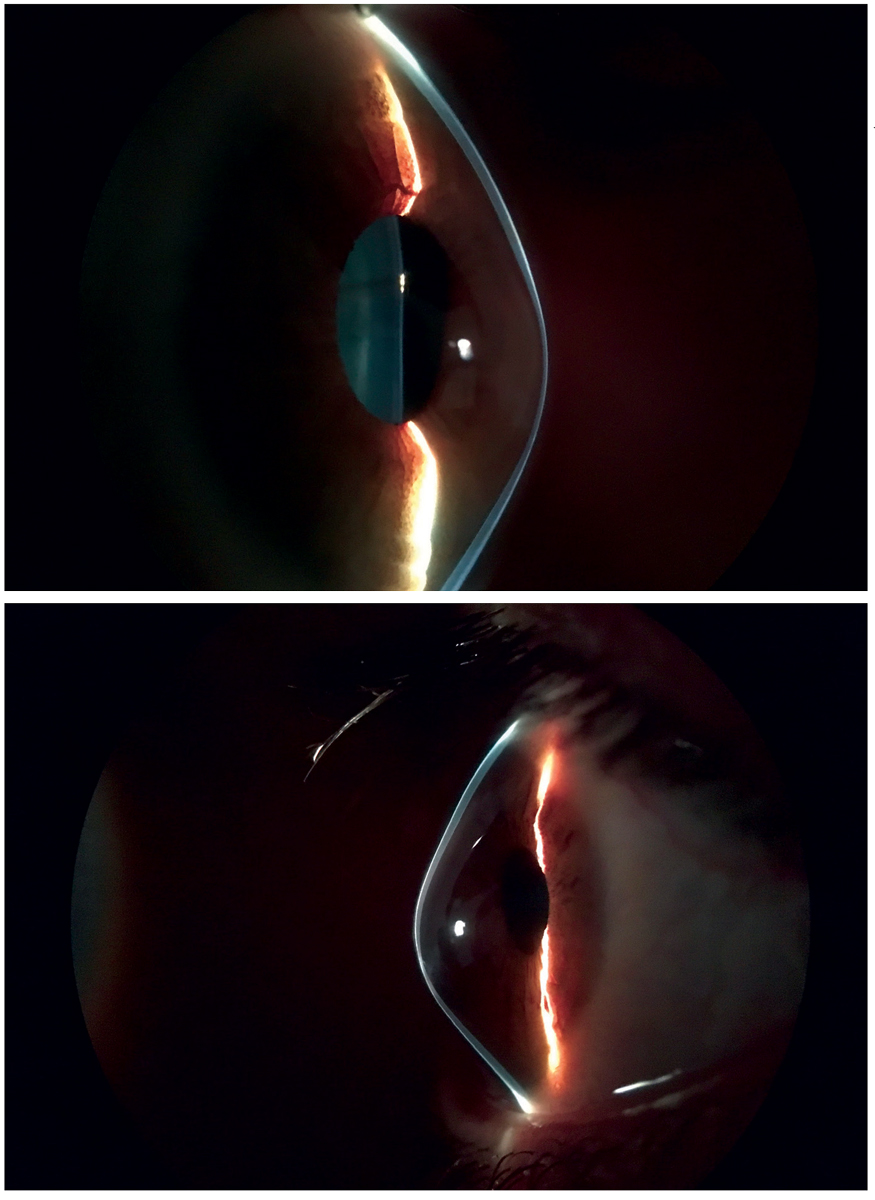
A prompt diagnosis during adolescence is more important than ever because this is the critical time when CXL has the strongest indication to prevent the most progression. In contrast, if a patient already displays Munson’s sign with a spectacle-corrected visual acuity (VA) of count fingers, a surgeon may feel these eyes are beyond the time to benefit from CXL (Figure 1). This would be analogous to how, in myopia management, it is valuable to initiate low-dose atropine therapy or peripheral defocus optical correction for a child with -1.00D myopia yet perhaps not so much for an adult with -12.00D myopia.
Testing for genetic risk of keratoconus was commercially introduced in February of 2020 and marketed as an important method to gain early diagnostic certainty. Genetic testing has not yet achieved widespread clinical acceptance as a useful standalone predictive test.1 Instead, tried-and-true clinical metrics continue as the basis for clinician keratoconus diagnoses.
There may be an emerging role for data analytics and artificial intelligence in improving a clinician’s ability to diagnose keratoconus. In the past, various indices were proposed to aid in this capacity, including the KISA% index based on corneal topography.2 Today with improved computational technologies, a neural network could be trained with databases of clinical metrics of those with and without keratoconus. The metrics could include best-spectacle corrected VA, intraocular pressure, corneal hysteresis, corneal thickness, corneal topography or tomography, aberrometry and pachymetry, as well as demographic data including age, race, family history of keratoconus, history of eye itching with severity and laterality, presence of allergy, asthma and eczema. A data-driven risk assessment of keratoconus may prove more sensitive and specific than that of an average clinician. Even so, the entry burden of multiple data fields may make real-time diagnosis using data analytics impractical. The greatest promise may require incorporation into electronic medical records or retrospective application to an existing patient electronic medical record database.
 |
|
Fig. 1. Consider whether CXL has a compelling indication in a patient with Munson’s sign. Photo: John Gelles, OD. Click image to enlarge. |
One of the most effective pathways for new keratoconus diagnosis is through LASIK consults. Since the approval of LASIK in 1999, screening for surgical candidacy has fueled diagnosis for many new cases of keratoconus. Studies found between 6.4% to 9.6% of prospective LASIK patients were ruled as non-candidates due to probable keratoconus, whereas an estimated one in 375 in the general population are expected to have keratoconus.3-6 Corneal topography or tomography, conducted across-the-board during LASIK consults, is arguably the single-most efficacious measurement to help a practitioner diagnose keratoconus. This unintended contribution to new keratoconus identification, however, is not optimized for early identification. Adolescents are non-candidates for LASIK, so they do not present for LASIK consultations.
This prompts the question, “Should corneal topography be part of a routine comprehensive eye examination?” For early diagnosis, standard inclusion of corneal topography may be a positive step for early detection of keratoconus; even so, it is not required now for a reasonably prudent practitioner. Furthermore, none of the major vision plans require a corneal topographer to join or stay on their panels. There are forces in play that may change this, creating new incentives that should lead to improved and widespread keratoconus screening. In the meantime, early identification of keratoconus in adolescents and young adults who may progress rapidly remains a challenge.
 |
|
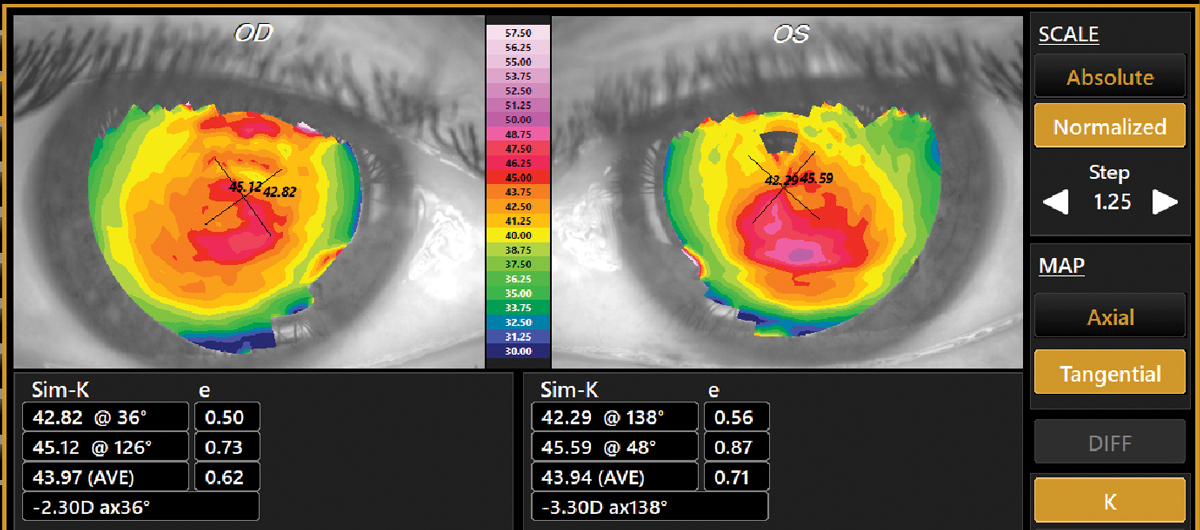
Fig. 2. Tangential topography of a 53-year-old male, which confirmed a first-time diagnosis of keratoconus. Best-spectacle corrected visual acuity was 20/20- in each eye with -0.50-2.00X038 OD and -0.50-2.50X137 OS. This was not “late onset” KCN but longstanding keratoconus that was not detected at any of his prior eye exams. Click image to enlarge. |
Challenge and Trend:
Improving and Expanding Keratoconus Screening
The corneal topographer is a foundational instrument for early identification, diagnosis and monitoring keratoconus rapidly and accurately, in the same manner the blood pressure cuff serves in identification, diagnosing and monitoring hypertension. Even so, not all practitioners have incorporated corneal topographers in their practices. One dynamic that may inspire movement toward adopting topography as part of routine examination is that there is now liability exposure for not diagnosing keratoconus and referring progressing disease for CXL in a timely manner. Sometime after the April 2016 FDA approval of the first CXL system, timely referral for this procedure became the standard of care for progressing keratoconus.
Today, more than 95% of commercial medical insurers provide some coverage for CXL.7 If damage to an eye or vision loss occurs proximate to failure to diagnose and/or refer in a timely manner, the practitioner has heightened exposure to claims of negligence. The mere fact that medicolegal claims of this nature already exist foreshadows increasing practitioner medicolegal liability when not diagnosing keratoconus in a timely manner. Incorporating corneal topography as part of routine examination can mitigate this risk by improving the practitioner’s ability to diagnose and monitor keratoconus. One large vision provider network has already incorporated instrumentation, combining topography and wavefront aberrometry as a routine element of their comprehensive eye examination.8 Combined corneal topography and aberrometry is expected to have greater power in diagnosing keratoconus than corneal topography alone.
Other factors driving increased keratoconus screening include heightened public awareness and patients’ desire to be screened. Over the past decade, professional sports stars, including NBA great Steph Curry and MLB outfielder Tommy Pham, have openly discussed and raised public recognition of keratoconus.9,10 Perhaps someday patients will commonly ask if their eye exam detects keratoconus in the same manner they ask if they have cataract, glaucoma or macular degeneration.
Finally, there is a possibility that a judicial ruling establishes topography as part of the comprehensive eye exam. This would be similar to the 1974 Helling v. Carey case, in which the Supreme Court of Washington held that tonometry was a necessary part of the routine eye exam not only for patients age 40 and older, but for all.11 However, most practitioners might prefer that their industry generate clinical care guidelines rather than leaving it to a judge or legislative body without clinical understanding and experience.
 |
|
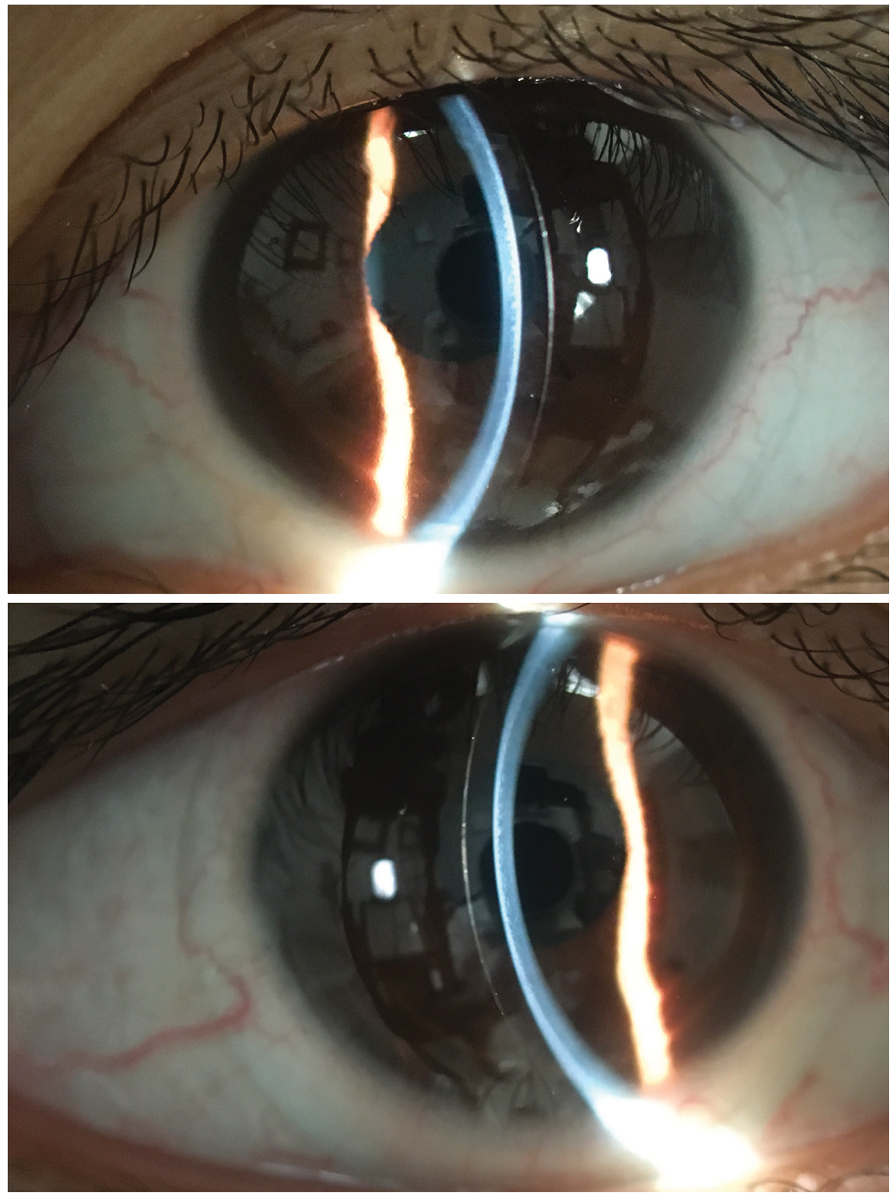
Fig. 3. Optic sections of ultrahigh Dk custom soft contact lenses for keratoconus. Click image to enlarge. |
Controversy:
Is Concern about Progression Overstated?
It is well understood that keratoconus is expected to progress during adolescence to early twenties. This warrants the attention of all clinicians to ensure a timely referral for CXL to arrest disease progression. Yet for a keratoconus patient who has reached presbyopia and beyond, is concern for progression warranted and is there a concomitant over-referral for CXL?
Keratoconus is understood to stabilize and arrest on its own by the third to fourth decade of life.12 Although keratoconus can have a later onset, the probability of late onset keratoconus is low. Cases of late onset keratoconus may in fact represent pre-existing disease that was not identified sooner (Figure 2). In some cases, corneal gas permeable (GP) contact lens wear may mask the diagnosis of keratoconus by confounding the topographical pattern through epithelial redistribution. If a corneal GP washout is not complete, the change in topographical appearance may masquerade as progressing keratoconus, whereas the cornea may, in fact, be returning to its natural shape.
Diagnosis of keratoconus later in life may correlate with milder and stable corneal distortion. Form fruste disease may escape detection due to the lack of suspicious findings. By the time classic clinical signs of keratoconus are detectable—Munson’s sign, Vogt’s striae and Fleischer rings—the keratoconus has existed for years. It is expected that a significant number of keratoconus patients diagnosed using these classic clinical signs may already have achieved stability.
With today’s emphasis on carefully monitoring for progressive corneal distortion, it is reasonable to wonder if CXL is worthwhile when the keratoconus may have stabilized on its own. Unnecessary CXL may represent an alpha risk of increased health care cost and complications from the CXL vs. beta risk of progression by a failed prognostication of nonprogression.
Standard epi-off CXL is not without its risks, with one paper citing a complication rate of between 1% to 10%.13 A large-scale review of 2,025 eyes undergoing accelerated CXL found haze formation in 9.1% in the early postoperative period and failure of treatment in 4.2% in the late period, with other common complications including loss of two or more Snellen lines in 2.4% and delayed epithelial healing in 1.8%.14 There are also rare reports of corneal melting and perforation after CXL.15
Even with this understanding of complication risk, practitioners and patients with keratoconus may take comfort in knowing that the overall rate of corneal transplantation has already plummeted by fivefold with the success of scleral contact lenses.16 Inducing fear during the counseling of keratoconus patients who are beyond their 40s to stimulate them to believe they may progress to corneal transplantation and that CXL prevents corneal transplantation may present an alpha risk for the patients that is not reasonable. A conservative approach for these seemingly late onset cases may include serial monitoring to confirm stability.
A standard of care is suggested for the intentional use of CXL that is moderated by age of onset and clinical metrics to support progression or the lack thereof, rather than an across-the-board application of CXL for all that are newly diagnosed. The personalized recommendation for intentional CXL would strike a balance between alpha and beta risk.
 |
|
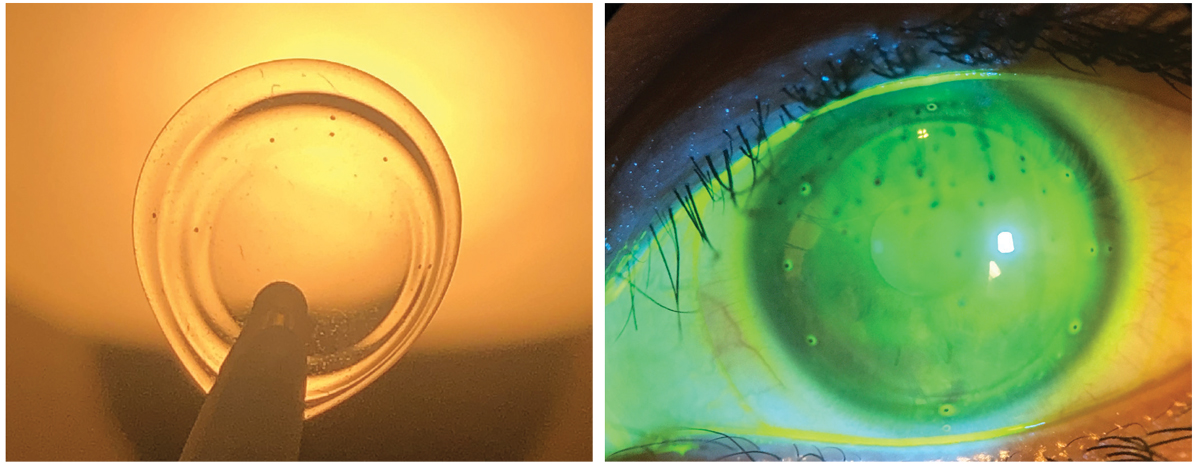
Fig. 4. Lens with registration marks at left. Same rotationally and translationally stabilized ultrahigh Dk soft lens on eye for wavefront-guided higher-order aberration correction for keratoconus. Click image to enlarge. |
Controversy:
Corneal Crosslinking or Contact Lens Treatment First?
For a newly diagnosed patient with keratoconus and poor best spectacle-corrected VA, is the first order treatment CXL or contact lens prescribing? There is no practitioner consensus yet; rather, the treatment plan is managed on a case-by-case basis with differences from one practitioner to another. It is reasonable to pursue both concurrently for many keratoconus patients since CXL is not expected to normalize the shape of the cornea sufficiently to reduce the higher order aberrations that impact best spectacle-corrected VA.
It is worth noting that medical insurance coverage for CXL may take months to even over a year to obtain, depending on administrative burden and criteria needed to demonstrate to the third-party payer to establish progression. It is advantageous that during this time, the patient benefits from vision improvement via contact lens prescribing and gains proficiency with lens handling. These adolescent patients need vision correction to perform well in the classroom and on the sports field. In lieu of being held in limbo waiting for the CXL approval, the patient benefits from contact lens wearing and the restoration of their visual performance immediately. Practitioners report that most patients in sclerals prior to CXL can wear the same lenses following surgical recovery. This observation suggests that it is reasonable to expect that contact lens re-prescribing may not be required after CXL.
Keratoconus patients on some vision care plans should know that undergoing CXL can potentially reduce their corneal powers below the threshold for reimbursement.17 One major plan sets a 53.00D steep keratometry criteria for the stepped-up necessary contact lens reimbursement for scleral prescribing. In the event of a reduced post-surgical steep keratometry meridian power, these patients may receive a surprise bill for out-of-pocket costs for subsequent scleral lens prescribing. At the same time, the same patient may enjoy peace of mind by minimizing the risk of disease progression. To avoid this coverage issue, initiating contact lens prescribing before CXL may be financially advantageous for the patient who may be teetering close to the third-party vision plan threshold for scleral lens reimbursement.
A practitioner prescribing contact lenses before CXL should know that corneal gas permeable lenses or hybrid contact lenses may confound the surgeon’s ability to assess the pre-treatment topography and the post treatment progression. Soft lenses and sclerals impact corneal shape less and are expected to cause less interference with topographical and tomographic evaluation for disease progression.
Challenge and Emerging Trend:
Wavefront-Guided Contact Lenses
Retrospective medical records review and prospective predictions conclude that custom soft lenses may be the lens of choice for sizeable number with keratoconus.18 Presently, custom soft contact lenses indicated for keratoconus have increased thickness and deliver oxygen transmissibility below the Holden-Mertz criteria for daily wear. They are lathe cut and have a high cost of goods, making disposability infeasible. New ultrahigh Dk lens materials and molding methods may deliver higher oxygen transmissibility along with reduced cost of goods that might allow a monthly disposable modality (Figure 3).
Also keep in mind that increased adoption of aberrometers by eyecare practitioners and software for converting simultaneous aberrometry and registration data to lens manufacturing is enabling wavefront-guided scleral contact lenses and custom soft lenses that manage the higher order aberrations that commonly reduce VA in keratoconus (Figure 4). The ultimate achievement of improved best corrected contact lens VA is anticipated.
Takeaways
Over the past 25 years, keratoconus treatment has advanced tremendously. While corneal gas permeable contact lenses and penetrating keratoplasty have ongoing roles for long-term keratoconus management, their frequency of use is muted today. This is largely due to the dramatic success of scleral lenses in restoring vision and collagen crosslinking in minimizing disease progression. New custom wavefront-guided soft lenses for keratoconus may emerge as a viable alternative for mild to moderate keratoconus.
Unfortunately, the greatest challenge from a public health perspective is that far too many adolescents and young adults with keratoconus remain undiagnosed, and there remains a cohort with known keratoconus who have not yet received the optimized treatment available today.
Dr. Chou practices at ReVision Optometry, a referral clinic for keratoconus and scleral lenses in San Diego. He reported the first US case of Intacs for keratoconus and is a past recipient of the National Keratoconus Foundation’s Top Doctor award. He has no relevant financial interests to disclose.
Dr. Legerton is the co-founder of SynergEyes, Innovega, Prolign Technologies and Ocular Surface Innovations. He is an inventor of more than 200 US and international patent cases and is honored with the American Optometric Association Outstanding Achievement Award, the American Academy of Optometry Founders’ Award, the Contact Lens Manufacturers Association Trailblazers Award and the Orthokeratology Academy of America Achievement Award.
1. Cole J. KCN genetic testing: where does it fit in? RCCL. 2022;159(1):10-3. 2. Rabinowitz YS, Rasheed K. KISA% index: a quantitative videokeratography algorithm embodying minimal topographic criteria for diagnosing keratoconus. J Cataract Refract Surg. 1999;25(10):1327-35. 3. Xu K, McKee HD, Jhanji V. Changing perspective of reasons for not performing laser-assisted in situ keratomileusis among candidates in a university eye clinic. Clin Exp Optom. 2013;96(1):20-4. 4. Sharma N, Singhvi A, Sinha R, Vajpayee RB. Reasons for not performing LASIK in refractive surgery candidates. J Refract Surg. 2005;21(5):496-8. 5. Hori-Komai Y, Toda I, Asano-Kato N, Tsubota K. Reasons for not performing refractive surgery. J Cataract Refract Surg. 2002;28(5):795-7. 6. Godefrooij DA, de Wit GA, Uiterwaal CS, Imhof SM, Wisse RP. Age-specific incidence and prevalence of keratoconus: a nationwide registration study. Am J Ophthalmol. 2017;175:169-72. 7. Insurance coverage for iLink corneal crosslinking. Glaukos. www.glaukos.com/cornea/reimbursement. Accessed January 17, 2023. 8. Clarifye: the digital eye exam that gives more information. LensCrafters. www.lenscrafters.com/clarifye. Accessed January 17, 2023. 9. Horsting L. Steph Curry, the NBA’s greatest shooter ever, has had vision problems his whole career. WarriorsWire. warriorswire.usatoday.com/2019/04/03/steph-curry-the-nbas-greatest-shooter-ever-has-had-vision-problems-his-whole-career. April 3, 2019. Accessed January 17, 2023. 10. Kolakowski R. Rays’ Tommy Pham shares story of eye condition that threatened career. Tampa Bay Times. www.tampabay.com/sports/rays/2019/10/16/rays-tommy-pham-shares-story-of-eye-condition-that-threatened-career. October 16, 2019. Accessed January 17, 2023. 11. Ruskiewicz JP, Godio L, Myrowitz E, France L. Cost and quality implications of changes in tonometry use by optometrists. J Am Optom Assoc. 1983;54(4):339-44. 12. Gordon-Shaag A, Millodot M, Shneor E, Liu Y. The genetic and environmental factors for keratoconus. Biomed Res Int. 2015;2015:795738. 13. Seiler TG, Schmidinger G, Fischinger I, et al. Komplikationen der vernetzung der hornhaut [Complications of corneal cross-linking]. Ophthalmologe. 2013;110(7):639-44. German. 14. Çakmak S, Sucu ME, Yildirim Y, et al. Complications of accelerated corneal collagen crosslinking: review of 2,025 eyes. Int Ophthalmol. 2020;40(12):3269-77. 15. Tillmann A, Kampik D, Borrelli M, et al. Acute corneal melt and perforation—a possible complication after riboflavin/UV-A crosslinking (CXL) in keratoconus. Am J Ophthalmol Case Rep. 2022;28:101705. 16. Ling JJ, Mian SI, Stein JD, et al. Impact of scleral contact lens use on the rate of corneal transplantation for keratoconus. Cornea. 2021;40(1):39-42. 17. Chou B. New tools to tame keratoconus. RevOptom. 2019;156(4):42-9. 18. Gelles JD. Contact lenses for visual rehabilitation in keratoconus: characteristics and outcomes of contact lenses for keratoconus an evidence-based fitting algorithm. Presented at Global Specialty Lens Symposium 2020. |


